Advanced Asymmetric Hydrogenation for Crizotinib Intermediate: Technical & Commercial Analysis
The pharmaceutical industry's relentless pursuit of efficiency in oncology drug synthesis has found a significant breakthrough in the preparation of Crizotinib intermediates, specifically detailed in Chinese Patent CN103319311A. This intellectual property introduces a revolutionary one-step asymmetric catalytic hydrogenation method for producing (1S)-1-(2,6-dichloro-3-fluorophenyl)ethanol, a critical chiral building block for the ALK inhibitor Crizotinib. Unlike traditional biocatalytic routes that suffer from prolonged reaction times and complex downstream processing, this novel chemical approach utilizes a highly specialized chiral iridium catalyst to achieve direct stereoselective reduction. For R&D directors and process chemists, this represents a paradigm shift from biological resolution to precise chemical catalysis, offering a pathway to significantly higher throughput and reduced operational complexity. The patent data underscores the viability of this method not just in theory, but through extensive experimental validation ranging from milligram-scale optimization to multi-kilogram pilot runs, establishing a robust foundation for commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (1S)-1-(2,6-dichloro-3-fluorophenyl)ethanol has relied heavily on enzymatic kinetic resolution strategies, as disclosed in prior art such as WO2006021881 and WO2007066187. These conventional pathways are inherently inefficient, typically requiring a multi-step sequence that begins with the reduction of substituted acetophenone followed by acetylation to form an ester intermediate. The critical bottleneck occurs during the enzymatic hydrolysis step, where lipases are employed to distinguish between enantiomers; however, this process is theoretically limited to a maximum yield of 50% for the desired isomer, necessitating the recycling or disposal of the unwanted (1R)-enantiomer. Furthermore, to utilize the discarded isomer, additional chemical steps involving mesylation and configuration inversion are required, drastically increasing the number of unit operations, solvent consumption, and waste generation. As illustrated in the reaction scheme below, the cumulative effect of these steps results in a lengthy production cycle, often taking up to seven days for completion, with poor reproducibility and severe sensitivity to reaction conditions that hinder reliable industrial scale-up.
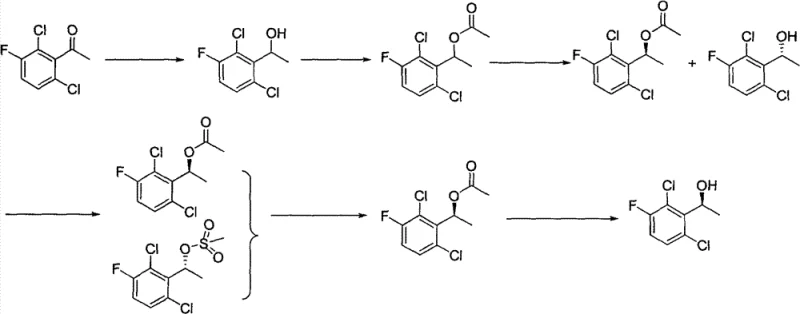
The Novel Approach
In stark contrast to the cumbersome enzymatic routes, the methodology presented in CN103319311A streamlines the entire synthesis into a single, direct asymmetric hydrogenation step. By employing a sophisticated chiral iridium catalyst in the presence of a base and hydrogen gas, the process converts the readily available ketone substrate directly into the target chiral alcohol with exceptional stereocontrol. This approach bypasses the theoretical yield ceiling of kinetic resolution, allowing for nearly quantitative conversion of the starting material, which translates to a dramatic improvement in atom economy and raw material utilization. The reaction conditions are remarkably mild yet effective, operating effectively at hydrogen pressures between 0.2 and 10 MPa and temperatures ranging from 0°C to 80°C. This simplification of the synthetic route not only reduces the physical footprint required for manufacturing but also minimizes the exposure of the product to potential degradation during multiple isolation and purification stages, thereby ensuring a cleaner impurity profile and higher overall process reliability for supply chain managers.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The cornerstone of this technological advancement lies in the unique architecture of the chiral catalyst, specifically the iridium complexes defined by Formula (I) and Formula (II) in the patent. These catalysts feature a P-N ligand system incorporating a bulky di-tert-butylphosphino (DTB) group and a chiral backbone derived from binaphthyl or similar scaffolds, which creates a highly defined chiral pocket around the metal center. This steric environment is crucial for differentiating the prochiral faces of the 1-(2,6-dichloro-3-fluorophenyl)ethanone substrate during the hydrogenation event. The mechanism likely proceeds through the formation of an active iridium-hydride species, where the coordination of the ketone oxygen and the subsequent migratory insertion of hydride are strictly governed by the chiral ligand's geometry. This precise control ensures that hydrogen delivery occurs exclusively to one face of the carbonyl group, resulting in the formation of the (1S)-alcohol with enantiomeric excess values consistently exceeding 99%. The structural integrity of the catalyst, as shown in the diagram below, allows it to maintain high activity even at extremely low loadings, a feature that is rare in asymmetric catalysis.
