Revolutionizing Ether Synthesis: Mild O-Allylation Technology for Commercial Scale-Up
Revolutionizing Ether Synthesis: Mild O-Allylation Technology for Commercial Scale-Up
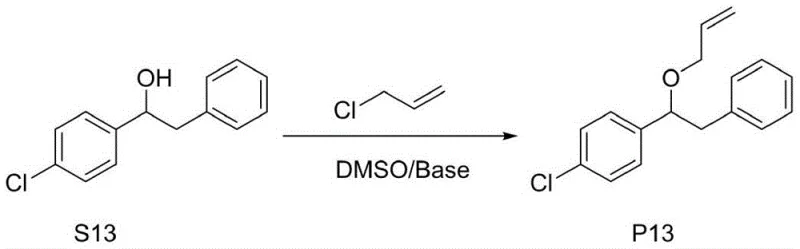
The landscape of organic synthesis for pharmaceutical intermediates is undergoing a significant transformation driven by the need for greener, more cost-effective processes. Patent CN110845416A introduces a groundbreaking O-allylation method for α-diaryl substituted ethanol that addresses critical pain points in traditional ether formation. This technology leverages a unique inorganic base-dimethyl sulfoxide (DMSO) super-base system to facilitate ether bond construction under remarkably mild conditions. For R&D directors and process chemists, this represents a departure from the hazardous and operationally complex protocols involving strong bases like sodium hydride or lithium amide. The ability to conduct these reactions in the presence of water, without the stringent requirement for anhydrous environments, lowers the barrier for industrial implementation. Furthermore, the process demonstrates exceptional versatility across a broad range of substrates, including those with sensitive functional groups, making it a robust candidate for the manufacturing of high-value active pharmaceutical ingredients (APIs) and fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of ether linkages, particularly in complex diaryl systems, has relied heavily on the Williamson ether synthesis or phase transfer catalysis (PTC), both of which present substantial challenges for large-scale manufacturing. Traditional laboratory methods often necessitate the use of potent, moisture-sensitive bases such as sodium hydride, sodium ethoxide, or potassium tert-butoxide to deprotonate the alcoholic hydroxyl group. These reagents demand strictly anhydrous reaction conditions; any introduction of moisture can lead to poor conversion rates or complete reaction failure, posing significant safety risks and equipment corrosion issues in a plant setting. Alternatively, industrial adaptations using phase transfer catalysts attempt to mitigate the anhydrous requirement but introduce new complexities. The screening for effective PTCs is often empirical and costly, as high-performance catalysts can be prohibitively expensive. Moreover, PTC processes typically require elevated temperatures ranging from 60°C to 120°C to achieve acceptable kinetics, which increases energy consumption and the risk of thermal degradation or side reactions. The removal of residual phase transfer catalysts from the final product adds further steps to the downstream processing, inflating both time and operational costs while complicating waste management protocols.
The Novel Approach
The innovative methodology disclosed in the patent data offers a compelling alternative by utilizing a DMSO-based super-base system generated in situ from inexpensive inorganic bases and water. This approach fundamentally alters the reaction landscape by enabling high-efficiency deprotonation of the alcoholic hydroxyl group without the need for hazardous pyrophoric reagents. By operating at mild temperatures between 35°C and 40°C, the process drastically reduces energy inputs and enhances safety profiles, making it inherently suitable for scale-up. The elimination of phase transfer catalysts not only simplifies the reaction mixture but also streamlines the workup procedure, as there is no need for complex extraction or chromatography steps solely dedicated to catalyst removal. This streamlined workflow translates directly into reduced cycle times and lower solvent consumption. The system's tolerance for aqueous conditions allows for the use of cheaper, commercially available inorganic bases like potassium hydroxide or sodium carbonate in their aqueous forms, bypassing the logistical challenges of handling and storing anhydrous solid bases. Consequently, this novel route provides a sustainable, economically viable pathway for producing O-allyl ethers that meets the rigorous quality standards required for pharmaceutical applications.
