Scalable Visible Light Catalysis for High-Purity Beta-Trifluoromethyl Alcohol Intermediates
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to introduce trifluoromethyl groups into organic scaffolds, as this modification profoundly enhances metabolic stability and lipophilicity. A groundbreaking approach detailed in patent CN111205185B introduces a visible light-catalyzed preparation method for beta-trifluoromethyl alcohols that addresses critical limitations in current synthetic methodologies. This innovative protocol utilizes aliphatic olefins and sodium trifluoromethanesulfinate as substrates, employing an organic manganese salt as a photocatalyst under an oxygen-containing atmosphere. By leveraging visible light energy at room temperature, this method achieves mild and efficient conversion, marking a significant departure from harsh thermal conditions. The general structural scope of the aliphatic olefin substrates is defined by specific carbon chain lengths and substituents, allowing for versatile application in complex molecule synthesis.
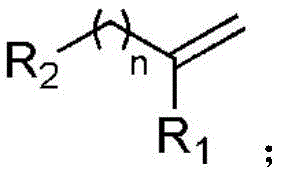
Furthermore, the resulting beta-trifluoromethyl alcohol products exhibit a robust structural framework suitable for downstream functionalization, as illustrated by the generalized product structure where the trifluoromethyl group and hydroxyl group are installed with high regioselectivity. This technology represents a pivotal shift towards greener chemistry, utilizing abundant raw materials and avoiding toxic reagents, thereby positioning it as a highly attractive route for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio. The ability to operate under ambient conditions without specialized high-pressure equipment further underscores its potential for immediate industrial adoption.
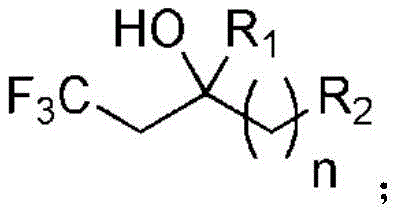
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-trifluoromethyl alcohols has been plagued by significant technical and economic hurdles that hinder large-scale manufacturing. Traditional thermal reactions often require expensive and specialized trifluoromethylating reagents, such as S-(trifluoromethyl)dibenzothiophenium tetrafluoroborate, which drastically inflate the cost of goods sold. Moreover, many existing protocols rely on noble metal catalysts like iridium complexes, which are not only costly but also pose challenges regarding residual metal removal in pharmaceutical applications. Another critical drawback is the reliance on dangerous oxidants; processes utilizing tert-butyl hydroperoxide or benzoquinone introduce substantial safety risks and generate hazardous waste streams that complicate environmental compliance. Additionally, the substrate scope of these conventional methods is frequently restricted to aromatic olefins, failing to accommodate the aliphatic chains prevalent in many drug candidates, thus limiting their utility in cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast, the visible light-catalyzed method described in the patent offers a transformative solution by replacing thermal energy with photon energy and expensive metals with earth-abundant manganese. This novel approach utilizes sodium trifluoromethanesulfinate, a stable and inexpensive solid, as the trifluoromethyl source, effectively lowering the raw material barrier. The use of air or oxygen as the terminal oxidant eliminates the need for hazardous peroxides, aligning perfectly with green chemistry principles and enhancing workplace safety. Crucially, this method demonstrates exceptional compatibility with various substituted aliphatic olefins, overcoming the selectivity issues that plague thermal reactions which often produce mixtures of alcohols and ketones. The reaction proceeds smoothly at room temperature (23-25°C), removing the energy burden associated with heating and allowing for simpler reactor designs, which is a key factor for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Mechanistic Insights into Mn-Catalyzed Visible Light Trifluoromethylation
The core of this technological advancement lies in the unique photoredox cycle facilitated by the manganese acetylacetonate catalyst under blue LED irradiation. Upon absorption of visible light photons, the manganese catalyst enters an excited state capable of engaging in single-electron transfer processes with the sodium trifluoromethanesulfinate. This interaction generates a trifluoromethyl radical species along with sulfur dioxide and a reduced manganese center. The highly reactive trifluoromethyl radical then adds across the carbon-carbon double bond of the aliphatic olefin substrate, forming a new carbon-carbon bond and a transient alkyl radical intermediate. This step is critical for determining the regioselectivity of the final product, ensuring the trifluoromethyl group attaches at the desired position relative to the functional groups.
Subsequently, the alkyl radical intermediate undergoes oxidation by molecular oxygen from the air, regenerating the active manganese catalyst and forming a peroxyl species that eventually collapses to yield the beta-trifluoromethyl alcohol. This oxidative turnover is essential for the catalytic cycle to continue without the accumulation of inactive metal species. The mild conditions prevent the over-oxidation of the alcohol to a ketone, a common side reaction in thermal methods, thereby ensuring high purity and minimizing the formation of difficult-to-separate impurities. The mechanistic elegance of using air as the oxidant not only simplifies the reaction setup but also ensures that the only byproduct is water or benign sulfur species, significantly reducing the environmental footprint of the synthesis.

