Advanced Visible Light Photocatalysis for Efficient Phenolic Ester Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient methodologies for constructing essential structural motifs, among which the ester bond remains a cornerstone. Patent CN108191651B introduces a groundbreaking preparation method for synthesizing phenolic esters through a thiocarboxylic acid-mediated visible light-catalyzed phenol acylation reaction. This innovation represents a significant departure from classical stoichiometric activation strategies, leveraging the power of photoredox catalysis to drive chemical transformations under exceptionally mild conditions. By utilizing readily available thiocarboxylic acids as acylating reagents and a ruthenium-based photosensitizer, this process achieves high-yielding, site-specific esterification at normal temperature and pressure. The technological breakthrough lies not only in the efficiency of the bond formation but also in the remarkable tolerance for diverse functional groups, positioning it as a highly reliable phenolic ester supplier technology for complex molecule assembly.
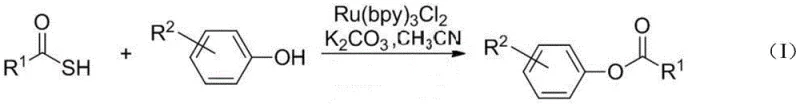
Traditional methods for constructing ester bonds, particularly phenolic esters, have long been plagued by significant operational and safety challenges that hinder scalable manufacturing. Conventional protocols typically rely on highly electrophilic and often hazardous activating reagents such as dicyclohexylcarbodiimide (DCC), diethyl azodicarboxylate (DEAD), or sensitive acid chlorides and anhydrides. These reagents frequently necessitate stringent anhydrous conditions, low temperatures, and the use of toxic solvents, leading to cumbersome operational procedures and substantial waste generation. Furthermore, a critical limitation of these classical approaches is their lack of chemoselectivity; when a substrate contains both phenolic and alcoholic hydroxyl groups, distinguishing between them to achieve site-specific phenolic esterification is notoriously difficult without extensive protecting group strategies. This lack of specificity increases synthetic steps, reduces overall atom economy, and escalates production costs, creating a bottleneck for the cost reduction in pharmaceutical intermediates manufacturing.
In stark contrast, the novel approach detailed in the patent utilizes a visible light-driven catalytic cycle that fundamentally alters the reaction landscape. By employing thiocarboxylic acids as stable and manageable acyl donors, the method circumvents the need for corrosive acid chlorides. The reaction proceeds efficiently under ambient atmospheric conditions using simple potassium carbonate as a base and acetonitrile as a solvent, driven by the energy of visible light rather than thermal activation. This shift to photochemical activation allows for the precise generation of acyl radical intermediates that selectively react with the phenolic oxygen. The result is a highly site-specific transformation that leaves aliphatic alcohols untouched, thereby eliminating the need for complex protection-deprotection sequences. This streamlined process not only simplifies the workflow but also enhances the safety profile of the manufacturing environment, making it an attractive option for the commercial scale-up of complex polymer additives and fine chemicals.
Mechanistic Insights into Ru(bpy)3Cl2-Catalyzed Phenolic Acylation
The core of this transformative technology lies in the sophisticated interplay between the photosensitizer and the substrate under visible light irradiation. The catalyst, tris(2,2'-bipyridine)ruthenium(II) dichloride hexahydrate (Ru(bpy)3Cl2), absorbs photons from the visible light source to reach an excited metal-to-ligand charge transfer (MLCT) state. In this excited state, the ruthenium complex possesses altered redox potentials that enable it to participate in single-electron transfer (SET) processes with the thiocarboxylic acid substrate. This interaction facilitates the homolytic cleavage of the sulfur-carbon bond or generates a thiyl radical species, ultimately leading to the formation of a highly reactive acyl radical. This radical species is then captured by the phenolic substrate, which has been activated by the mild base, to form the desired ester bond. The catalytic cycle is closed by the regeneration of the ground-state ruthenium catalyst, ensuring that only catalytic amounts of the expensive metal complex are required to drive the reaction to completion with high turnover numbers.
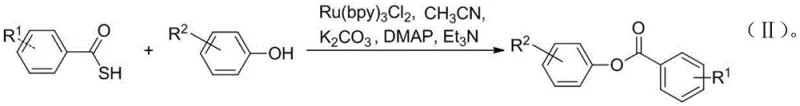
Beyond the fundamental bond formation, the mechanism offers profound advantages regarding impurity control and product purity, which are critical concerns for any R&D Director. The mildness of the visible light conditions ensures that sensitive functional groups on the aromatic rings, such as halogens, nitro groups, cyano groups, and esters, remain intact throughout the reaction. Traditional thermal acylation methods often induce side reactions like nucleophilic aromatic substitution or hydrolysis of sensitive esters under harsh basic or acidic conditions. However, the radical nature of this photocatalytic pathway is remarkably chemoselective for the O-H bond of the phenol. This specificity minimizes the formation of by-products and simplifies the downstream purification process, often requiring only a standard silica gel column chromatography to achieve high-purity phenolic ester products. The ability to maintain the integrity of complex molecular architectures while installing the ester functionality makes this method particularly valuable for late-stage functionalization in drug discovery pipelines.
