Advanced Synthesis of 2-Arylamino-1,3,4-Oxadiazoles for High-Purity Pharmaceutical Intermediates
Introduction to Patent CN102229578A Technology
The pharmaceutical and agrochemical industries continuously demand more efficient and environmentally benign pathways for constructing privileged heterocyclic scaffolds. Patent CN102229578A introduces a groundbreaking preparation method for 2-arylamino-1,3,4-oxadiazole compounds, a structural motif prevalent in numerous bioactive molecules ranging from anticonvulsants to anti-inflammatory agents. This technology leverages the unique reactivity of isoselenocyanates reacting with acylhydrazides or diacylhydrazides in common organic solvents. Unlike legacy processes that rely on aggressive dehydration or desulfurization reagents, this novel approach operates under remarkably mild thermal conditions, typically between 20°C and 135°C, ensuring high product integrity. The strategic advantage lies in its operational simplicity and the inherent ability to recover elemental selenium, transforming a potential waste stream into a reusable asset. For R&D directors and procurement specialists alike, this represents a significant leap forward in process chemistry, offering a robust platform for the commercial scale-up of complex pharmaceutical intermediates with reduced ecological footprints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylamino-1,3,4-oxadiazoles has been plagued by significant technical and economic hurdles associated with traditional cyclization strategies. Conventional routes often necessitate the use of semicarbazide derivatives subjected to harsh dehydration conditions employing reagents such as phosphorus oxychloride, concentrated sulfuric acid, or Burgess reagent. These chemicals are not only highly corrosive and toxic, posing severe safety risks to personnel and equipment, but they also generate substantial quantities of hazardous waste that require costly disposal protocols. Alternatively, thiosemicarbazide derivatives require desulfurization using agents like iodine, bromine, or mercury salts, which introduce heavy metal contamination risks and complicate purification workflows. Furthermore, reactions involving 1,3,4-oxadiazol-2-ones with organic amines often suffer from low yields when targeting arylamino substitutions, leading to poor atom economy and inflated production costs. These inefficiencies create bottlenecks in supply chains, extending lead times and compromising the reliability of high-purity intermediate delivery.
The Novel Approach
In stark contrast, the methodology disclosed in CN102229578A circumvents these challenges by utilizing isoselenocyanates as key building blocks. This innovative pathway eliminates the need for external dehydrating or desulfurizing agents entirely, relying instead on the intrinsic thermodynamic drive of the cyclization reaction to expel elemental selenium. The process is adaptable to a wide array of solvents including dichloromethane, ethanol, and DMF, providing flexibility for process optimization. Crucially, the reaction conditions are温和 (mild), typically proceeding efficiently at temperatures between 50°C and 100°C over durations of 3 to 8 hours. This gentler approach preserves sensitive functional groups on the aromatic rings, thereby expanding the scope of accessible derivatives. The post-treatment is exceptionally straightforward, involving simple filtration to isolate the selenium byproduct followed by recrystallization of the target compound. This streamlined workflow drastically reduces processing time and solvent consumption, establishing a new benchmark for efficiency in heterocyclic synthesis.
Mechanistic Insights into Isoselenocyanate-Mediated Cyclization
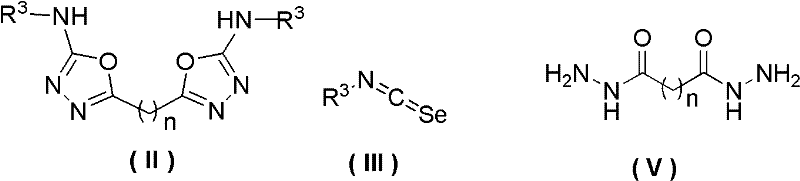
The core of this technological advancement lies in the nucleophilic addition-elimination mechanism facilitated by the isoselenocyanate functionality. When the acylhydrazide nucleophile attacks the electrophilic carbon of the isoselenocyanate group, a selenourea intermediate is transiently formed. This intermediate subsequently undergoes an intramolecular cyclization where the carbonyl oxygen attacks the adjacent carbon, driven by the formation of the stable 1,3,4-oxadiazole ring system. The driving force for this transformation is the precipitation of elemental selenium, which shifts the equilibrium towards product formation according to Le Chatelier's principle. This mechanistic pathway is distinct from oxygen- or sulfur-based analogues because the selenium-carbon bond is weaker and more labile under thermal conditions, allowing for spontaneous extrusion of selenium without the need for oxidative or reductive auxiliaries. Understanding this mechanism is vital for R&D teams aiming to optimize reaction kinetics, as the rate-determining step often involves the initial nucleophilic attack, which can be modulated by solvent polarity and temperature control.
