Scalable Production of High-Purity 2-Methylene Glutaric Acid via Novel Composite Catalysis
The chemical industry is constantly seeking more efficient and cost-effective pathways for producing high-value intermediates, and the synthesis of 2-methylene glutaric acid represents a critical area of innovation for polymer and agrochemical sectors. A recent technological breakthrough, detailed in patent CN114149316A, introduces a novel preparation method that fundamentally optimizes the catalytic system used in the dimerization of acrylates. This patent discloses a sophisticated approach utilizing a composite catalyst mixture of organic phosphorus and tertiary amine, which addresses long-standing challenges regarding yield, selectivity, and process safety. By shifting away from traditional single-component catalysts, this method achieves a remarkable improvement in the yield of the 2-methylene glutaric acid diester intermediate, subsequently boosting the final acid production efficiency. For global procurement and R&D teams, understanding this shift is vital, as it signals a move towards more sustainable and economically viable manufacturing processes for this essential fine chemical building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methylene glutaric acid diesters has relied heavily on the use of single organic phosphine catalysts, such as tributylphosphine, often under harsh conditions that pose significant operational risks. These traditional methods suffer from several critical drawbacks, including the high toxicity and flammability of the catalysts, which necessitate stringent safety protocols and increase handling costs. Furthermore, the use of单一 organic phosphine catalysts frequently leads to substantial side reactions, particularly the polymerization of the acrylate raw material, which drastically reduces the atom economy and complicates downstream purification. The recovery of high-boiling-point solvents in these older processes is energy-intensive and difficult, contributing to a larger environmental footprint. Additionally, the high cost of pure organic phosphine reagents places a heavy burden on the overall production economics, making the final product less competitive in price-sensitive markets like agricultural chemicals and commodity polymers.
The Novel Approach
The innovative methodology presented in the patent data overcomes these deficiencies by introducing a synergistic composite catalyst system that balances reactivity with selectivity. By combining organic phosphine with a tertiary amine, the new process leverages the high reactivity of the phosphine component while utilizing the tertiary amine to modulate selectivity and reduce costs. This dual-catalyst approach allows the reaction to proceed effectively at lower temperatures, typically between 50-60°C, which mitigates the thermal decomposition risks associated with organic phosphines. Moreover, the implementation of an intermittent dropping mode for the catalyst addition ensures that the catalyst concentration remains optimal throughout the reaction, effectively suppressing the formation of unwanted acrylate polymers. This results in a cleaner reaction profile, higher yields of the desired diester intermediate, and a simplified workup procedure that enhances the overall sustainability of the manufacturing process.
Mechanistic Insights into Composite Catalyzed Dimerization
The core of this technological advancement lies in the intricate interplay between the organic phosphine and the tertiary amine within the catalytic cycle. The organic phosphine component, such as tributylphosphine, acts as the primary nucleophile to initiate the dimerization of the acrylate monomer, forming the necessary carbon-carbon bonds to create the glutaric acid backbone. However, without modulation, this high reactivity can lead to uncontrolled polymerization. The tertiary amine component serves as a crucial co-catalyst that enhances the selectivity of the reaction towards the desired dimer rather than higher-order polymers. This synergy allows the system to maintain high catalytic activity even at reduced temperatures, ensuring that the reaction proceeds to completion without the thermal degradation of sensitive reagents. The specific molar ratio of organic phosphorus to tertiary amine, optimized at approximately 1:2, is critical for maximizing this synergistic effect, ensuring that the electronic and steric properties of the catalyst complex are perfectly tuned for the transformation.
Impurity control is another significant aspect of this mechanistic design, particularly regarding the suppression of acrylate polymer byproducts. In conventional systems, the continuous presence of a high concentration of catalyst can trigger rapid chain growth of the acrylate monomer, leading to viscous polymeric wastes that are difficult to separate. The novel intermittent dropping strategy described in the patent keeps the instantaneous concentration of the active catalyst species low within the reaction bulk. This kinetic control effectively starves the polymerization pathway while still allowing the slower, selective dimerization to occur. Consequently, the generation of acrylate polymer byproducts is reduced by a significant margin, reported in the data as a reduction of roughly 14-16% compared to traditional methods. This purity enhancement not only improves the yield of the intermediate ester but also simplifies the subsequent hydrolysis and acidification steps, leading to a final product with superior quality specifications suitable for high-end applications.
