Revolutionizing Ticagrelor Intermediate Production: A Safer, Scalable Synthetic Strategy
Revolutionizing Ticagrelor Intermediate Production: A Safer, Scalable Synthetic Strategy
The global demand for potent antiplatelet agents continues to surge, driven by the increasing prevalence of acute coronary syndromes and the widespread adoption of dual antiplatelet therapy protocols. At the heart of this therapeutic class lies Ticagrelor, a molecule whose complex architecture demands highly efficient and robust supply chains for its key building blocks. Recent intellectual property developments, specifically patent CN113024471A, have unveiled a transformative synthetic methodology for producing 4,6-dichloro-2-propylmercapto-5-aminopyridine, a pivotal intermediate in the Ticagrelor value chain. This technological breakthrough addresses long-standing bottlenecks in pharmaceutical manufacturing by replacing hazardous nitration processes with a sophisticated protective group strategy. For R&D directors and supply chain leaders, understanding this shift is critical, as it promises not only enhanced safety profiles but also substantial improvements in yield consistency and environmental compliance. The following analysis dissects the mechanistic advantages and commercial implications of this novel route, positioning it as a cornerstone for future reliable pharmaceutical intermediates supplier partnerships.
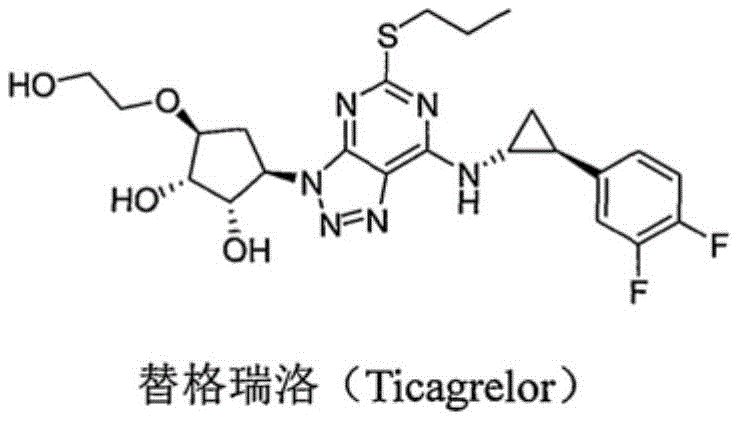
Ticagrelor represents a significant advancement in cardiovascular medicine, functioning as a reversible P2Y12 receptor antagonist with rapid onset and offset of action. The molecular complexity of this drug necessitates a multi-step synthesis where the integrity of the pyrimidine core is paramount. As illustrated in the structural diagram, the presence of the cyclopentyl triol moiety and the specific halogenated pyrimidine ring requires precise chemical construction. The intermediate 4,6-dichloro-2-propylmercapto-5-aminopyridine serves as the foundational scaffold upon which the rest of the molecule is built. Any inefficiency or impurity at this stage propagates through the entire synthesis, impacting final API quality and cost. Therefore, optimizing the production of this specific heterocycle is not merely a chemical exercise but a strategic imperative for maintaining competitive advantage in the cardiovascular drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4,6-dichloro-2-propylmercapto-5-aminopyridine has relied on pathways fraught with significant operational and environmental hazards. Traditional routes, such as those disclosed in earlier patents like WO2011036479, typically involve the nitration of a pyrimidine precursor using concentrated nitric acid. This step introduces severe safety risks associated with exothermic reactions and the handling of corrosive acids, while simultaneously generating large volumes of acidic wastewater that require costly treatment. Furthermore, the subsequent reduction of the nitro group often necessitates either catalytic hydrogenation using expensive noble metals, which are prone to poisoning, or metal reduction using iron, which produces massive quantities of iron sludge waste. These legacy methods create a heavy burden on waste management systems and complicate the purification process, often resulting in lower overall yields and inconsistent batch-to-batch quality. The reliance on such archaic chemistry limits the ability of manufacturers to scale production efficiently while meeting modern green chemistry standards.