How to Synthesize Beta-Trifluoromethyl Alcohol Efficiently
The practical implementation of this synthesis involves a straightforward procedure that can be easily adapted for both laboratory discovery and pilot plant operations. The process begins with the precise weighing of the aliphatic olefin substrate and four equivalents of sodium trifluoromethanesulfinate to ensure complete conversion. These solids are introduced into a reaction vessel containing a mixed solvent system of 1,4-dioxane and acetone, which provides the optimal polarity for solubilizing both organic and inorganic components. Following the addition of 10 mol% manganese acetylacetonate, the mixture is subjected to irradiation from a 40W blue LED lamp while being stirred in an open-air environment or under an oxygen balloon. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the aliphatic olefin substrate and sodium trifluoromethanesulfinate (4 equivalents) in a solvent system of 1,4-dioxane and acetone.
- Add 10 mol% of manganese acetylacetonate (Mn(acac)3) as the catalyst to the reaction vessel under an oxygen-containing atmosphere such as air.
- Irradiate the mixture with a 40W blue LED light source at room temperature (23-25°C) for 24 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible light technology translates into tangible strategic benefits that extend beyond simple yield improvements. The shift from noble metal catalysts to manganese represents a fundamental change in the cost structure of the synthesis, as manganese salts are orders of magnitude cheaper and more readily available globally than iridium or ruthenium complexes. This substitution removes a major bottleneck in raw material sourcing, ensuring a more resilient supply chain that is less susceptible to geopolitical fluctuations affecting precious metal markets. Furthermore, the elimination of hazardous peroxides reduces the regulatory burden and insurance costs associated with storing and handling dangerous chemicals, directly impacting the bottom line through lower operational overheads.
- Cost Reduction in Manufacturing: The replacement of expensive iridium catalysts with commercially available manganese acetylacetonate results in a drastic reduction in catalyst costs, which is particularly significant when scaling to multi-kilogram production. Additionally, the use of air as a free and abundant oxidant removes the recurring expense of purchasing stoichiometric chemical oxidants, further driving down the variable costs per batch. The mild room temperature conditions also lead to substantial energy savings by eliminating the need for heating mantles or oil baths, contributing to a leaner and more cost-efficient manufacturing process overall.
- Enhanced Supply Chain Reliability: By utilizing sodium trifluoromethanesulfinate, a stable solid reagent, the process avoids the logistical complexities and safety hazards associated with handling gaseous trifluoromethyl sources like trifluoroiodomethane. This stability allows for easier transportation and storage, reducing lead times and ensuring a consistent supply of key starting materials for continuous production runs. The robustness of the reaction across a wide range of aliphatic olefin substrates means that a single standardized protocol can be used for multiple products, simplifying inventory management and reducing the need for specialized equipment for different campaigns.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is highly amenable to flow chemistry technologies, which offer superior light penetration and heat dissipation compared to batch reactors, facilitating seamless scale-up from grams to tons. The green profile of the reaction, characterized by the use of non-toxic catalysts and benign oxidants, simplifies waste treatment procedures and ensures compliance with increasingly stringent environmental regulations. This eco-friendly approach not only mitigates the risk of regulatory fines but also enhances the corporate sustainability profile, which is a growing requirement for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this visible light catalyzed synthesis, providing clarity for R&D teams evaluating this technology for their pipelines. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development decisions. Understanding these nuances is crucial for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the advantages of using manganese catalysts over iridium for trifluoromethylation?
A: Manganese catalysts like Mn(acac)3 are significantly more cost-effective and commercially available compared to expensive noble metal catalysts like iridium, reducing overall production costs while maintaining high efficiency under visible light conditions.
Q: Is this process suitable for aliphatic olefins?
A: Yes, unlike many traditional thermal methods limited to aromatic olefins, this visible light protocol is specifically designed for aliphatic olefins, expanding the scope for synthesizing diverse fluorinated building blocks.
Q: What safety benefits does using air as an oxidant provide?
A: Utilizing air or oxygen as the terminal oxidant eliminates the need for hazardous organic peroxides or stoichiometric oxidants, significantly enhancing operational safety and simplifying waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Trifluoromethyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light catalysis in modern organic synthesis and have integrated these advanced capabilities into our CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of beta-trifluoromethyl alcohol meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project, unlocking new possibilities for your drug discovery programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule, where we can provide specific COA data and route feasibility assessments to demonstrate how this green chemistry approach can optimize your supply chain and reduce overall manufacturing costs.
