Advanced Manufacturing of Pregabalin Chiral Intermediates via Novel Grignard Route
Introduction to Next-Generation Pregabalin Intermediate Manufacturing
The global demand for Pregabalin, a potent calcium channel regulator approved for treating partial epilepsy and neuropathic pain, continues to drive innovation in its supply chain. Central to the efficient production of this high-value Active Pharmaceutical Ingredient (API) is the availability of optically pure chiral intermediates. Patent CN105481801B discloses a groundbreaking preparation method for (S)-4-isobutyl-dihydro-3H-furan-2-one, a critical precursor that enables the synthesis of high-purity Pregabalin. This technical insight report analyzes the proprietary three-step synthetic route detailed in the patent, which leverages a Grignard-mediated ring-opening strategy followed by sulfonylation and cyclization. For R&D directors and procurement leaders, understanding this methodology is essential, as it represents a significant departure from legacy processes that rely on expensive starting materials or hazardous reagents. By shifting towards commodity chemicals and mild reaction conditions, this technology offers a robust pathway for cost reduction in pharmaceutical intermediate manufacturing while ensuring stringent stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key lactone intermediate has been plagued by economic and safety inefficiencies. One prominent prior art method, reported by Bellotti et al., utilizes a beta-chiral acid ester as the starting material. While this route is conceptually short, it suffers from severe commercial drawbacks: the beta-chiral acid ester is prohibitively expensive and difficult to source in bulk quantities. Furthermore, the reduction step necessitates the use of borane-methyl sulfide, a reagent known for its toxicity, flammability, and complex waste disposal requirements. These factors create substantial barriers to safe, large-scale production.
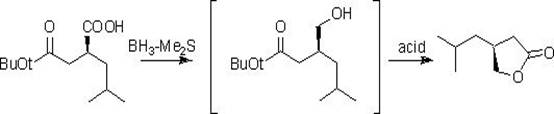
Another approach developed by Ok et al. attempts to utilize chiral epoxypropane derivatives. Although this method starts with cheaper materials, it introduces significant technical risks regarding regioselectivity. The chiral epoxide possesses multiple reactive sites, requiring extremely strict temperature control to prevent the formation of unwanted chiral isomers during the Grignard attack. In a commercial setting, maintaining such precise control over competing reaction pathways is difficult, often leading to lower optical purity and challenging purification burdens that erode overall process efficiency.
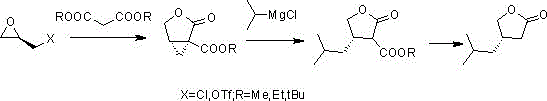
The Novel Approach
The methodology outlined in CN105481801B overcomes these historical bottlenecks through a streamlined three-step sequence that prioritizes safety and scalability. The process initiates with the regioselective ring-opening of an S-epoxypropane compound (Compound 2) using a Grignard reagent, specifically isopropyl magnesium bromide or chloride. Unlike previous attempts, this step is conducted under mild conditions (-10 to 0°C), which effectively suppresses side reactions while delivering high yields of the chiral hydroxy compound (Compound 3). The subsequent conversion of the hydroxyl group into a superior leaving group (such as a triflate or tosylate) activates the molecule for a clean nucleophilic substitution. Finally, the reaction with acetate or malonate derivatives, followed by acid-catalyzed hydrolysis and lactonization, efficiently constructs the target furanone ring. This route eliminates the need for dangerous borane reagents and expensive chiral acids, replacing them with stable, commodity-grade chemicals.
Mechanistic Insights into Grignard-Mediated Lactonization
The core innovation of this synthesis lies in the precise manipulation of stereochemistry during the Grignard addition and the subsequent cyclization. In the first step, the Grignard reagent attacks the less hindered carbon of the chiral epoxide ring. The patent specifies a molar ratio of 1:1.1 to 1:2.0 for the epoxide to Grignard reagent, ensuring complete conversion without excessive exothermicity. The use of solvents like THF or toluene facilitates this interaction, while the low temperature range (-10 to 0°C) is critical for preserving the chiral integrity of the resulting alcohol. This careful control prevents the racemization that often occurs in less optimized epoxide openings, ensuring that the downstream product retains the necessary (S)-configuration required for biological activity.
Following the formation of the chiral alcohol, the mechanism shifts to an SN2-type substitution facilitated by the installation of a sulfonate leaving group. Whether using trifluoromethanesulfonic anhydride to create a triflate or p-toluenesulfonyl chloride for a tosylate, the goal is to create a highly electrophilic center. When reacted with enolates generated from dimethyl malonate or tert-butyl acetate (using bases like sodium tert-butoxide or LDA), the displacement occurs with inversion of configuration, which is accounted for in the stereochemical design of the starting epoxide. The final stage involves a tandem hydrolysis and intramolecular esterification. Under acidic reflux conditions, the ester groups are hydrolyzed to carboxylic acids, which then spontaneously cyclize onto the adjacent ether oxygen to form the thermodynamically stable five-membered lactone ring. This cascade effectively locks the chiral center into the rigid furanone structure, yielding the target intermediate with high optical purity.
