Advanced Microwave Catalysis for High-Purity Podophyllotoxin-PEG Derivatives Commercialization
The pharmaceutical industry continuously seeks innovative solutions to enhance the therapeutic index of potent anticancer agents, and patent CN101314043B represents a significant breakthrough in the field of podophyllotoxin modification. This specific intellectual property details a novel preparation method for a carboxyl monomethoxy polyethylene glycol derivative of podophyllotoxin, addressing critical limitations such as poor water solubility and severe toxicity associated with the native compound. By leveraging microwave catalysis technology, the inventors have developed a streamlined synthetic route that couples podophyllotoxin with polyethylene glycol (PEG) chains through an ester linkage, resulting in a prodrug with superior stability and bioavailability. This technological advancement is not merely a laboratory curiosity but a viable pathway for industrial production, offering a robust alternative to traditional conjugation methods that often suffer from prolonged reaction times and complex purification protocols. For R&D directors and procurement specialists alike, understanding the nuances of this microwave-assisted esterification is crucial for evaluating its potential in large-scale API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing podophyllotoxin derivatives, particularly esterification with high molecular weight polymers like PEG, are fraught with inefficiencies that hinder commercial scalability. Conventional thermal heating often requires extended reaction periods, sometimes lasting several hours to days, to drive the equilibrium towards the desired ester product, which inevitably leads to higher energy consumption and increased operational costs. Furthermore, the use of toxic organic solvents and the difficulty in removing byproducts such as dicyclohexylurea (DCU) from the viscous polymer matrix create significant downstream processing challenges. These legacy techniques frequently result in lower overall yields and inconsistent product quality, posing risks to supply chain reliability and cost-effectiveness in competitive pharmaceutical markets. The inherent sluggishness of reacting a bulky polymer chain with a sterically hindered natural product like podophyllotoxin under standard conditions necessitates harsh catalysts or excessive reagent loading, which complicates impurity control and regulatory compliance.
The Novel Approach
In stark contrast, the methodology outlined in CN101314043B utilizes microwave irradiation to overcome these kinetic barriers, achieving complete esterification in merely 25 to 35 minutes under optimized conditions. This novel approach employs a specific molar ratio of reactants, typically around 1:1.5:2:2 for podophyllotoxin, mPEG-COOH, DCC, and DMAP respectively, ensuring high conversion efficiency without the need for excessive reagent waste. The rapid dielectric heating provided by the microwave field facilitates uniform energy distribution throughout the reaction mixture, significantly accelerating the formation of the activated O-acylisourea intermediate and its subsequent nucleophilic attack by the podophyllotoxin hydroxyl group. This results in a dramatic reduction in processing time and a marked improvement in the purity profile of the final white solid product, which can be easily isolated through simple filtration and recrystallization steps. Such efficiency translates directly into enhanced throughput capabilities for manufacturers aiming to produce high-purity pharmaceutical intermediates at a commercial scale.
Mechanistic Insights into Microwave-Assisted Steglich Esterification
The core chemical transformation driving this synthesis is a variation of the Steglich esterification, facilitated by the coupling agent dicyclohexylcarbodiimide (DCC) and the nucleophilic catalyst 4-dimethylaminopyridine (DMAP). In this mechanism, the carboxyl group of the monomethoxy polyethylene glycol first reacts with DCC to form a highly reactive O-acylisourea intermediate, which is prone to rearrangement but is rapidly intercepted by the catalyst. DMAP acts as a potent acyl transfer agent, forming an even more reactive acylpyridinium species that is readily attacked by the secondary hydroxyl group at the C-4 position of the podophyllotoxin scaffold. The application of microwave energy at 510W provides the necessary activation energy to overcome the steric hindrance imposed by the bulky PEG chain and the rigid lignan structure of podophyllotoxin, ensuring the reaction proceeds to completion within the short 30-minute window. This precise control over reaction kinetics minimizes side reactions such as racemization or degradation of the sensitive lactone ring, which is critical for maintaining the biological activity of the final prodrug.
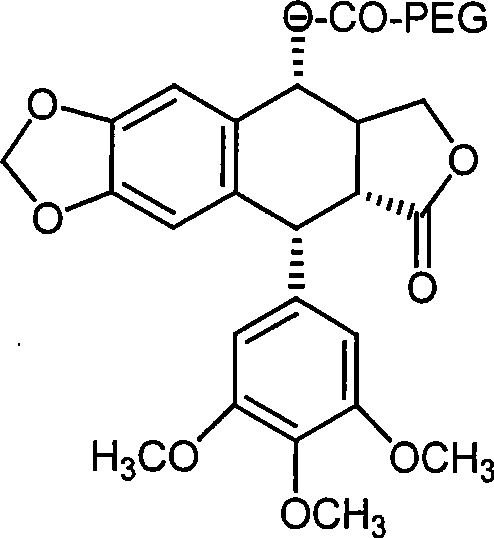
Impurity control in this process is meticulously managed through a multi-step workup procedure that leverages the solubility differences between the product and the byproducts. Following the microwave reaction, the mixture is dried and treated with anhydrous methylene chloride to dissolve the organic components, allowing for the physical removal of the insoluble dicyclohexylurea precipitate via filtration. Subsequent washing with dilute hydrochloric acid effectively removes residual DMAP and any unreacted basic impurities, while a final recrystallization from isopropyl alcohol ensures the removal of trace organic contaminants and unreacted starting materials. This rigorous purification protocol guarantees a final product with high chemical purity and consistent physical properties, such as the defined melting points observed in the embodiments (50–53°C and 59–62°C depending on PEG molecular weight). For quality assurance teams, this reproducible purification strategy offers a clear roadmap for establishing robust specification limits and analytical testing methods.