Mechanistic Insights into DMSO-Mediated O-Allylation
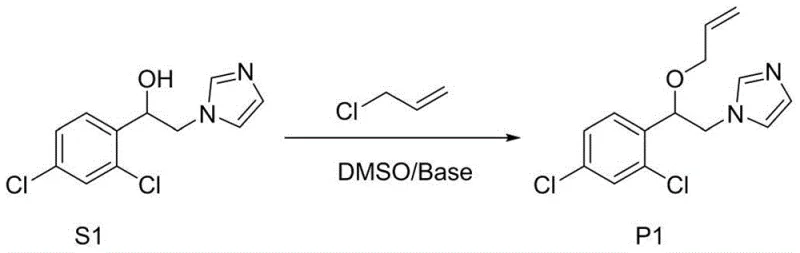
The efficacy of this synthetic route lies in the unique physicochemical properties of the dimethyl sulfoxide and inorganic base combination, which creates a "super-base" environment capable of efficient proton abstraction. In this system, the hydroxide ions from the inorganic base interact with the polar aprotic solvent DMSO, significantly enhancing their basicity and nucleophilicity compared to purely aqueous environments. This enhanced basicity allows for the rapid and quantitative deprotonation of the α-diaryl substituted ethanol substrate to form the corresponding alkoxide intermediate. Unlike traditional methods where the alkoxide might be unstable or prone to side reactions in protic solvents, the DMSO medium stabilizes the anionic species while maintaining high reactivity towards the electrophile, allyl chloride. The nucleophilic attack of the alkoxide on the allyl chloride proceeds via an SN2 mechanism, facilitated by the excellent solvating power of DMSO which helps to separate ion pairs and increase the availability of the naked alkoxide nucleophile. This mechanistic pathway ensures high conversion rates and minimizes the formation of elimination byproducts that are common when using bulky bases or high temperatures.
From an impurity control perspective, the mild thermal conditions (35-40°C) play a pivotal role in maintaining product integrity. High-temperature etherifications often suffer from competing elimination reactions where the alkyl halide loses hydrogen chloride to form dienes, or where the alcohol substrate undergoes dehydration. By keeping the reaction temperature low, the kinetic energy of the molecules is insufficient to overcome the activation barriers for these undesirable pathways, thereby channeling the reaction selectively towards the desired etherification. Additionally, the absence of transition metal catalysts or complex organic additives means that the impurity profile of the crude product is significantly cleaner. Analytical data from the patent examples indicates that crude GC purities can reach levels as high as 96.4% prior to any purification, suggesting that the reaction is highly chemoselective. This inherent purity reduces the burden on downstream purification units, whether crystallization or column chromatography, ensuring that the final API intermediate meets stringent regulatory specifications for residual solvents and heavy metals.
How to Synthesize O-Allyl Alpha-Diaryl Ethers Efficiently
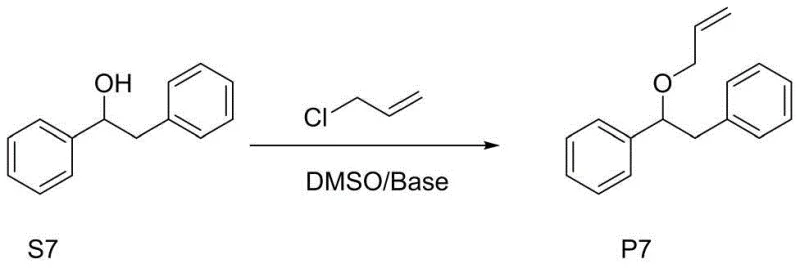
The practical implementation of this O-allylation technology involves a straightforward sequence of mixing and temperature control steps that are easily adaptable to standard reactor configurations. The process begins with the dissolution of the specific α-diaryl substituted ethanol substrate in dimethyl sulfoxide at ambient temperature, creating a homogeneous solution ready for activation. Following this, an aqueous solution of the chosen inorganic base, such as potassium hydroxide, is introduced dropwise to the reaction vessel. Critical to the success of this step is the maintenance of the reaction temperature below 40°C during the addition to manage the exotherm associated with neutralization and solvation. Once the base is fully added, the mixture is stirred to ensure complete formation of the reactive alkoxide species. Subsequently, the allylating agent, typically allyl chloride dissolved in DMSO, is added slowly to the activated mixture. The reaction is then maintained at a controlled temperature of 35-40°C until monitoring techniques such as TLC or HPLC confirm the complete consumption of the starting material. The detailed standardized synthesis steps for specific substrates are outlined in the guide below.
- Dissolve the alpha-diaryl substituted ethanol substrate in dimethyl sulfoxide (DMSO) at room temperature to form the initial reaction mixture.
- Prepare an aqueous solution of inorganic base (e.g., KOH or NaOH) and add it dropwise to the substrate solution while maintaining temperature below 40°C.
