Advanced Synthesis of Nitrogenous Heterocyclic Ferrocene Derivatives for Commercial Scale-up and High Purity Applications
Introduction to Patent CN110862421A
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for complex organometallic structures, particularly those exhibiting redox activity and lipid solubility. Patent CN110862421A discloses a groundbreaking synthetic method for nitrogenous heterocyclic ferrocene derivatives, addressing critical safety and scalability challenges inherent in traditional organometallic synthesis. This technology leverages a strategic three-step sequence involving formylation, hydroxylamination, and reduction to construct the target amino-substituted ferrocene architecture. Unlike legacy processes that rely on extreme conditions, this approach utilizes mild thermal parameters and avoids explosive reagents, marking a significant evolution in the manufacturing of redox-active pharmaceutical intermediates. The method ensures that the central iron ion maintains its reversible redox properties while facilitating smooth passage through cell membranes, a crucial attribute for bioactive applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized ferrocene derivatives has been plagued by severe safety hazards and operational complexities that hinder industrial adoption. Traditional routes frequently necessitate the use of sodium azide, a highly toxic and potentially explosive reagent that demands rigorous safety protocols and specialized waste treatment infrastructure. Furthermore, many established pathways rely on high-pressure hydrogenation reactions to reduce nitro or azide groups, requiring expensive autoclaves and posing significant risks of containment failure. These stringent equipment requirements not only inflate capital expenditure but also limit the flexibility of production facilities, making it difficult to adapt to fluctuating market demands. Additionally, the harsh conditions often associated with these legacy methods can lead to the decomposition of the sensitive ferrocene core, resulting in lower yields and complex impurity profiles that are costly to remove.
The Novel Approach
In stark contrast, the novel approach detailed in CN110862421A circumvents these pitfalls by employing a温和 (mild) and stepwise functionalization strategy that prioritizes safety and efficiency. By introducing an aldehyde group via organolithium chemistry followed by oxime formation and subsequent reduction, the process entirely eliminates the need for high-pressure hydrogenation or azide chemistry. The reaction conditions are maintained between 0°C and 40°C, allowing for precise thermal control that preserves the integrity of the heterocyclic ferrocene structure. This shift from hazardous high-energy transformations to controlled nucleophilic substitutions and reductions drastically simplifies the operational workflow. Consequently, the new method facilitates easier separation of intermediates through standard extraction techniques, thereby enhancing the overall process robustness and making it highly suitable for large-scale commercial application without compromising on safety standards.
Mechanistic Insights into Organolithium Formylation and Oxime Reduction
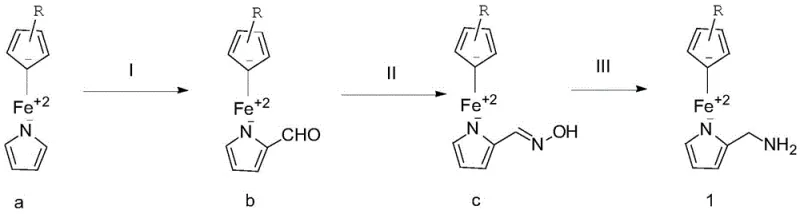
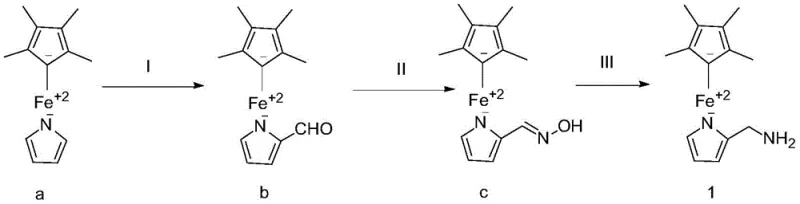
The core of this synthetic innovation lies in the precise execution of the initial formylation step, where regioselectivity and kinetic control are paramount. The process utilizes n-butyllithium to deprotonate the nitrogen-containing heterocycle, generating a reactive lithiated species that subsequently attacks dimethylformamide (DMF) to install the aldehyde functionality. This organolithium-mediated transformation is conducted at low temperatures (0-5°C during addition) to prevent over-lithiation or degradation of the ferrocene moiety. Following the formylation, the aldehyde intermediate undergoes condensation with hydroxylamine hydrochloride in the presence of a mild base such as sodium carbonate. This hydroxylamination step proceeds efficiently in ethanol-water mixtures, forming the oxime intermediate with high conversion rates. The final reduction utilizing lithium aluminum hydride cleanly converts the oxime to the primary amine, completing the installation of the nitrogenous side chain while maintaining the oxidation state of the iron center.
