Efficient Catalyst-Free Synthesis of 3,4-Dihydro-1,3-Oxazin-2-One Derivatives for Pharma
Efficient Catalyst-Free Synthesis of 3,4-Dihydro-1,3-Oxazin-2-One Derivatives for Pharma
The pharmaceutical industry is constantly seeking greener, more efficient pathways to synthesize complex heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN101973954A, which discloses a novel, environmentally benign method for the synthesis of 3,4-dihydro-[1,3]-oxazine-2-ketone derivatives. This technology represents a paradigm shift from traditional metal-catalyzed processes to a catalyst-free, aqueous-organic system, addressing major pain points regarding product purity, environmental impact, and operational safety. By utilizing readily available aryl-substituted nitroalkenes and 1,3-dicarbonyl compounds in a dimethylsulfoxide (DMSO) and water mixed system, this process achieves direct one-pot cyclization at moderate temperatures ranging from 50-100°C. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: the elimination of expensive transition metals simplifies downstream processing, while the use of water as a co-solvent drastically reduces raw material costs and waste disposal burdens, positioning it as a highly attractive route for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 3,4-dihydro-[1,3]-oxazin-2-one derivatives relied heavily on methodologies that introduced significant operational and regulatory hurdles. One prominent conventional approach, reported by Jagdale et al., utilized a Biginelli condensation reaction catalyzed by copper trifluoromethanesulfonate (Cu(OTf)2) in acetonitrile solvent. While this method could achieve decent yields, the reliance on a transition metal catalyst poses a severe risk of metal residue contamination in the final product, necessitating costly and time-consuming purification steps to meet stringent pharmaceutical standards. Furthermore, the use of acetonitrile, a toxic organic solvent, raises serious occupational health and safety concerns for plant operators and complicates environmental compliance due to volatile organic compound (VOC) emissions. Another existing method by Vovk et al. employed isonitriles and cyclohexanediones in toluene; however, the synthesis of chloro-substituted isonitriles is inherently complex, and these reagents often suffer from poor stability, limiting their utility in large-scale industrial applications. These legacy technologies collectively suffer from high input costs, safety hazards, and difficult waste management profiles.
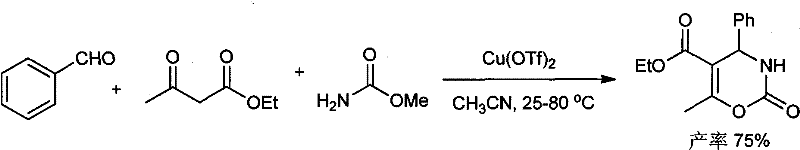
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN101973954A introduces a streamlined, catalyst-free protocol that leverages the unique reactivity of nitroalkenes in aqueous media. The core innovation lies in the use of a DMSO/Water binary solvent system, which facilitates the reaction between aryl-substituted nitrovinyl compounds and 1,3-dicarbonyl compounds without the need for any external catalyst. This approach not only eliminates the risk of heavy metal contamination but also capitalizes on the "on-water" effect, where hydrophobic interactions in the aqueous phase can accelerate reaction rates and improve selectivity. The reaction proceeds smoothly at temperatures between 50-100°C, with optimized conditions identified at 70°C, yielding the target oxazine derivatives in high purity. By replacing toxic solvents like acetonitrile or toluene with a water-rich system, this method significantly lowers the environmental footprint and operational costs, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for sustainable manufacturing practices.
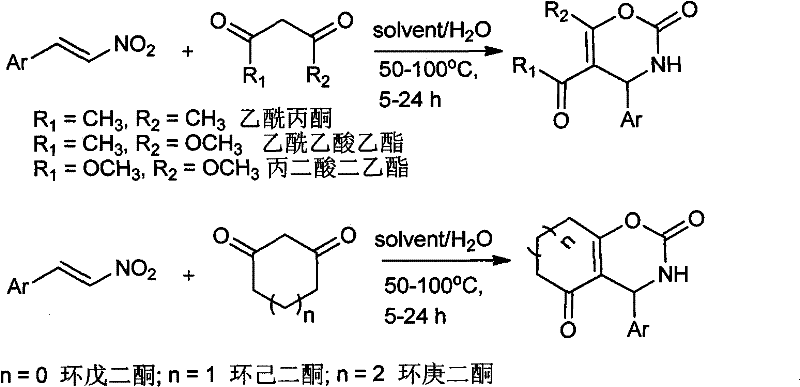
Mechanistic Insights into Catalyst-Free Aqueous Cyclization
The mechanistic pathway of this transformation is a fascinating example of how solvent engineering can replace traditional catalysis. In the absence of a Lewis acid catalyst like copper, the reaction is driven by the inherent electrophilicity of the nitroalkene and the nucleophilicity of the enol form of the 1,3-dicarbonyl compound. The presence of water plays a dual role: firstly, it acts as a proton shuttle, facilitating the tautomerization of the 1,3-dicarbonyl compound to its reactive enol form, which is crucial for the initial Michael addition step. Secondly, the high polarity of the water/DMSO mixture stabilizes the charged transition states and intermediates formed during the cyclization process. The reaction likely proceeds via a conjugate addition of the 1,3-dicarbonyl to the beta-position of the nitroalkene, followed by an intramolecular cyclization involving the carbonyl oxygen and the nitro group (which acts as a leaving group or is reduced in situ depending on specific conditions, though here it forms the oxazine ring directly through condensation). This cascade occurs efficiently under atmospheric conditions without the need for inert gas protection, demonstrating remarkable robustness against moisture and oxygen, which are typically detrimental to sensitive organometallic catalytic cycles.