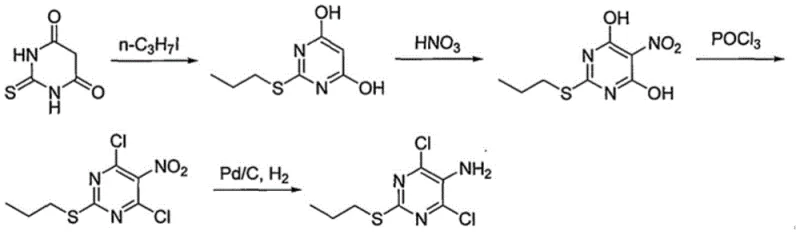
The Novel Approach
In stark contrast, the methodology outlined in CN113024471A introduces a paradigm shift by eliminating the nitration step entirely. Instead of building the nitrogen functionality through dangerous oxidation-reduction cycles, this innovative route utilizes a pre-functionalized starting material: a protected diethyl aminomalonate. By engaging this protected precursor in a condensation reaction with thiourea, the pyrimidine ring is constructed with the amino group already in place but safely masked. This strategic use of a protecting group prevents unwanted side reactions during the subsequent alkylation phase, a common failure point in unprotected syntheses. The result is a streamlined four-step process that bypasses the need for high-pressure hydrogenation equipment and avoids the generation of heavy metal waste. This approach not only simplifies the equipment requirements, making it accessible for a wider range of manufacturing facilities, but also aligns perfectly with the industry's drive towards sustainable and cost reduction in API manufacturing.
Mechanistic Insights into Protective Group-Mediated Cyclization
The core innovation of this synthetic pathway lies in the meticulous control of chemoselectivity during the alkylation step. In conventional attempts to synthesize this intermediate starting from unprotected aminomalonate derivatives, the free amino group acts as a competing nucleophile. When exposed to alkylating agents like bromopropane, the nitrogen atom readily undergoes N-alkylation, leading to a complex mixture of mono- and di-substituted impurities that are difficult to separate. The new method circumvents this by employing an acetyl-protected aminomalonate. The electron-withdrawing nature of the acetyl group significantly reduces the nucleophilicity of the nitrogen atom, effectively "turning off" its reactivity towards the alkyl halide. Consequently, when the reaction mixture is treated with bromopropane under basic conditions, the alkylation occurs exclusively at the sulfur atom of the thiourea-derived moiety. This high degree of regioselectivity ensures that the propyl group is installed precisely where needed, minimizing the formation of byproducts and maximizing the yield of the desired sulfide intermediate.
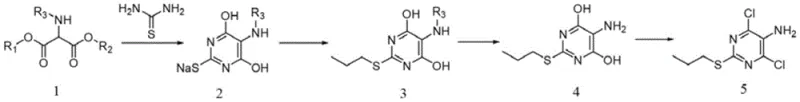
Following the successful S-alkylation, the process proceeds through a carefully orchestrated deprotection and chlorination sequence. The removal of the acetyl protecting group is achieved under mild acidic conditions, typically using hydrochloric acid in methanol, which regenerates the free amino group without compromising the integrity of the newly formed thioether bond. This step is crucial as it restores the reactivity of the amine for potential downstream modifications while maintaining the purity established in the previous step. Finally, the introduction of chlorine atoms at the 4 and 6 positions of the pyrimidine ring is accomplished using phosphorus oxychloride in the presence of an organic base. This chlorination step converts the hydroxyl groups into chlorides, yielding the final target molecule, 4,6-dichloro-2-propylmercapto-5-aminopyridine. The entire sequence demonstrates a masterful application of protective group chemistry to solve a persistent selectivity problem, offering a robust solution for the commercial scale-up of complex heterocycles.
How to Synthesize 4,6-Dichloro-2-Propylmercapto-5-Aminopyridine Efficiently
The execution of this synthetic route requires precise control over reaction parameters to fully realize its potential benefits. The process begins with the cyclization of the protected aminomalonate and thiourea, followed by the critical alkylation step where temperature and base concentration must be tightly regulated to prevent deprotection or over-alkylation. Subsequent steps involve acid-mediated deprotection and final chlorination, each requiring specific workup procedures to ensure high purity. The detailed standardized operating procedures, including specific molar ratios, solvent choices, and temperature profiles for each transformation, are essential for reproducibility. For technical teams looking to implement this technology, adhering to the optimized conditions described in the patent examples is vital to achieving the reported yields and purity levels. The detailed standardized synthesis steps are provided below.
- Cyclization: React protected diethyl aminomalonate with thiourea under alkaline catalysis to form the pyrimidine ring structure.
- Selective Alkylation: Perform S-alkylation using bromopropane in a mixed solvent system with controlled base concentration to ensure high selectivity.