- Slowly add allyl chloride dissolved in DMSO, stir at 35-40°C until completion, then quench with water and extract with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DMSO-based O-allylation process offers tangible strategic advantages centered around cost stability and operational resilience. The primary driver for cost reduction is the substitution of expensive, specialty reagents with commodity chemicals. Traditional methods relying on sodium hydride or specialized phase transfer catalysts incur high raw material costs and require specialized storage infrastructure due to their hazardous nature. In contrast, the new method utilizes ubiquitous inorganic bases like potassium hydroxide and sodium carbonate, which are available in bulk quantities at a fraction of the cost. Furthermore, the elimination of the anhydrous requirement removes the need for expensive drying agents, molecular sieves, and the energy-intensive processes associated with maintaining moisture-free environments. This simplification of the bill of materials (BOM) directly contributes to a lower cost of goods sold (COGS), providing a competitive edge in price-sensitive markets. The simplified workup procedure also reduces solvent usage and waste disposal costs, aligning with modern sustainability goals and reducing the environmental compliance burden.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental simplification of the reaction engineering. By avoiding the use of pyrophoric bases like sodium hydride, facilities eliminate the need for inert atmosphere gloveboxes or nitrogen-blanketed reactors specifically for reagent handling, thereby reducing capital expenditure on specialized equipment. The ability to use aqueous base solutions means that water, the cheapest solvent, becomes a co-solvent rather than a contaminant, reducing the volume of organic solvents required for the reaction medium. Additionally, the high conversion rates observed in the patent examples minimize the amount of unreacted starting material that needs to be recovered or disposed of, improving overall atom economy. The absence of phase transfer catalysts removes a significant line item from the raw material budget and eliminates the downstream costs associated with removing these often difficult-to-separate organic salts from the product stream.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on widely available commodity chemicals significantly de-risks the production schedule. Reagents such as potassium hydroxide, sodium hydroxide, and dimethyl sulfoxide are produced globally in massive volumes, ensuring a stable supply even during market fluctuations that might affect niche fine chemicals. This abundance prevents bottlenecks that could arise from the scarcity of specialized catalysts or anhydrous reagents. Moreover, the mild reaction conditions reduce the wear and tear on reactor vessels and piping, leading to lower maintenance downtime and higher asset utilization rates. The robustness of the process against minor variations in moisture content also means that raw material specifications can be slightly relaxed without compromising yield, allowing procurement teams to source from a broader range of suppliers and negotiate better terms based on flexibility.
- Scalability and Environmental Compliance: The scalability of this O-allylation method is supported by its excellent thermal profile and manageable exotherm. Operating at temperatures near 40°C allows for efficient heat removal using standard cooling water systems, avoiding the need for cryogenic cooling or complex temperature control loops required for highly exothermic reactions with strong bases. This thermal safety margin is crucial for scaling from kilogram to multi-ton batches, ensuring consistent product quality and operator safety. Environmentally, the process generates less hazardous waste; the aqueous waste streams containing inorganic salts are easier to treat than organic waste laden with phase transfer catalysts or reactive metal residues. The high purity of the crude product reduces the need for extensive chromatographic purification, which is a major source of solvent waste in pharmaceutical manufacturing. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this O-allylation technology, based on the detailed experimental data provided in the patent documentation. These insights are intended to assist process development teams in evaluating the feasibility of adopting this route for their specific intermediate manufacturing needs. Understanding the nuances of base selection, temperature control, and purification strategies is essential for maximizing the yield and purity of the final ether products. The answers provided reflect the specific conditions and outcomes observed in the patent examples, offering a realistic baseline for expected performance in a production environment.
Q: Does this O-allylation process require strictly anhydrous conditions?
A: No, unlike traditional Williamson ether synthesis which requires rigorous anhydrous environments and strong bases like sodium hydride, this patented method utilizes an inorganic base-dimethyl sulfoxide super-base system that tolerates aqueous conditions, significantly simplifying industrial operations.
Q: What types of inorganic bases are compatible with this synthesis method?
A: The process is highly versatile regarding base selection, successfully employing alkali metal hydroxides such as sodium hydroxide and potassium hydroxide, as well as carbonates and bicarbonates, allowing procurement teams to optimize based on availability and cost.
Q: How does this method improve impurity profiles compared to phase transfer catalysis?
A: By eliminating the need for expensive phase transfer catalysts and operating at mild temperatures (35-40°C), the method reduces side reactions and simplifies post-treatment, resulting in crude products with high GC purity (often >95%) and easier refinement to pharmaceutical grades.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Diaryl Substituted Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert process chemists has thoroughly analyzed the potential of the DMSO-mediated O-allylation technology described in CN110845416A and is prepared to leverage this knowledge for your custom synthesis projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control and efficient solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards, providing you with a reliable supply of high-quality materials for your drug development pipelines.
We invite you to collaborate with us to explore how this advanced etherification technology can optimize your specific manufacturing processes. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current production volumes and cost structures. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a supplier, but to a strategic partner committed to driving efficiency and innovation in your supply chain. Please contact our technical procurement team today to request specific COA data for related intermediates and to discuss detailed route feasibility assessments for your target molecules. Let us help you achieve your production goals with superior chemistry and unmatched service reliability.