Revolutionizing Phosphine Oxide Manufacturing: A Green, Solvent-Free Catalytic Route for High-Purity Intermediates
The chemical industry is currently witnessing a pivotal shift towards sustainable manufacturing paradigms, particularly in the synthesis of high-value organophosphorus compounds which serve as critical ligands and intermediates in pharmaceutical development. Patent CN109678901B introduces a groundbreaking methodology for the synthesis of trisubstituted phosphine oxide compounds that fundamentally alters the traditional approach to C-P(O) bond formation. This innovation leverages a halosilane-catalyzed dehydration strategy, replacing hazardous alkyl halides with benign, widely available alcohols as the alkylating agents. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes while adhering to increasingly stringent environmental regulations. The core of this technology lies in its ability to directly couple dialkyl phosphine oxides with monohydric or dihydric alcohols under mild, solvent-free conditions, generating water as the sole byproduct. This not only simplifies the purification workflow but also drastically reduces the E-factor of the process, making it an attractive candidate for the green manufacturing of complex API intermediates and specialized ligands required in asymmetric catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of trisubstituted phosphine oxides has relied heavily on the Michaelis-Arbuzov reaction, which necessitates the use of toxic and often unstable alkyl halides as electrophiles. These traditional pathways are fraught with significant operational hazards, including the generation of micromolecular halohydrocarbon byproducts that pose severe environmental pollution risks and require complex waste treatment protocols. Furthermore, alternative transition-metal catalyzed coupling methods, while effective, introduce substantial cost burdens due to the requirement for expensive precious metal catalysts, specialized ligands, and large excesses of base. These factors collectively contribute to a cumbersome downstream processing burden, where the removal of trace metal residues becomes a critical bottleneck for pharmaceutical applications. Additionally, previous attempts to utilize alkoxyphosphines have been hindered by their extreme sensitivity to moisture and air, leading to poor shelf-life and the formation of unwanted self-rearrangement byproducts that compromise the purity of the final active ingredient.
The Novel Approach
In stark contrast to these legacy methods, the novel halosilane-catalyzed dehydration route offers a streamlined, atom-economical solution that bypasses the need for toxic halides or expensive transition metals. By utilizing stable dialkyl phosphine oxides and common alcohols, the process achieves high selectivity under remarkably mild thermal conditions, typically ranging from 80°C to 120°C. The absence of solvent not only reduces raw material costs but also enhances the reaction concentration, driving the equilibrium towards product formation through the continuous removal of water. This methodology demonstrates exceptional versatility, accommodating a broad spectrum of substrates including benzyl, allyl, and aliphatic alcohols, as well as diols for the synthesis of bis-phosphine oxides. The visual representation of this general transformation highlights the simplicity and elegance of the direct dehydration mechanism.
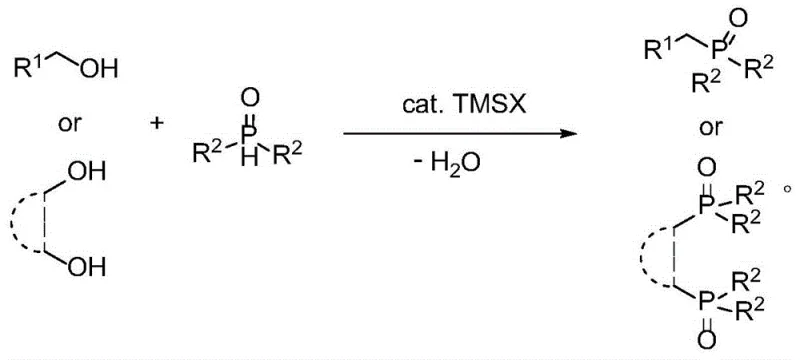
The strategic implementation of this catalytic system allows for the direct construction of the C-P(O) bond with minimal waste generation. Unlike the stepwise approaches reported in prior literature, which suffer from low atom economy and lengthy reaction times, this one-pot procedure consolidates the synthesis into a single efficient operation. The use of trimethylsilyl iodide (TMSI) as the catalyst of choice has been shown to activate the hydroxyl group of the alcohol effectively, facilitating the nucleophilic attack by the phosphine oxide without the need for harsh activating agents. This results in a cleaner reaction profile, significantly reducing the burden on purification teams and enabling the production of high-purity intermediates suitable for sensitive downstream applications in drug discovery and materials science.
Mechanistic Insights into Halosilane-Catalyzed Dehydration
The mechanistic underpinning of this transformation involves the activation of the alcohol substrate by the halosilane catalyst, likely forming a transient silyl ether intermediate that is more susceptible to nucleophilic displacement by the phosphine oxide. The phosphorus center, acting as a nucleophile, attacks the activated carbon center, leading to the formation of the new carbon-phosphorus bond and the subsequent elimination of the silanol species which condenses to release water. This catalytic cycle is highly efficient because the halosilane is regenerated or acts in a turnover capacity that minimizes catalyst loading requirements. Detailed experimental optimization revealed that the nature of the halogen on the silicon atom plays a crucial role in catalytic efficiency, with iodine providing superior leaving group ability compared to chlorine or bromine. This subtle electronic effect translates into dramatic differences in reaction outcomes, where TMSI consistently outperforms other halosilanes in terms of both conversion rate and isolated yield.