How to Synthesize Phenolic Esters Efficiently
The practical implementation of this visible light-catalyzed protocol is designed for ease of operation and scalability, bridging the gap between academic innovation and industrial application. The general procedure involves dissolving the thiocarboxylic acid and the phenolic substrate in acetonitrile, followed by the addition of the ruthenium photocatalyst and potassium carbonate base. The reaction mixture is then stirred under open-air conditions at room temperature while being irradiated with a standard 45W fluorescent lamp. This setup eliminates the need for specialized high-pressure reactors or cryogenic cooling systems, significantly lowering the barrier to entry for adoption. After the reaction reaches completion, typically within a few hours, the workup involves a straightforward aqueous extraction and purification sequence. For a detailed standardized synthesis guide including specific molar ratios and purification parameters, please refer to the structured instructions below.
- Dissolve the thiocarboxylic acid substrate and phenolic compound in acetonitrile solvent within a reaction vessel.
- Add the photocatalyst Ru(bpy)3Cl2 and the base K2CO3 to the reaction mixture under ambient atmosphere.
- Irradiate the mixture with a 45W fluorescent lamp at room temperature, followed by standard aqueous workup and silica gel purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this visible light-catalyzed synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like acid chlorides and coupling agents such as DCC drastically reduces the safety risks associated with storage, handling, and disposal, leading to substantial cost savings in terms of safety infrastructure and waste management compliance. Furthermore, the use of ambient temperature and pressure conditions significantly lowers the energy consumption profile of the manufacturing process compared to traditional thermal methods that require prolonged heating or cooling. This energy efficiency translates directly into reduced operational expenditures, enhancing the overall cost competitiveness of the final phenolic ester products in the global market.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the use of inexpensive and commercially available starting materials. Thiocarboxylic acids are generally more stable and easier to handle than their acid chloride counterparts, reducing losses due to hydrolysis during storage and transport. Additionally, the catalytic nature of the ruthenium complex means that only minute quantities are needed to drive the reaction, minimizing the cost contribution of the precious metal catalyst. The simplified workup procedure, which avoids complex quenching steps often required for reactive acylating agents, further reduces labor time and solvent usage, collectively driving down the cost of goods sold (COGS) for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the robustness of the reaction conditions. Because the process tolerates a wide range of functional groups and operates under mild conditions, it is less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or moisture levels. This reliability ensures consistent product quality and yield, which is crucial for maintaining steady supply lines to downstream customers. Moreover, the stability of the thiocarboxylic acid reagents allows for longer shelf lives and more flexible inventory management, reducing the risk of supply disruptions caused by reagent degradation.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the simplicity of the reactor requirements. The use of visible light sources, such as LED arrays or fluorescent lamps, can be easily integrated into existing flow chemistry setups or batch reactors without major capital investment. The environmental footprint is also markedly reduced, as the process generates fewer toxic by-products and utilizes greener solvent systems. This alignment with green chemistry principles not only aids in meeting increasingly stringent environmental regulations but also enhances the corporate sustainability profile, which is becoming a key differentiator in supplier selection processes for multinational corporations.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial applicability of this patented technology, we have compiled a set of frequently asked questions based on the experimental data and mechanistic understanding provided in the patent documentation. These answers are designed to provide clarity on the operational parameters, substrate scope, and potential limitations of the visible light-catalyzed phenolic ester synthesis. Understanding these details is essential for evaluating the feasibility of integrating this method into your existing manufacturing workflows or research programs.
Q: What is the primary advantage of this visible light catalyzed method over traditional acylation?
A: The primary advantage is the exceptional chemoselectivity and mild reaction conditions. Unlike traditional methods requiring corrosive acid chlorides or harsh activating agents, this protocol operates at room temperature and atmospheric pressure, specifically targeting phenolic hydroxyl groups even in the presence of alcoholic hydroxyls.
Q: What is the role of the Ru(bpy)3Cl2 catalyst in this reaction?
A: Ru(bpy)3Cl2 acts as a photosensitizer that absorbs visible light energy to generate excited states. These excited states facilitate the single-electron transfer processes necessary to activate the thiocarboxylic acid, generating the reactive acyl radical species required for the esterification without needing high thermal energy.
Q: Does this method tolerate sensitive functional groups on the substrate?
A: Yes, the method demonstrates broad functional group tolerance. The mild nature of the visible light catalysis allows for the successful conversion of substrates containing halogens, nitro groups, cyano groups, and esters without degradation or side reactions, making it ideal for complex pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenolic Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modern organic synthesis and are committed to delivering high-quality phenolic esters produced via this advanced methodology. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative patent can be realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of phenolic ester meets the exacting standards required by the global pharmaceutical and agrochemical industries. We understand that consistency and reliability are paramount, and our dedicated technical team is prepared to support your specific project needs with precision and expertise.
We invite you to explore how this cutting-edge synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our phenolic ester portfolio and to discuss route feasibility assessments for your custom synthesis projects. Let us partner with you to bring efficient, sustainable, and cost-effective chemical solutions to market faster.
