Advanced Synthesis of Flupenthixol EP Impurity H for Global Quality Control Standards
Introduction to Novel Impurity Synthesis Technology
The pharmaceutical industry relies heavily on precise impurity profiling to ensure the safety and efficacy of active pharmaceutical ingredients (APIs). Patent CN115850232B introduces a groundbreaking preparation method for Flupenthixol EP Impurity H, a critical reference standard required for the quality control of Flupenthixol, a potent neuroleptic agent. This patent discloses a robust six-step synthetic route that transforms 2-trifluoromethylthioxanthone into the target impurity, (E)-2-(4-(3-(2-(trifluoromethyl)-9H-thioxanthene-9-yl)allyl)piperazin-1-yl)ethan-1-ol. By addressing the previous technical void in the synthesis of this specific compound, the invention provides a reliable pathway for generating high-quality impurity standards. This development is particularly significant for regulatory compliance, as it enables manufacturers to accurately quantify trace impurities that could impact patient safety. The methodology emphasizes the use of readily available raw materials and optimized reaction conditions to achieve superior yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of patent CN115850232B, the chemical community faced a significant challenge known as a technical blank regarding the specific synthesis of Flupenthixol EP Impurity H. Without a dedicated synthetic route, obtaining this impurity for analytical method validation was exceptionally difficult, often relying on isolation from crude reaction mixtures which is inefficient and yields insufficient quantities. The lack of a defined protocol meant that quality control laboratories struggled to establish accurate detection limits for this specific degradation product or synthetic byproduct. Furthermore, alternative routes attempting to modify the Flupenthixol molecule directly often resulted in complex mixtures of geometric isomers, complicating the purification process. This scarcity of reliable reference material posed a risk to the consistent quality assurance of Flupenthixol formulations in the global market. Consequently, there was an urgent need for a dedicated, linear synthesis that could reliably produce this specific structural analog.
The Novel Approach
The inventive approach detailed in the patent overcomes these historical limitations by constructing the impurity molecule from the ground up using a logical, step-wise assembly. Starting from the commercially viable 2-trifluoromethylthioxanthone, the route systematically builds the requisite side chain through a sequence of reduction, formylation, olefination, and amination. This strategy avoids the pitfalls of modifying the final API, instead focusing on a precursor-based synthesis that allows for better control over stereochemistry and purity. The use of a Wittig reaction ensures the formation of the desired (E)-alkene geometry, which is crucial for matching the impurity profile found in the actual drug substance. By breaking the synthesis down into six distinct, manageable transformations, the method significantly enhances reproducibility. This structured approach not only fills the technical gap but also establishes a new benchmark for producing complex thioxanthene derivatives with high fidelity.
Mechanistic Insights into the Six-Step Synthetic Cascade
The core of this technology lies in its elegant manipulation of the thioxanthene scaffold through a series of classic yet highly optimized organic transformations. The process initiates with the reduction of the ketone moiety in 2-trifluoromethylthioxanthone using borane dimethyl sulfide, effectively converting the carbonyl group into a methylene bridge to form the 9H-thioxanthene core. Subsequent lithiation at the 9-position followed by quenching with methyl formate introduces the aldehyde functionality, setting the stage for chain extension. The pivotal step involves a Wittig olefination using carboethoxymethylene triphenylphosphine, which installs the acrylic ester side chain with high stereoselectivity for the trans-configuration. Following this, selective reduction of the ester to an allylic alcohol using DIBAL-H preserves the double bond integrity while preparing the molecule for functionalization.
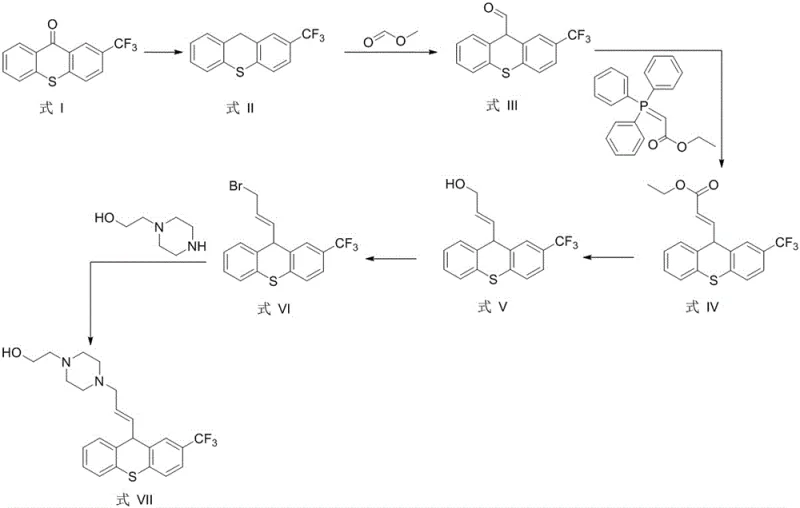
The final stages of the synthesis involve converting the allylic alcohol into a reactive bromide intermediate using phosphorus tribromide, which then undergoes a nucleophilic substitution with N-hydroxyethylpiperazine. This final coupling reaction attaches the piperazine tail, completing the structural requirements of Flupenthixol EP Impurity H. Each step is meticulously controlled with specific temperature ranges, such as maintaining -78°C during lithiation to prevent side reactions, and utilizing appropriate quenching agents like saturated ammonium chloride or sodium potassium tartrate to ensure clean workups. The mechanistic precision ensures that the final product matches the spectral data required for certification as a reference standard. This level of control over the reaction pathway minimizes the formation of regioisomers and other related substances, thereby simplifying the downstream purification processes.
