Revolutionizing Butene Liquid Crystal Intermediates: Efficient Mesylate Synthesis for High-Performance Displays
The rapid evolution of the flat-panel display industry demands increasingly sophisticated liquid crystal materials capable of delivering faster response times and wider viewing angles. Central to the manufacturing of high-grade STN and TFT mixed crystals is the efficient production of butene liquid crystal monomers. A pivotal breakthrough in this domain is detailed in patent CN101407482B, which introduces a superior intermediate for synthesizing these critical compounds. This patent discloses a novel class of alkyl-cyclohexyl methanesulfonates that serve as precursors for butene liquid crystals, offering a distinct advantage over traditional p-toluenesulfonate intermediates. By shifting the synthetic strategy from tosylation to mesylation, manufacturers can achieve significant improvements in reaction kinetics, cost efficiency, and final product purity. This report analyzes the technical merits of this innovation, providing actionable insights for R&D directors seeking robust synthetic routes and procurement managers aiming to optimize supply chain costs for electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of butene liquid crystal monomers has relied heavily on the use of p-toluenesulfonic acid esters (tosylates) as key intermediates. As illustrated in the traditional synthetic pathway, the process typically involves reacting an alkyl-cyclohexyl methanol with p-toluenesulfonyl chloride (TsCl) in the presence of a base. While chemically feasible, this conventional approach suffers from several inherent drawbacks that hinder large-scale efficiency. The primary issue lies in the post-treatment complexity; the reaction mixture requires repeated washing with water and multiple solvent extractions to isolate the product, as the tosylate derivatives often exhibit poor solubility characteristics that complicate phase separation. Furthermore, the starting alcohols in traditional methods frequently fail to transform completely, leading to low crude purity that necessitates energy-intensive recrystallization steps. These additional purification stages not only increase solvent consumption and waste generation but also result in substantial product loss, ultimately driving up the cost of goods sold (COGS) for the final liquid crystal material.
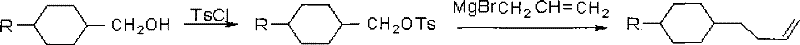
The Novel Approach
In stark contrast, the methodology presented in CN101407482B replaces the bulky tosyl group with a methanesulfonyl (mesyl) group, fundamentally altering the physicochemical properties of the intermediate. The new process utilizes methanesulfonyl chloride (MsCl), a reagent that is not only more economically accessible but also possesses a lower molecular weight, thereby offering a higher mole count per unit mass compared to TsCl. This switch facilitates a much cleaner reaction profile where the raw alcohol converts quantitatively to the methanesulfonate ester with minimal side products. The most transformative aspect of this novel approach is the streamlined workup procedure. Instead of laborious extractions, the process employs a precipitation technique where the reaction solvent is partially distilled, and water is added to induce product crystallization. This simple filtration and washing step effectively removes inorganic salts and unreacted reagents, yielding a high-purity product directly without the need for recrystallization. This paradigm shift represents a significant leap forward in process chemistry for electronic materials.
Mechanistic Insights into Methanesulfonyl Chloride Esterification
The core chemical transformation driving this innovation is the nucleophilic substitution of the hydroxyl group on the cyclohexyl-methanol derivative by the methanesulfonyl group. In the presence of a basic catalyst such as triethylamine or pyridine, the alcohol oxygen attacks the sulfur atom of the methanesulfonyl chloride. The use of polar aprotic solvents like tetrahydrofuran (THF) or dioxane is critical here, as these solvents provide excellent solubility for both the organic substrates and the inorganic byproducts during the reaction phase. Unlike traditional methods where solubility mismatches often lead to heterogeneous reaction conditions, the THF-based system ensures a homogeneous reaction environment, allowing for rapid and complete conversion even at moderate temperatures ranging from 0°C to 80°C. The mechanistic efficiency is further enhanced by the high reactivity of the methanesulfonyl chloride intermediate formed in situ, which reacts swiftly with the alcohol to form the stable sulfonate ester bond.
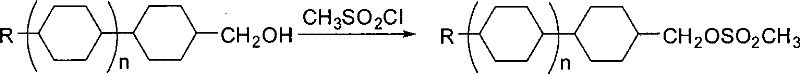
Impurity control is another area where this mechanism excels. In traditional tosylation, the formation of chlorosulfite intermediates can lead to complex byproduct profiles that are difficult to separate. However, the mesylation pathway described in the patent minimizes these side reactions. The subsequent workup leverages the differential solubility of the product versus the impurities. By adding water to the THF residue, the methanesulfonate product, which is poorly soluble in water, precipitates out as a solid. Meanwhile, the triethylamine hydrochloride salt and any excess methanesulfonyl chloride hydrolysis products remain dissolved in the aqueous-organic phase. A final wash with aqueous ethanol serves to remove any residual surface impurities and decolorize the product. This elegant separation mechanism ensures that the final intermediate meets the stringent purity specifications required for high-performance liquid crystal applications, typically achieving HPLC purity levels exceeding 97%.