- Deprotection and Chlorination: Remove the amino protecting group via acid hydrolysis, followed by chlorination using phosphorus oxychloride to yield the final dichloro product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical advantages that extend beyond simple yield improvements. By fundamentally altering the chemistry to avoid hazardous reagents and complex waste streams, the process directly impacts the bottom line through reduced operational expenditures and mitigated regulatory risks. The elimination of nitration and metal reduction steps removes the need for specialized corrosion-resistant equipment and expensive catalyst recovery systems, thereby lowering capital expenditure barriers. Furthermore, the simplified purification profile reduces the consumption of solvents and energy during downstream processing. These factors combine to create a more resilient supply chain capable of delivering high-purity Ticagrelor intermediates with greater consistency and reliability, ensuring uninterrupted production schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the complete removal of the nitration and subsequent reduction steps. Traditional methods rely on costly catalytic hydrogenation or generate vast amounts of iron sludge that incur high disposal fees. By switching to a protective group strategy, the new process utilizes inexpensive, commodity-grade reagents like bromopropane and thiourea. Additionally, the avoidance of noble metal catalysts eliminates the risk of catalyst poisoning and the associated costs of metal recovery or replacement. The streamlined workflow also reduces labor hours and utility consumption, as fewer distinct unit operations are required to reach the final intermediate. This holistic reduction in material and processing costs translates into a more competitive pricing structure for the final API, allowing pharmaceutical companies to optimize their COGS without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized reagents or processes that are sensitive to minor deviations. The new method enhances reliability by utilizing robust chemistry that is less prone to failure. The starting materials, such as protected aminomalonates, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. Moreover, the process operates under milder conditions compared to high-pressure hydrogenation or aggressive nitration, reducing the likelihood of unplanned shutdowns due to equipment failure or safety incidents. The high selectivity of the alkylation step ensures consistent batch quality, minimizing the need for reprocessing or rejection of out-of-specification material. This predictability allows supply chain planners to maintain leaner inventories while confidently meeting delivery commitments to API customers.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthetic route becomes a critical factor in vendor selection. This novel method is inherently greener, as it avoids the generation of nitrous oxide emissions and heavy metal-contaminated wastewater. The absence of iron sludge simplifies waste treatment protocols and reduces the environmental liability associated with production. From a scalability perspective, the reaction conditions are easily transferable from laboratory to pilot and commercial scales without requiring exotic engineering solutions. The use of standard solvents and ambient pressure reactions facilitates rapid capacity expansion to meet surging market demand. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental regulations but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthetic pathway is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding safety, yield optimization, and scalability, drawing directly from the experimental data and comparative analysis provided in the patent literature. These insights are intended to clarify how the protective group strategy resolves historical challenges in Ticagrelor intermediate synthesis. Stakeholders are encouraged to review these points to assess the feasibility of integrating this technology into their existing manufacturing frameworks.
Q: Why is the new synthetic route safer than traditional nitration methods?
A: Traditional routes often utilize hazardous nitric acid for nitration and subsequent metal reduction, generating significant acidic waste and iron sludge. The novel method disclosed in CN113024471A completely eliminates the nitration step, utilizing a protective group strategy that avoids high-risk reactions and heavy metal waste, significantly improving environmental compliance and operational safety.
Q: How does the amino protection strategy improve reaction yield?
A: In conventional approaches starting from unprotected aminomalonate, the free amino group competes during the alkylation step, leading to N-alkylation impurities and low conversion rates. By employing a protected aminomalonate derivative, the new method ensures exclusive S-alkylation on the sulfur atom, drastically reducing impurity profiles and enhancing the overall yield of the critical propylmercapto intermediate.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial scalability. It utilizes commercially available raw materials like diethyl aminomalonate and thiourea, avoids expensive noble metal catalysts required for hydrogenation, and operates under mild conditions. The simplified post-treatment procedures, such as filtration and crystallization rather than complex chromatographic separations, make it highly viable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dichloro-2-Propylmercapto-5-Aminopyridine Supplier
The transition to safer and more efficient synthetic routes requires a manufacturing partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, ensuring that the delicate balance of the protective group strategy is maintained throughout the production lifecycle. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4,6-dichloro-2-propylmercapto-5-aminopyridine meets the highest international standards. Our commitment to continuous improvement means we are constantly optimizing processes to deliver superior value to our global partners.
We invite pharmaceutical companies and contract manufacturers to explore the potential of this advanced synthetic technology for their Ticagrelor supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovation can drive efficiency in your operations. Contact us today to discuss how we can support your long-term strategic goals with reliable, high-quality intermediates.