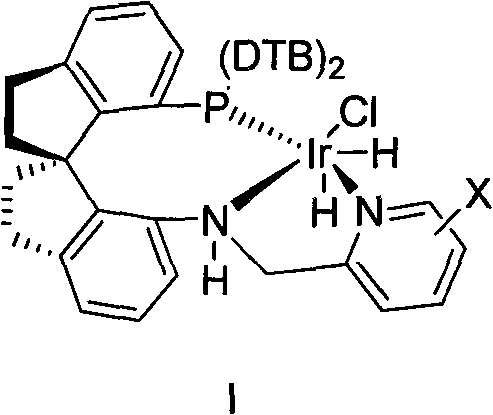
Beyond mere stereocontrol, the catalyst system exhibits remarkable stability and turnover efficiency, which is vital for managing trace metal impurities in pharmaceutical intermediates. The patent highlights that effective catalysis can be achieved with a molar ratio of catalyst to substrate ranging from 1:100,000 to 1:1,000,000. This ultra-low loading capability implies that the residual iridium content in the final product is inherently minimized, simplifying the downstream purification process and reducing the burden on heavy metal scavenging technologies. From an impurity control perspective, the direct nature of the reaction avoids the formation of by-products associated with acetylation or mesylation reagents, such as acetic acid esters or sulfonates. Consequently, the crude product profile is significantly cleaner, dominated primarily by the unreacted ketone which can be easily removed or recycled. This mechanistic elegance provides R&D teams with a robust platform for quality-by-design, ensuring that the critical quality attributes of the intermediate, particularly optical purity and chemical purity, are built into the process rather than relying solely on end-of-line testing.
How to Synthesize (1S)-1-(2,6-dichloro-3-fluorophenyl)ethanol Efficiently
Implementing this advanced hydrogenation protocol requires careful attention to reaction parameters to maximize the benefits of the chiral catalyst system. The process is designed to be operationally simple, utilizing standard high-pressure reactor equipment commonly found in fine chemical facilities. The selection of the base, such as potassium tert-butoxide or sodium ethoxide, plays a critical role in activating the catalyst precursor and maintaining the reaction cycle. Solvent choice is also flexible, with options including ethanol, THF, and toluene, allowing process engineers to optimize for solubility and safety profiles. The following guide outlines the standardized procedural framework derived from the patent examples, serving as a starting point for technology transfer and scale-up activities.
- Prepare the reaction system by charging 1-(2,6-dichloro-3-fluorophenyl)ethanone, a chiral iridium catalyst (Formula I or II), and an alkali base into a reactor under nitrogen protection.
- Add the selected solvent (e.g., ethanol, THF, or toluene) and pressurize the autoclave with hydrogen gas to a pressure range of 0.2 to 10 MPa.