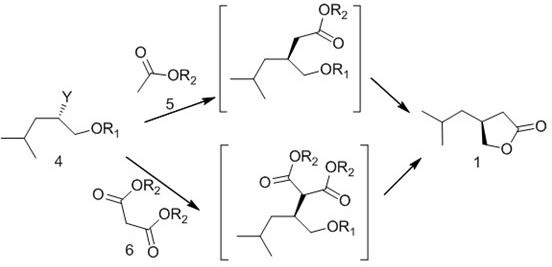
How to Synthesize (S)-4-isobutyl-dihydro-3H-furan-2-one Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to maximize yield and purity. The process begins with the preparation of the Grignard adduct, followed by activation and cyclization. Operators must ensure that moisture is rigorously excluded during the Grignard and sulfonylation steps to prevent reagent degradation. The final acid treatment serves a dual purpose: cleaving protecting groups and driving the equilibrium toward the cyclic lactone. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures required to execute this chemistry successfully, please refer to the standardized protocol below.
- Perform regioselective ring-opening of S-epoxypropane derivative using isopropyl magnesium bromide or chloride at -10 to 0°C to form the chiral hydroxy compound.
- Convert the hydroxyl group into a superior leaving group (triflate or tosylate) using sulfonylation reagents like trifluoroacetic anhydride or p-toluenesulfonyl chloride.
- Execute nucleophilic substitution with acetate or malonate derivatives followed by acid-catalyzed hydrolysis and intramolecular lactonization to yield the target furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic benefits beyond mere technical feasibility. The shift away from proprietary, high-cost starting materials like beta-chiral acid esters to ubiquitous commodity chemicals fundamentally alters the cost structure of the intermediate. By utilizing reagents such as isopropyl magnesium halides and dialkyl malonates, manufacturers can leverage established global supply chains, reducing vulnerability to price volatility and sourcing bottlenecks. Furthermore, the elimination of hazardous borane reagents simplifies regulatory compliance and waste management, leading to significant operational cost savings.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive chiral building blocks with affordable alternatives. The use of standard Grignard reagents and malonates, which are produced at massive scales for various industries, ensures a low baseline material cost. Additionally, the avoidance of cryogenic conditions (often below -78°C in other chiral syntheses) in favor of moderate cooling (-10 to 0°C) drastically reduces energy consumption and refrigeration infrastructure costs. The high overall yield reported in the examples further amplifies these savings by minimizing raw material waste per kilogram of finished product.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the reliance on chemically stable and widely available reagents. Unlike specialized chiral acids that may have limited suppliers, isopropyl magnesium bromide and sulfonyl chlorides are stocked by numerous chemical distributors worldwide. This diversity of supply sources mitigates the risk of production stoppages due to single-vendor dependencies. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different batches or vendors.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is significantly cleaner. The removal of borane-methyl sulfide eliminates a major toxicological hazard, simplifying plant safety protocols and reducing the burden on wastewater treatment facilities. The reaction solvents, primarily THF, dichloromethane, and ethyl acetate, are well-understood and easily recoverable through distillation, supporting green chemistry initiatives. The simplicity of the workup—often involving standard aqueous washes and crystallization—facilitates easy scale-up from pilot plants to multi-ton commercial reactors without the need for complex purification technologies like preparative HPLC.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pregabalin intermediate synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations.
Q: What are the primary safety advantages of this new synthesis route compared to traditional borane reduction methods?
A: The novel route eliminates the use of hazardous borane-methyl sulfide reducing agents, which pose significant safety risks and environmental pollution concerns in large-scale manufacturing. Instead, it utilizes standard Grignard reagents and mild sulfonylation conditions, drastically improving operational safety profiles.
Q: How does this method address the selectivity issues found in previous epoxide-based syntheses?
A: By strictly controlling the reaction temperature between -10°C and 0°C during the Grignard addition and utilizing specific leaving groups like triflates, the process minimizes the formation of chiral isomers. This precise control prevents the competing reaction pathways that often plague earlier epoxide opening strategies.
Q: Is this synthesis route suitable for multi-ton commercial scale-up?
A: Yes, the process is designed for scalability. It relies on cheap, commercially available raw materials such as isopropyl magnesium halides and malonates, and avoids cryogenic conditions or exotic catalysts. The workup procedures involve standard extraction and crystallization, making it highly amenable to industrial production from 100 kgs to 100 MT.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-isobutyl-dihydro-3H-furan-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals depends on a secure and high-quality supply of critical intermediates. As a leading CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this Grignard-based synthesis, including rigorous moisture control and low-temperature reactor capabilities. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of (S)-4-isobutyl-dihydro-3H-furan-2-one meets the exacting standards required for API synthesis, guaranteeing consistency and reliability for your downstream processes.
We invite you to collaborate with us to optimize your supply chain for Pregabalin production. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this advanced route can improve your margins. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing for this high-value chiral intermediate.