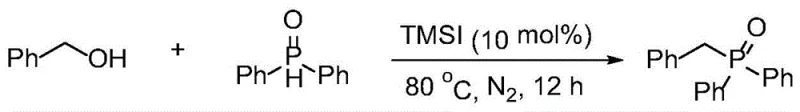
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of phosphonium salts or metal-complexed side products that are common in transition-metal catalysis. The stability of the dialkyl phosphine oxide starting material ensures that the reaction proceeds without the competing self-rearrangement pathways observed with alkoxyphosphines. This inherent stability simplifies the impurity profile, making it easier to meet the rigorous specifications required for GMP manufacturing. Furthermore, the reaction tolerance extends to various functional groups, allowing for the synthesis of diverse phosphine oxide derivatives without the need for extensive protecting group strategies. The ability to tolerate heteroaryl systems, such as thiophenes, and fused ring systems like naphthalenes, underscores the robustness of this catalytic system for constructing complex molecular architectures found in modern medicinal chemistry.
How to Synthesize Benzyldiphenylphosphine Oxide Efficiently
The synthesis of benzyldiphenylphosphine oxide serves as a benchmark reaction to demonstrate the efficacy of this novel protocol. By combining diphenylphosphine oxide with benzyl alcohol in the presence of a catalytic amount of trimethylsilyl iodide, chemists can achieve near-quantitative yields under solvent-free conditions. The procedure is operationally simple, requiring only the mixing of reagents under an inert atmosphere followed by heating, which makes it highly amenable to automation and scale-up. For detailed standard operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Charge a reactor with diphenylphosphine oxide and the desired alcohol (benzyl, allyl, or aliphatic) under an inert nitrogen atmosphere.
- Add the halosilane catalyst, specifically trimethylsilyl iodide (TMSI), at a loading of 10 mol% to initiate the dehydration coupling.
- Heat the reaction mixture to 80°C without solvent for 12 hours, then purify the resulting trisubstituted phosphine oxide via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this halosilane-catalyzed technology presents a compelling value proposition centered on cost efficiency and supply security. The substitution of expensive and toxic alkyl halides with commodity alcohols significantly lowers the raw material cost base, while the elimination of transition metal catalysts removes the need for costly scavenging resins and specialized waste disposal services. This shift not only reduces the direct cost of goods sold but also mitigates regulatory risks associated with heavy metal limits in pharmaceutical products. The simplified workflow, characterized by solvent-free conditions and aqueous byproducts, translates into reduced energy consumption for solvent recovery and distillation, further enhancing the overall economic viability of the manufacturing process.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost reagents with inexpensive, bulk-available alcohols and the removal of precious metal catalysts from the bill of materials. By eliminating the need for ligands and strong bases, the process reduces the complexity of the reaction mixture, which in turn lowers the consumption of chromatography media and solvents during purification. This streamlined approach results in substantial cost savings across the entire production lifecycle, from raw material acquisition to final product isolation, making it a highly competitive route for large-scale manufacturing.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous starting materials like dialkyl phosphine oxides and alcohols ensures a more resilient supply chain compared to methods dependent on sensitive or regulated alkyl halides. The robustness of the reagents allows for longer storage times and reduced spoilage, minimizing inventory write-offs and ensuring consistent production schedules. Additionally, the use of commercially available halosilane catalysts, which are standard reagents in the fine chemical industry, guarantees a secure supply source without the geopolitical risks often associated with rare earth or precious metal catalysts.
- Scalability and Environmental Compliance: The solvent-free nature of this reaction significantly simplifies the engineering requirements for scale-up, as there is no need for large-volume solvent handling systems or explosion-proof infrastructure. The generation of water as the only byproduct aligns perfectly with green chemistry principles, reducing the environmental footprint and easing the burden on wastewater treatment facilities. This compliance with environmental standards facilitates faster regulatory approvals and supports corporate sustainability goals, positioning the manufacturer as a leader in eco-friendly chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for process development teams.
Q: What are the primary advantages of using alcohols over alkyl halides in phosphine oxide synthesis?
A: Using alcohols eliminates the toxicity and instability associated with alkyl halides, reduces hazardous waste generation, and utilizes cheaper, more abundant starting materials, significantly improving the environmental profile and cost-efficiency of the manufacturing process.
Q: Why is trimethylsilyl iodide (TMSI) preferred over other halosilanes in this reaction?
A: Experimental data indicates that TMSI provides superior catalytic activity compared to TMSCl or TMSBr, achieving isolation yields up to 95% under mild conditions, whereas chlorosilanes often result in significantly lower conversion rates and require harsher conditions.
Q: Is this method scalable for industrial production of ligand intermediates?
A: Yes, the patent explicitly demonstrates successful scale-up potential, noting that the solvent-free nature and simple workup procedures facilitate easy amplification from gram-scale laboratory synthesis to multi-kilogram commercial production without complex engineering requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Phosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this halosilane-catalyzed technology for the production of high-purity phosphine oxide intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trisubstituted phosphine oxide delivered meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can optimize your specific project requirements. By leveraging our expertise in green chemistry and process intensification, we can provide a Customized Cost-Saving Analysis tailored to your production volumes. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical phosphine-based intermediates.