Impurity control is intrinsically built into the mechanistic design of this route, primarily through the moderation of reaction temperatures and the selection of compatible solvent systems. By avoiding high-temperature regimes, the method suppresses common side reactions such as polymerization of the heterocyclic ring or unwanted substitution on the cyclopentadienyl ligands. The use of specific extraction solvents like ethyl acetate and diethyl ether at distinct stages allows for the selective partitioning of the desired product from inorganic salts and unreacted starting materials. For instance, the quenching of the lithiation step with water followed by ethyl acetate extraction effectively removes lithium salts, while the final ether extraction isolates the amine product from aluminum byproducts. This meticulous attention to workup procedures ensures that the final nitrogenous heterocyclic ferrocene derivative achieves purity levels exceeding 97%, minimizing the burden on downstream purification units and ensuring a consistent quality profile for sensitive pharmaceutical applications.
How to Synthesize Nitrogenous Heterocyclic Ferrocene Derivative Efficiently
The practical implementation of this synthesis requires careful adherence to the specified stoichiometry and thermal gradients to maximize yield and safety. The process begins with the preparation of the aldehyde intermediate under inert atmosphere, followed by the aqueous-organic biphasic oxime formation, and concludes with an anhydrous reduction step. Operators must ensure strict temperature control during the n-butyllithium addition to mitigate exothermic risks, while the subsequent reduction with lithium aluminum hydride demands dry conditions to prevent premature reagent decomposition. The detailed standardized synthesis steps, including specific molar ratios and quenching protocols, are outlined below to guide process engineers in replicating this high-efficiency route.
- Step 1: Introduce an aldehyde group onto the nitrogen-containing heterocycle of the raw material using n-butyllithium and DMF at 0-40°C.
- Step 2: Perform a hydroxylamination reaction on the aldehyde intermediate using hydroxylamine hydrochloride and a base like sodium carbonate at 0-35°C.
- Step 3: Reduce the hydroxylamine group of the resulting intermediate using lithium aluminum hydride to obtain the final amine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthetic methodology offers profound strategic advantages regarding cost structure and operational continuity. By eliminating the requirement for high-pressure reactors and hazardous azide reagents, manufacturers can significantly reduce both capital investment in specialized equipment and the ongoing costs associated with safety compliance and waste disposal. The reliance on commodity chemicals such as DMF, n-butyllithium, and sodium carbonate ensures a stable supply of raw materials, insulating production schedules from the volatility often seen with exotic or regulated precursors. Furthermore, the simplified workup procedures involving standard liquid-liquid extractions reduce processing time and solvent consumption, contributing to a leaner and more cost-effective manufacturing operation that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of high-pressure hydrogenation equipment and sodium azide reagents leads to substantial savings in both CapEx and OpEx. Without the need for specialized autoclaves or extensive safety monitoring for explosive materials, facility overheads are drastically lowered. Additionally, the high purity of the intermediates reduces the need for expensive chromatographic purification, further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents like n-butyllithium and hydroxylamine hydrochloride mitigates the risk of supply disruptions common with niche catalysts. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling suppliers to respond rapidly to demand fluctuations. This reliability is critical for maintaining continuous production lines in the pharmaceutical sector, where downtime can have cascading effects on downstream drug formulation.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of heavy metal catalysts simplify the scale-up process from pilot to commercial production. Waste streams are easier to treat due to the lack of azide residues or complex metal complexes, ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces disposal costs but also enhances the sustainability profile of the supply chain, a key metric for modern corporate responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthetic route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, scalability, and product quality. Understanding these aspects is essential for technical teams evaluating the feasibility of integrating this method into existing production portfolios.
Q: Why is this synthetic method safer than conventional ferrocene derivative production?
A: Conventional methods often require hazardous reagents like sodium azide or high-pressure hydrogenation equipment. This patented method operates at mild temperatures (0-40°C) and ambient pressure using standard reagents, significantly reducing operational risks.
Q: What represents the key advantage for supply chain stability in this process?
A: The process utilizes commercially available and stable reagents such as n-butyllithium, DMF, and lithium aluminum hydride. This eliminates dependency on specialized or dangerous precursors, ensuring consistent raw material availability and reducing lead time for high-purity intermediates.
Q: How does this method impact the purity profile of the final product?
A: By maintaining mild reaction temperatures and utilizing specific extraction protocols (e.g., ethyl acetate/diethyl ether), the method minimizes side reactions and decomposition. Examples in the patent demonstrate purities exceeding 97%, simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogenous Heterocyclic Ferrocene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic methodology for the development of next-generation redox-active therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle organolithium chemistry and sensitive reductions with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee batch-to-batch consistency. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us support your journey towards commercial success with reliable, high-performance chemical solutions.