How to Synthesize Alkyl-cyclohexyl Methanesulfonate Efficiently
The synthesis of these critical intermediates is designed for operational simplicity and robustness, making it ideal for both laboratory optimization and industrial scale-up. The process begins with the dissolution of the specific cyclohexyl-methanol derivative and a stoichiometric amount of base in anhydrous THF. Methanesulfonyl chloride is then introduced dropwise under controlled temperature conditions to manage the exotherm. Following the reaction period, the solvent is partially recovered via distillation, a step that not only concentrates the product but also allows for the recycling of valuable solvent resources. The addition of water triggers the precipitation of the crude product, which is then isolated by filtration. This standardized approach eliminates the variability associated with extraction efficiencies and ensures consistent batch-to-batch quality. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios tailored to different substrate variations, please refer to the standardized synthesis guide below.
- Dissolve the raw alcohol substrate and a basic catalyst (triethylamine or pyridine) in an anhydrous polar aprotic solvent such as THF or dioxane.
- Add methanesulfonyl chloride dropwise at controlled temperatures ranging from -10°C to 100°C, maintaining a molar ratio of alcohol to reagent between 1: 0.9 and 1:10.
- Upon completion, recover the solvent via distillation, add water to precipitate the product, and wash the solid with aqueous ethanol to obtain high-purity mesylate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders in the electronic chemicals sector, the adoption of this mesylation technology offers compelling strategic advantages beyond mere technical performance. The shift from tosylates to mesylates directly addresses key pain points related to raw material costs, processing time, and environmental compliance. By optimizing the molecular weight of the leaving group and simplifying the isolation procedure, manufacturers can realize substantial reductions in overall production costs while simultaneously enhancing supply reliability. These benefits are critical in a market where demand for high-quality display materials is volatile and price sensitivity is high.
- Cost Reduction in Manufacturing: The economic argument for this process is robust. Methanesulfonyl chloride is significantly less expensive than p-toluenesulfonyl chloride on a molar basis, and its lower molecular weight means that less mass of reagent is required to achieve the same molar conversion. Furthermore, the elimination of multiple solvent extraction steps drastically reduces the volume of organic solvents consumed. The ability to recover and reuse the reaction solvent (THF) without complex treatment further lowers utility and material costs. This cumulative effect results in a leaner cost structure for the production of liquid crystal intermediates.
- Enhanced Supply Chain Reliability: Simplified processing translates directly to improved throughput and shorter lead times. The precipitation-based workup is inherently faster and less prone to operational bottlenecks than multi-stage liquid-liquid extractions, which often suffer from emulsion formation or phase separation delays. This efficiency allows for quicker turnover of reactor vessels, increasing the overall capacity of the manufacturing facility without the need for capital expansion. Consequently, suppliers can respond more agilely to fluctuating market demands, ensuring a steady flow of high-purity intermediates to downstream liquid crystal blenders.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method is superior. The reduction in solvent usage and the avoidance of chlorinated extraction solvents minimize the generation of hazardous waste. The process generates primarily aqueous waste streams containing dissolved salts, which are easier and cheaper to treat than complex organic waste mixtures. Additionally, the high yield and purity reduce the need for reprocessing off-spec batches, aligning with green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this methanesulfonate synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: Why is methanesulfonate preferred over p-toluenesulfonate for butene liquid crystal synthesis?
A: Methanesulfonates offer higher reactivity in subsequent Grignard coupling reactions due to the smaller steric hindrance of the methyl group compared to the tolyl group. Additionally, methanesulfonyl chloride is significantly cheaper and has a lower molecular weight, providing better atom economy and cost efficiency.
Q: How does the new workup procedure improve product purity?
A: The novel process utilizes a precipitation method where water is added to the reaction residue, causing the product to separate out while impurities and salts remain in the solution. This eliminates the need for complex solvent extractions and recrystallization, reducing product loss and ensuring high HPLC purity (>97%).
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method is highly scalable. It uses common solvents like THF which can be recovered and reused, and the simplified workup (filtration and washing) is much easier to manage on a large scale compared to multi-step liquid-liquid extractions required by traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl-cyclohexyl Methanesulfonate Supplier
As the global demand for advanced display technologies continues to surge, securing a reliable source of high-purity liquid crystal intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in fine chemical synthesis to deliver superior electronic materials. Our technical team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative mesylation processes described in CN101407482B can be seamlessly transferred to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of alkyl-cyclohexyl methanesulfonate meets the exacting standards required for TFT and STN liquid crystal formulations.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your operations. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our intermediates in your downstream applications. Let us help you drive down costs and enhance the reliability of your liquid crystal supply chain with our proven manufacturing capabilities.