- Maintain the reaction temperature between 0°C and 80°C with stirring until conversion is complete, followed by standard aqueous workup and purification to isolate the chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from enzymatic resolution to this asymmetric hydrogenation technology offers profound strategic advantages that extend beyond simple technical metrics. The most immediate impact is seen in the drastic reduction of manufacturing lead times; by collapsing a multi-day, multi-step sequence into a single reaction that completes in hours, production throughput can be multiplied significantly without expanding facility capacity. This acceleration directly enhances supply chain resilience, allowing manufacturers to respond more agilely to market demand fluctuations for Crizotinib and related oncology therapies. Furthermore, the elimination of expensive biocatalysts and the associated buffering systems removes a volatile cost component from the bill of materials, replacing it with a stable, chemically synthesized catalyst that can be sourced reliably. The ability to run reactions at high substrate concentrations, up to 5 mol/L as noted in the patent, further drives down solvent costs and waste disposal fees, contributing to a leaner and more sustainable manufacturing model.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is compelling, primarily driven by the elimination of auxiliary reagents and the maximization of raw material yield. In the conventional enzymatic route, the theoretical yield cap of 50% necessitates either purchasing double the starting material or investing in complex recycling loops for the unwanted enantiomer, both of which inflate costs. The new catalytic method achieves near-quantitative yields, effectively doubling the output per kilogram of starting ketone compared to the best-case scenario of the old method. Additionally, the ultra-low catalyst loading means that the cost contribution of the precious metal iridium is negligible on a per-kilogram basis, removing the financial risk associated with fluctuating precious metal prices. The simplification of the workup procedure, which avoids multiple extraction and chromatography steps required for enzyme removal, further reduces labor and utility costs, resulting in substantial overall cost savings for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized biological materials which can suffer from batch-to-batch variability and limited shelf life. By switching to a purely chemical catalytic system, manufacturers secure a more robust supply chain that is less susceptible to biological contamination or enzyme deactivation issues. The starting material, 1-(2,6-dichloro-3-fluorophenyl)ethanone, is a commodity chemical available from multiple global suppliers, reducing single-source dependency risks. Moreover, the reaction's tolerance to a wide range of temperatures and pressures provides operational flexibility, ensuring that production schedules are not derailed by minor equipment deviations. This reliability is crucial for maintaining the steady flow of intermediates required for continuous API manufacturing, preventing costly stock-outs that could disrupt the availability of life-saving cancer medications.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical manufacturing is under increasing scrutiny, and this new process aligns perfectly with green chemistry principles. The reduction in solvent volume due to high-concentration processing directly lowers the E-factor (mass of waste per mass of product), facilitating easier compliance with environmental regulations. The absence of enzymatic proteins and complex buffer salts simplifies wastewater treatment, reducing the load on effluent processing plants. Scalability has been proven in the patent data, which successfully demonstrates the transition from gram-scale vials to 100-liter autoclaves producing over 20 kilograms of product in a single batch. This proven track record de-risks the capital investment required for scale-up, assuring stakeholders that the technology is ready for commercial deployment at the tonnage scale required by the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims within patent CN103319311A, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production lines.
Q: What is the primary advantage of the iridium-catalyzed method over enzymatic resolution?
A: The iridium-catalyzed asymmetric hydrogenation described in CN103319311A eliminates the need for multi-step protection/deprotection and enzymatic hydrolysis, reducing the process from roughly five steps to a single direct transformation with near-quantitative yield.
Q: What level of optical purity can be achieved with this catalytic system?
A: The patented process consistently achieves enantiomeric excess (ee) values greater than 99%, with specific embodiments reporting up to 99.8% ee, meeting the stringent requirements for oncology API synthesis.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly demonstrates scalability from gram-level laboratory experiments to 100-liter pilot scales (producing over 20 kg per batch), utilizing robust conditions like moderate hydrogen pressure and common solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S)-1-(2,6-dichloro-3-fluorophenyl)ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced oncology drugs depends on the availability of high-quality, cost-effective intermediates. Our technical team has thoroughly analyzed the capabilities presented in CN103319311A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this efficient hydrogenation process to fruition. We are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including the critical >99% ee requirement for this chiral alcohol. Our commitment to excellence ensures that every batch delivered meets the exacting standards required by global regulatory bodies, providing our partners with peace of mind regarding product quality and consistency.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this transformative synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us help you secure a competitive advantage in the Crizotinib supply chain through innovation and operational excellence.