How to Synthesize Podophyllotoxin-PEG Derivative Efficiently
The synthesis of this water-soluble prodrug is designed for operational simplicity, requiring standard laboratory equipment adapted for microwave chemistry. The process begins with the precise weighing and homogeneous mixing of the four key components: podophyllotoxin, carboxyl monomethoxy polyethylene glycol (with a molecular weight preferably between 2000 and 20000), DCC, and DMAP. Once the mixture is prepared, it is subjected to microwave irradiation, where the rapid heating drives the esterification to completion, after which the crude solid is processed through a series of dissolution, filtration, and washing steps to isolate the pure derivative. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistency, and the specific step-by-step guidelines are provided below for technical reference.
- Mix podophyllotoxin, carboxyl monomethoxy polyethylene glycol (mPEG-COOH), dicyclohexylcarbodiimide (DCC), and DMAP in a molar ratio of approximately 1: 1.5:2:2.
- Subject the homogeneous mixture to microwave irradiation at 510W for 30 minutes to complete the esterification reaction efficiently.
- Purify the crude product by dissolving in methylene chloride, filtering off dicyclohexylurea, washing with dilute HCl, and recrystallizing from isopropyl alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this microwave-catalyzed synthesis route offers substantial strategic advantages for procurement managers and supply chain heads focused on cost optimization and reliability. The drastic reduction in reaction time from hours to minutes directly correlates to increased equipment utilization rates, allowing manufacturing facilities to produce larger batches within the same timeframe without significant capital investment in new reactors. This efficiency gain is compounded by the simplified workup procedure, which eliminates the need for complex chromatographic separations often required in traditional polymer conjugation, thereby reducing solvent consumption and waste disposal costs significantly. Furthermore, the use of readily available reagents like DCC and DMAP, combined with the robustness of the microwave protocol, ensures a stable supply of raw materials and minimizes the risk of production delays due to reagent scarcity or process failure.
- Cost Reduction in Manufacturing: The elimination of prolonged heating cycles and the reduction in solvent usage lead to a significant decrease in utility and material costs per kilogram of product. By avoiding the need for expensive transition metal catalysts or specialized anhydrous conditions required by other coupling methods, the overall cost of goods sold (COGS) is optimized, making the final API intermediate more price-competitive in the global market. Additionally, the high yield reported in the patent embodiments suggests minimal loss of valuable starting materials, further enhancing the economic viability of the process for large-scale production runs.
- Enhanced Supply Chain Reliability: The simplicity and speed of the microwave-assisted method reduce the complexity of the manufacturing schedule, allowing for more flexible production planning and faster response to market demand fluctuations. Since the reaction is less sensitive to minor variations in conditions compared to long-duration thermal processes, the risk of batch failure is minimized, ensuring a consistent and reliable flow of high-purity intermediates to downstream drug formulation partners. This reliability is crucial for maintaining uninterrupted supply chains for critical anticancer medications derived from podophyllotoxin scaffolds.
- Scalability and Environmental Compliance: The process demonstrates excellent potential for scale-up, particularly with the advent of continuous flow microwave reactors that can handle industrial volumes while maintaining the precise thermal control seen in batch experiments. The reduced solvent footprint and the ability to recycle certain wash solvents contribute to a greener manufacturing profile, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This environmental compliance not only mitigates regulatory risk but also enhances the brand reputation of suppliers who can demonstrate eco-friendly production practices for complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent literature. These inquiries address common concerns regarding reaction parameters, product specifications, and the practical implications of implementing microwave chemistry in a GMP environment. Understanding these details is vital for assessing the feasibility of integrating this synthetic route into existing manufacturing workflows and for ensuring that the final product meets the rigorous quality standards required for clinical applications.
Q: What are the advantages of using microwave catalysis for podophyllotoxin esterification?
A: Microwave catalysis significantly reduces reaction time from hours to minutes (e.g., 30 minutes), improves reaction selectivity, and enhances yields compared to conventional heating methods, while simplifying the workup process.
Q: How does PEGylation improve the pharmacological profile of podophyllotoxin?
A: Conjugating podophyllotoxin with polyethylene glycol (PEG) drastically improves its water solubility and stability, reduces systemic toxicity, and allows for a controlled slow-release mechanism via enzymatic hydrolysis in the body.
Q: What is the typical molecular weight range for the PEG chain in this derivative?
A: The patent specifies a preferred molecular weight range for the carboxyl monomethoxy polyethylene glycol (mPEG-COOH) between 2000 and 20000 Daltons to optimize solubility and drug release properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Podophyllotoxin-PEG Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the microwave-catalyzed production of podophyllotoxin derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials regardless of their development stage. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of podophyllotoxin-PEG derivative meets the highest standards for safety and efficacy required by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthesis technology for their anticancer drug development programs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you accelerate your timeline to market with a supply chain partner dedicated to innovation, quality, and commercial success.