From an impurity control perspective, the absence of metal catalysts fundamentally alters the impurity profile of the final product. In traditional metal-catalyzed reactions, side products often arise from metal-mediated decomposition or incomplete catalyst removal, leading to complex impurity spectra that are difficult to characterize and remove. In this aqueous system, the primary byproducts are likely limited to unreacted starting materials or simple hydrolysis products, which are generally easier to separate via standard extraction or crystallization techniques. The use of DMSO, a high-boiling polar aprotic solvent, ensures that all reactants remain in solution during the reaction phase, promoting homogeneity and consistent reaction kinetics. Upon completion, the addition of brine and extraction with ethyl acetate allows for the efficient partitioning of the organic product away from the water-soluble DMSO and inorganic salts. This simplified workup procedure not only enhances the overall yield—reported up to 88% in optimized examples—but also ensures a cleaner crude product, reducing the load on final purification columns and aligning perfectly with the requirements for high-purity pharmaceutical intermediate manufacturing.
How to Synthesize 3,4-Dihydro-1,3-Oxazin-2-One Derivatives Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow the optimized parameters derived from the patent examples to ensure maximum efficiency and reproducibility. The process begins by charging a reaction vessel with the aryl-substituted nitroalkene and the 1,3-dicarbonyl compound, typically in a molar ratio of 1:2 to drive the reaction to completion. The solvent system is critical; a 1:1 volume ratio of DMSO to water has been identified as the optimal medium, providing the right balance of solubility for organic reactants and the beneficial "on-water" acceleration effects. The mixture is then heated to 70°C and maintained under stirring for approximately 12 hours. Monitoring via TLC is recommended to confirm conversion. Post-reaction, the mixture is treated with saturated brine and extracted with ethyl acetate, followed by drying over anhydrous sodium sulfate and solvent removal. The detailed standardized synthesis steps see the guide below.
- Mix aryl-substituted nitroalkene and 1,3-dicarbonyl compound (e.g., acetylacetone) in a reaction vessel.
- Add a solvent system comprising Dimethyl Sulfoxide (DMSO) and Water, preferably in a 1: 1 volume ratio.
- Heat the mixture to 70°C and stir for approximately 12 hours under atmospheric conditions to achieve high yields up to 88%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free synthesis route offers transformative advantages that extend far beyond simple chemistry. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized transition metal catalysts like copper trifluoromethanesulfonate, which can be expensive and subject to supply volatility, manufacturers can rely on commodity chemicals that are universally available and price-stable. Furthermore, the removal of toxic solvents like acetonitrile reduces the regulatory burden associated with solvent recovery and disposal, leading to substantial cost savings in waste management and environmental compliance. The robustness of the reaction under atmospheric conditions also means that facilities do not require expensive inert gas infrastructure, lowering both capital expenditure (CAPEX) for new plants and operational expenditure (OPEX) for existing ones. These factors combine to create a manufacturing process that is not only cheaper but also significantly more resilient to market fluctuations and supply disruptions.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials, as these reagents are often among the most expensive components in a synthetic sequence. Additionally, the absence of metal residues negates the need for specialized scavenging resins or complex chromatographic purification steps designed to lower metal content to ppm levels, which are resource-intensive and time-consuming. The use of water as a primary co-solvent further drives down costs, as it is the cheapest and most abundant solvent available, replacing costly organic solvents that require rigorous recycling protocols. This streamlined process flow results in a leaner manufacturing operation with lower variable costs per kilogram of product, directly improving margin potential for high-volume API production.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks such as nitroalkenes and acetylacetone ensures a stable and diversified supply base, reducing the risk of production stoppages due to single-source supplier issues. The simplicity of the reaction conditions—operating at moderate temperatures without inert gas protection—means that the process can be easily transferred between different manufacturing sites or scaled up in multipurpose reactors without requiring specialized equipment modifications. This flexibility enhances supply continuity, allowing manufacturers to respond rapidly to changes in demand from downstream pharmaceutical clients. Moreover, the reduced hazard profile of the reagents and solvents simplifies logistics and storage requirements, minimizing the risk of delays caused by hazardous material transport regulations.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method make it highly scalable and compliant with increasingly stringent global environmental regulations. The aqueous waste stream generated is significantly less toxic than those from traditional organic solvent-heavy processes, simplifying wastewater treatment and reducing the carbon footprint of the manufacturing site. The high atom economy and efficient conversion rates minimize the generation of chemical waste, aligning with corporate sustainability goals and ESG (Environmental, Social, and Governance) criteria. This environmental compatibility not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain, facilitating smoother audits and approvals from major multinational drug companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for R&D teams evaluating the route for potential integration into their existing pipelines.
Q: What are the optimal reaction conditions for this oxazine synthesis?
A: According to patent CN101973954A, the optimal conditions involve using a DMSO/Water (1:1) solvent system at a temperature of 70°C for 12 hours, which balances reaction time and yield effectively.
Q: Does this method require expensive transition metal catalysts?
A: No, a key advantage of this technology is that it operates without any additional catalyst, eliminating the risk of heavy metal residues and reducing purification costs significantly.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully reacting various aryl-substituted nitroalkenes (including those with electron-withdrawing or donating groups) with 1,3-dicarbonyl compounds like acetylacetone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydro-1,3-Oxazin-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this catalyst-free synthesis route to revolutionize the production of antiviral intermediates and other critical pharmaceutical building blocks. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required by this technology, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that consistency and quality are paramount in the pharmaceutical industry, and our dedicated technical team is committed to delivering products that exceed expectations.
We invite you to explore how this innovative synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us partner with you to bring high-quality, cost-effective pharmaceutical intermediates to market faster and more sustainably.