How to Synthesize 2-Methylene Glutaric Acid Efficiently
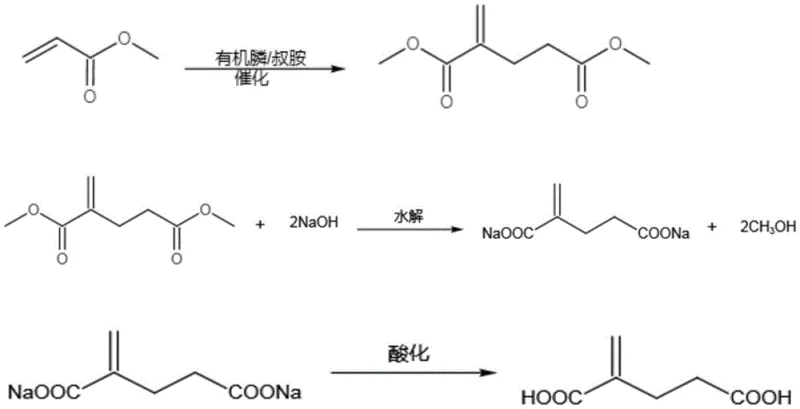
The practical execution of this synthesis involves a carefully controlled two-step sequence designed for maximum efficiency and safety in an industrial setting. The first stage focuses on the construction of the carbon skeleton through the dimerization of low-molecular-weight acrylates, such as methyl acrylate, using the optimized composite catalyst system under mild thermal conditions. The second stage involves the hydrolysis of the resulting diester intermediate using a two-phase aqueous system, which eliminates the need for additional organic solvents and facilitates the easy recovery of alcohol byproducts. This streamlined approach minimizes waste generation and energy consumption, aligning with modern green chemistry principles. For detailed operational parameters and standardized protocols, please refer to the technical guide below.
- Synthesize the 2-methylene glutaric acid diester intermediate by intermittently adding a composite catalyst (organic phosphine and tertiary amine) to low-molecular-weight acrylate at 50-60°C.
- Perform reflux hydrolysis of the diester intermediate with aqueous sodium hydroxide in a two-phase system for 3-5 hours to form the disodium salt.
- Acidify the mother liquor to pH ≤ 1 to precipitate the final 2-methylene glutaric acid product, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift to a composite catalyst system directly addresses the volatility and high cost associated with pure organic phosphine reagents by substituting a significant portion of the expensive catalyst with more affordable and readily available tertiary amines. This substitution results in a substantial reduction in raw material costs per kilogram of finished product, enhancing the overall margin structure for manufacturers. Furthermore, the improved selectivity of the process means that less raw material is wasted on forming polymeric byproducts, leading to better atom economy and reduced disposal costs for hazardous waste streams. These factors combine to create a more robust and cost-competitive supply chain for 2-methylene glutaric acid derivatives.
- Cost Reduction in Manufacturing: The replacement of a portion of the expensive organic phosphine catalyst with cheaper tertiary amines leads to a direct decrease in catalyst expenditure, which is a significant variable cost in this synthesis. Additionally, the reduction in byproduct formation means that less feedstock is lost to waste, effectively lowering the cost of goods sold. The ability to operate at lower temperatures also reduces energy consumption for heating and cooling, contributing to further operational savings. These cumulative efficiencies allow for a more aggressive pricing strategy in the market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reagents required for this process, including methyl acrylate, sodium hydroxide, and common tertiary amines, are commodity chemicals with stable and widespread global supply chains. This reduces the risk of production stoppages due to raw material shortages, which can occur with specialized or niche catalysts. The simplified process flow, which avoids complex solvent recovery systems and hazardous high-temperature operations, also minimizes the risk of unplanned downtime due to equipment failure or safety incidents. This reliability ensures consistent delivery schedules for downstream customers in the pharmaceutical and agrochemical industries.
- Scalability and Environmental Compliance: The solvent-free or low-solvent nature of the hydrolysis step significantly reduces the volume of volatile organic compounds (VOCs) emitted during production, easing the burden on environmental control systems. The tertiary amine components in the waste stream are generally easier to degrade biologically compared to persistent organophosphorus compounds, simplifying wastewater treatment compliance. The process is inherently scalable, as the intermittent addition of the catalyst can be easily automated and controlled in large-scale reactors, facilitating the transition from pilot plant to commercial tonnage production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-methylene glutaric acid using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical properties of the process for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this material into your specific product formulations or manufacturing lines.
Q: What are the advantages of the composite catalyst over traditional single catalysts?
A: The composite catalyst system, combining organic phosphine and tertiary amine, significantly reduces the formation of acrylate polymer byproducts compared to single organic phosphine catalysts. It also lowers overall catalyst costs since tertiary amines are generally less expensive, and it allows the reaction to proceed efficiently at lower temperatures (50-60°C), enhancing safety and operability.
Q: How does the intermittent dropping mode affect the reaction yield?
A: By adding the composite catalyst in intermittent portions rather than continuously or all at once, the concentration of the catalyst in the reaction system remains low. This specific feeding strategy effectively suppresses the polymerization of the acrylate raw material, thereby increasing the selectivity for the desired 2-methylene glutaric acid diester and improving the overall yield to the 75-78% range.
Q: Is the hydrolysis step performed in a solvent?
A: No, the hydrolysis step utilizes a solvent-free two-phase system. The intermediate diester is mixed directly with an aqueous sodium hydroxide solution. Upon heating and refluxing, the two phases mutually dissolve as hydrolysis proceeds, simplifying the process, reducing solvent waste, and facilitating the recovery of byproduct alcohols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylene Glutaric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity intermediates like 2-methylene glutaric acid in the development of next-generation polymers and agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, such as acrylate polymers, guaranteeing the performance consistency required for high-end applications in chip manufacturing additives and specialty resins. We are committed to leveraging advanced catalytic technologies to deliver superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your projects.
