Advanced PTC Synthesis of 2-Nitro-1-Aryl Alcohols for Scalable Pharma Manufacturing
The global demand for high-quality pharmaceutical intermediates necessitates synthetic routes that balance efficiency with operational simplicity. Patent CN101857547A introduces a transformative preparation method for 2-nitro-1-aryl alcohols, addressing critical bottlenecks in traditional nitroaldol (Henry) reactions. These compounds serve as pivotal precursors in the synthesis of numerous active pharmaceutical ingredients (APIs), yet their production has historically been plagued by low yields and difficult purification processes. The disclosed innovation utilizes a phase transfer catalysis (PTC) system employing sodium bicarbonate as a mild base, effectively circumventing the harsh conditions typically required for such transformations. By shifting from aggressive alkalis to a buffered bicarbonate system, the technology ensures the stability of the sensitive beta-nitro alcohol moiety against dehydration. This strategic adjustment not only enhances the chemical yield but also streamlines the downstream processing, offering a robust solution for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio.
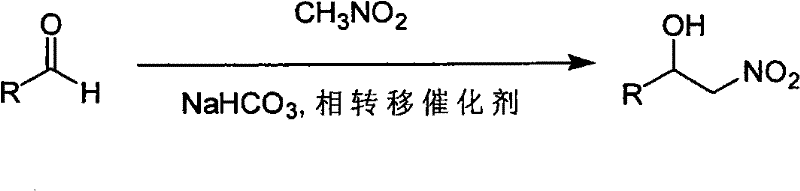
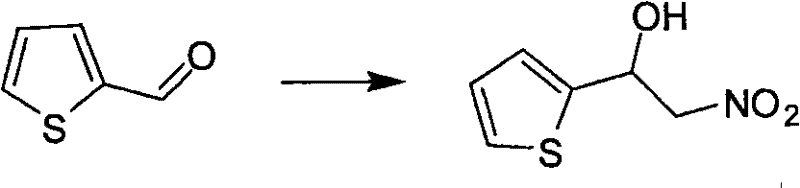
Traditional synthetic methodologies for generating 2-nitro-1-aryl alcohols predominantly rely on strong inorganic bases such as sodium hydroxide or sodium carbonate to deprotonate nitromethane. While thermodynamically feasible, these aggressive conditions frequently trigger a secondary elimination reaction, causing the newly formed hydroxyl group to dehydrate and form unwanted nitroalkene byproducts. This side reaction drastically diminishes the isolated yield of the target alcohol and creates a complex impurity profile that is notoriously difficult to separate, often requiring expensive chromatographic purification. Furthermore, the exothermic nature of reactions involving strong bases poses safety risks during scale-up, limiting the feasibility of commercial production. In stark contrast, the novel approach detailed in the patent employs saturated aqueous sodium bicarbonate in conjunction with a quaternary ammonium salt, specifically tetrabutylammonium iodide (TBAI). This mild basic environment is sufficient to initiate the nucleophilic attack of the nitronate anion on the aldehyde carbonyl but is too weak to facilitate the elimination of water from the product. Consequently, the reaction proceeds with high selectivity towards the alcohol, as evidenced by the successful synthesis of diverse substrates ranging from simple benzaldehydes to complex heterocycles like thiophene derivatives shown below.
The mechanistic elegance of this phase transfer catalyzed Henry reaction lies in the synergistic interaction between the weak base and the lipophilic catalyst within a biphasic solvent system. The tetrabutylammonium cation acts as a molecular ferry, transporting the nitronate anion generated in the aqueous bicarbonate phase into the organic phase where the hydrophobic aldehyde substrate resides. This intimate contact significantly accelerates the reaction rate at ambient temperatures, eliminating the need for energy-intensive heating or cooling cycles. Crucially, the use of bicarbonate serves as a kinetic control mechanism; by maintaining a lower pH compared to hydroxide bases, the concentration of the highly reactive nitronate species is moderated, preventing the rapid over-reaction or decomposition of sensitive functional groups. This controlled reactivity is paramount for impurity control, as it suppresses the formation of polymeric byproducts and nitroalkenes that typically arise from uncontrolled base catalysis. The result is a cleaner reaction matrix that allows for straightforward isolation of the product through simple extraction and crystallization, thereby ensuring the delivery of high-purity 2-nitro-1-aryl alcohol suitable for stringent regulatory standards.
For procurement specialists and supply chain managers, the transition to this bicarbonate-mediated PTC protocol represents a substantial opportunity for cost reduction in API manufacturing. The replacement of hazardous strong bases with inexpensive, non-toxic sodium bicarbonate eliminates the need for specialized corrosion-resistant equipment and rigorous safety protocols associated with handling caustic sodas. Moreover, the ambient temperature operation significantly lowers energy consumption compared to processes requiring reflux or cryogenic control, directly impacting the utility costs of large-scale production. The simplified workup procedure, which avoids complex distillation or column chromatography, reduces solvent usage and waste generation, aligning with modern green chemistry principles and environmental compliance mandates. From a supply chain reliability perspective, the robustness of this method against variations in raw material quality ensures consistent batch-to-batch performance, reducing the risk of production delays. The ability to easily scale this reaction from laboratory grams to multi-ton commercial quantities without re-optimizing critical parameters provides a secure foundation for long-term supply continuity of these essential fine chemical intermediates.
- Dissolve the corresponding aromatic aldehyde in a mixed solvent system of ethyl acetate and saturated aqueous sodium bicarbonate.
- Add the phase transfer catalyst, tetrabutylammonium iodide (TBAI), to the mixture and stir at room temperature to facilitate interfacial contact.
- Introduce nitromethane to the reaction system, maintain stirring overnight at room temperature, and proceed with standard organic workup including washing and drying.
Frequently Asked Questions (FAQ)
Q: Why is sodium bicarbonate preferred over sodium hydroxide for this Henry reaction?
A: Sodium bicarbonate acts as a mild base that effectively catalyzes the nitroaldol addition without promoting the subsequent dehydration of the beta-nitro alcohol product. Stronger bases like NaOH often lead to the formation of nitroalkenes via elimination, significantly reducing the yield of the desired alcohol and complicating purification.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is specifically designed for scalability. It operates at room temperature without the need for cryogenic cooling or anhydrous conditions, and the workup involves simple liquid-liquid extraction, making it highly suitable for large-scale commercial manufacturing of complex intermediates.
Q: What types of aldehyde substrates are compatible with this PTC method?
A: The method demonstrates broad substrate scope, successfully reacting with various aromatic aldehydes including those with electron-withdrawing groups (like cyano or fluoro), heteroaromatic aldehydes (such as thiophene and pyridine derivatives), and polycyclic aromatic aldehydes (like naphthaldehyde and pyrenecarboxaldehyde).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-1-Aryl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your drug development pipeline depends heavily on the quality and availability of key building blocks like 2-nitro-1-aryl alcohols. Our technical team has extensively analyzed the potential of the PTC-based synthesis route described in CN101857547A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering these intermediates with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for pharmaceutical applications. Our facility is equipped to handle the specific solvent systems and extraction protocols necessary to maximize yield while minimizing impurities, guaranteeing a supply chain that supports your critical timelines.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage potential partners to contact us directly to obtain specific COA data for our current inventory and to discuss route feasibility assessments for custom derivatives. Let us help you streamline your sourcing strategy and secure a competitive advantage through superior chemical manufacturing solutions.