How to Synthesize Flupenthixol EP Impurity H Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters outlined in the patent to ensure maximum yield and purity. The process begins with the dissolution of the starting ketone in a solvent such as tetrahydrofuran or dioxane, followed by the controlled addition of the reducing agent under heating conditions. Subsequent steps demand precise temperature management, particularly during the cryogenic lithiation and DIBAL-H reduction phases, to maintain chemoselectivity. The workflow integrates standard unit operations including extraction, drying, and column chromatography to isolate intermediates at each stage. For a comprehensive breakdown of the specific molar ratios, solvent choices, and purification techniques required for each of the six steps, please refer to the standardized guide below.
- Reduce 2-trifluoromethylthioxanthone using borane dimethyl sulfide to form the thioxanthene intermediate.
- Perform lithiation followed by formylation with methyl formate to introduce the aldehyde group at the 9-position.
- Execute a Wittig reaction with carboethoxymethylene triphenylphosphine to establish the acrylate side chain.
- Reduce the ester to an allylic alcohol using DIBAL-H, followed by bromination with phosphorus tribromide.
- Complete the synthesis via nucleophilic substitution with N-hydroxyethylpiperazine to yield the final impurity standard.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the methodology described in patent CN115850232B offers substantial strategic benefits for sourcing high-purity pharmaceutical intermediates. The reliance on 2-trifluoromethylthioxanthone as a starting material is a key advantage, as this compound is commercially available and cost-effective, reducing the dependency on custom-synthesized precursors. The linear nature of the six-step sequence allows for straightforward scaling, as each reaction utilizes common reagents and solvents that are easily sourced in bulk quantities. This accessibility translates directly into enhanced supply chain reliability, minimizing the risk of production delays caused by raw material shortages. Furthermore, the high yields reported in the patent embodiments suggest a material-efficient process that reduces waste generation and lowers the overall cost of goods sold.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts or exotic reagents, relying instead on fundamental organic chemistry tools like borane reductions and Wittig reagents. This simplification of the reagent profile significantly lowers the direct material costs associated with production. Additionally, the high efficiency of the reduction and substitution steps minimizes the loss of valuable intermediates, further driving down the cost per gram of the final impurity standard. By avoiding complex enzymatic or biocatalytic steps, the process remains compatible with standard chemical manufacturing infrastructure, preventing the need for costly capital investment in specialized equipment.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as methyl formate, phosphorus tribromide, and N-hydroxyethylpiperazine ensures that the supply chain is resilient against market volatility. These reagents are produced by multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard laboratory temperatures and pressures, facilitates technology transfer between different manufacturing sites. This flexibility allows for diversified production strategies, ensuring continuous availability of Flupenthixol EP Impurity H even during regional disruptions. Consequently, pharmaceutical companies can maintain uninterrupted quality control operations without fearing stockouts of critical reference materials.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like ethyl acetate and dichloromethane which have established recovery and recycling protocols in industrial settings. The absence of heavy metal contaminants simplifies the waste treatment process, aligning with increasingly stringent environmental regulations. The stepwise purification via column chromatography, while labor-intensive on a small scale, can be adapted to crystallization or distillation methods upon scale-up to improve throughput. This adaptability ensures that the manufacturing process can grow from kilogram-scale development to multi-ton commercial production without requiring a complete process redesign. Such scalability is essential for meeting the growing global demand for high-quality impurity standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Flupenthixol EP Impurity H. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this new supply source into their existing quality assurance frameworks. The answers reflect the practical realities of executing this synthesis in a regulated environment.
Q: What is the primary starting material for synthesizing Flupenthixol EP Impurity H?
A: According to patent CN115850232B, the synthesis initiates with 2-trifluoromethylthioxanthone, a commercially available ketone that serves as the core scaffold for the thioxanthene ring system.
Q: How does this new method improve upon previous impurity sourcing strategies?
A: Prior to this invention, there was a technical blank in the literature regarding the specific preparation of this impurity. This method fills that gap by providing a reproducible, high-yield route using accessible reagents, ensuring a stable supply of reference standards.
Q: Is this synthesis scalable for industrial reference standard production?
A: Yes, the process utilizes standard organic transformations such as Wittig olefination and nucleophilic substitution which are well-suited for scale-up. The use of common solvents like THF and DCM further supports manufacturability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flupenthixol EP Impurity H Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN115850232B to serve the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major API manufacturers. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation. By leveraging this novel six-step route, we can provide Flupenthixol EP Impurity H with superior consistency and competitive lead times. Our dedication to technical excellence ensures that every batch meets the rigorous demands of international pharmacopoeias.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities align with your supply chain goals. Contact us today to secure a reliable supply of this critical reference standard and enhance the quality control of your Flupenthixol formulations.