From an impurity control perspective, this mechanism offers superior selectivity compared to acid-catalyzed dehydrations. Traditional methods often promote side reactions such as hydrolysis of the hydrazide or over-dehydration leading to polymeric byproducts. In the isoselenocyanate route, the primary byproduct is elemental selenium, which is insoluble in most reaction media and can be removed quantitatively by simple filtration. This physical separation ensures that the crude product contains minimal inorganic contaminants, simplifying downstream purification. Moreover, the absence of strong acids or bases prevents the degradation of acid-sensitive substituents on the aryl rings, such as esters or ethers, which might otherwise decompose under conventional conditions. The result is a cleaner reaction profile with higher crude purity, reducing the burden on final crystallization steps and ensuring consistent quality for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 2-Arylamino-1,3,4-Oxadiazole Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent selection to maximize yield and facilitate selenium recovery. The patent outlines a generalized protocol where isoselenocyanates and hydrazides are combined in a molar ratio ranging from 1:0.8 to 1:2, with a preferred range of 1:0.8 to 1.2 to minimize excess reagent waste. The reaction mixture is heated to the optimal temperature window, monitored until completion, and then processed to isolate the solid selenium and the liquid product phase. Detailed standardized operating procedures regarding specific heating rates, stirring speeds, and filtration techniques are critical for reproducibility at scale. For comprehensive step-by-step instructions tailored to specific substrates, please refer to the technical guide below.
- Dissolve isoselenocyanate (Formula III) in a suitable organic solvent such as dichloromethane, ethanol, or DMF.
- Add the corresponding acylhydrazide (Formula IV) or diacylhydrazide (Formula V) to the reaction mixture.
- Heat the mixture to 50-100°C for 3-8 hours, then filter to recover selenium powder and recrystallize the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this isoselenocyanate-based methodology translates into tangible operational improvements and risk mitigation. The elimination of hazardous dehydrating agents like POCl3 removes the need for specialized corrosion-resistant reactors and complex scrubbing systems, thereby lowering capital expenditure requirements for manufacturing facilities. Additionally, the ability to recover and recycle selenium powder creates a closed-loop material flow that reduces raw material procurement costs over time. The mild reaction conditions also enhance workplace safety by minimizing exposure to toxic fumes and exothermic runaway risks, which can lower insurance premiums and regulatory compliance burdens. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting fluctuating market demands without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the dependency on expensive and single-use dehydration reagents such as DCC or Burgess reagent. Since the selenium byproduct is solid and easily separable, the downstream purification workload is drastically reduced, leading to lower labor and utility costs per kilogram of product. Furthermore, the high yields reported in the patent examples, often exceeding 90%, ensure maximum conversion of starting materials into valuable intermediates, minimizing waste disposal fees and improving overall atom economy.
- Enhanced Supply Chain Reliability: Sourcing hazardous reagents often involves long lead times and strict transportation regulations due to their classification as dangerous goods. By shifting to a chemistry that utilizes stable isoselenocyanates and common solvents like ethanol or ethyl acetate, the supply chain becomes less vulnerable to logistical disruptions. The robustness of the reaction across a wide temperature range (20-135°C) also provides flexibility in manufacturing scheduling, allowing production to proceed even if precise temperature control equipment is temporarily unavailable, thus ensuring continuous supply continuity for downstream API synthesis.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which relies on filtration and recrystallization rather than complex chromatographic separations or aqueous washes, makes this process highly scalable from pilot plant to multi-ton production. The generation of elemental selenium as the sole major byproduct simplifies waste management, as selenium can be collected and returned to suppliers for regeneration into isoselenocyanates. This aligns with green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding heavy metal discharge and solvent emissions, securing long-term operational licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and claims within CN102229578A, providing clarity on substrate compatibility and process parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: What are the advantages of the isoselenocyanate route over traditional dehydration methods?
A: Unlike traditional methods using toxic dehydrating agents like POCl3 or DCC, this process operates under mild conditions without requiring harsh reagents, significantly simplifying post-treatment and reducing environmental impact.
Q: Can the selenium byproduct be recovered and reused?
A: Yes, the reaction generates elemental selenium powder as a byproduct which can be easily separated by filtration and recycled, contributing to lower raw material costs and improved sustainability.
Q: What types of substituents are compatible with this synthesis method?
A: The method demonstrates broad substrate scope, accommodating various aryl groups including chlorophenyl, bromophenyl, methoxyphenyl, and heterocycles like pyridyl and furyl, making it versatile for diverse intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylamino-1,3,4-Oxadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-arylamino-1,3,4-oxadiazole meets the highest international standards. Our commitment to process excellence means we can leverage the efficiencies of the isoselenocyanate route to deliver cost-competitive solutions without sacrificing quality.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning, ensuring a reliable partnership for your long-term success.
