Advanced Synthesis of High-Purity (S)-Oxiracetam via TBS Protection Strategy
Introduction to Patent CN114621128A
The pharmaceutical industry constantly seeks more efficient routes for producing nootropic agents, and the recent disclosure in patent CN114621128A represents a significant breakthrough in the manufacturing of (S)-oxiracetam. This specific enantiomer is a critical active pharmaceutical ingredient (API) known for its cognitive enhancement properties, yet its production has historically been plagued by purification challenges. The patent details a novel preparation method that fundamentally alters the downstream processing landscape by introducing a strategic protection-deprotection sequence. By shifting away from traditional, labor-intensive purification techniques, this technology offers a robust pathway to achieving exceptional chemical and optical purity. For R&D directors and supply chain managers, understanding this shift is crucial, as it directly impacts the feasibility of large-scale commercial production and the economic viability of the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-oxiracetam involved the direct condensation of S-4-chloro-3-hydroxybutyric acid ethyl ester with glycinamide, followed by arduous purification steps. The primary bottleneck in these conventional processes lies in the physicochemical properties of the target molecule itself; (S)-oxiracetam exhibits strong hydrophilicity, making it exceptionally difficult to separate from the various polar salt compounds generated during the reaction, such as glycinamide residues, sodium bicarbonate, and ammonium chloride. Because these impurities share similar polarity profiles with the product, standard crystallization techniques often fail to yield a pure solid. Consequently, manufacturers were forced to rely on silica gel column chromatography or ion exchange resin columns to isolate the product. These methods are not only operationally cumbersome and time-consuming but also require vast quantities of organic solvents, driving up both the environmental footprint and the overall production cost significantly.
The Novel Approach
The innovative methodology described in the patent circumvents these purification hurdles by temporarily modifying the molecular structure of the product. Instead of attempting to purify the highly polar (S)-oxiracetam directly, the process introduces a protective group immediately after the initial condensation reaction. Specifically, the hydroxyl group of the crude (S)-oxiracetam is shielded using a silyl protecting group, transforming the molecule into a less polar, more hydrophobic intermediate known as Intermediate I. This structural modification drastically changes the solubility profile of the compound, allowing polar salt impurities to be easily removed through simple aqueous extraction and washing. Furthermore, the protected intermediate possesses superior crystallinity, enabling high-purity isolation through straightforward recrystallization rather than complex chromatography. This strategic detour simplifies the entire workflow, reducing solvent consumption and operational complexity while simultaneously enhancing the final yield.
Mechanistic Insights into TBS-Protection and Crystallization
The core of this technological advancement lies in the precise application of protective group chemistry to manipulate physical properties. The reaction begins with the condensation of the chiral starting material, S-4-chloro-3-hydroxybutyric acid ethyl ester, with glycinamide under alkaline conditions to form the crude lactam ring. Once formed, the crude mixture is treated with tert-butyldimethylchlorosilane (TBSCl) in the presence of an acid-binding agent such as triethylamine or imidazole. This step selectively silylates the hydroxyl group at the 4-position of the pyrrolidine ring. The introduction of the bulky tert-butyldimethylsilyl (TBS) group effectively masks the hydrogen-bonding capability of the hydroxyl moiety, which is the primary driver of the molecule's hydrophilicity. By converting this polar group into a lipophilic silyl ether, the intermediate becomes soluble in organic solvents like ethyl acetate or toluene, facilitating the phase separation of water-soluble inorganic salts.
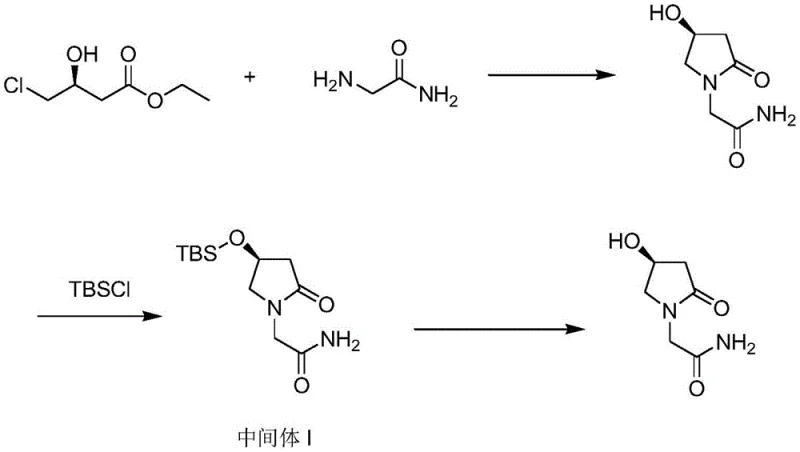
Following the protection step, the process leverages the enhanced crystallinity of Intermediate I for purification. The patent highlights that the TBS-protected intermediate can be recrystallized from solvents such as isopropyl ether or n-hexane to achieve a purity of over 99.8% by HPLC. This is a critical control point, as the crystallization lattice effectively excludes remaining organic impurities. The final step involves the removal of the protecting group using an acid catalyst, such as hydrochloric acid or methanesulfonic acid, in a solvent like dioxane or ethanol. This deprotection restores the free hydroxyl group, yielding the final (S)-oxiracetam. The beauty of this mechanism is that the final recrystallization of the deprotected product is now feasible and efficient, resulting in a final API with both chemical and optical purity exceeding 99.9%, a benchmark that is difficult to reach with traditional ion-exchange methods.
How to Synthesize (S)-Oxiracetam Efficiently
Implementing this synthesis route requires careful control of reaction parameters to maximize the benefits of the protection strategy. The process is designed to be scalable, moving from the initial alkaline condensation to the final acidic deprotection with minimal unit operations. Operators must ensure that the molar ratio of the protecting reagent to the substrate is optimized, typically between 1:1 and 2.5, to ensure complete conversion without excessive reagent waste. The temperature during the protection phase is maintained between 0°C and 60°C to balance reaction rate and selectivity. Once the high-purity Intermediate I is secured via crystallization, the deprotection step is conducted under reflux conditions to drive the equilibrium towards the final product. For a detailed, step-by-step breakdown of the exact reagents, solvent volumes, and workup procedures described in the patent examples, please refer to the standardized synthesis guide below.
- Condense S-4-chloro-3-hydroxybutyric acid ethyl ester with glycinamide under alkaline conditions to form crude (S)-oxiracetam.
- React the crude solution with tert-butyldimethylchlorosilane (TBSCl) to form the protected Intermediate I.
- Crystallize Intermediate I in a solvent like isopropyl ether to remove polar salt impurities.
- Treat the high-purity Intermediate I with an acid catalyst to remove the protecting group and obtain final (S)-oxiracetam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this TBS-protection methodology offers profound economic and logistical benefits. The most immediate impact is the elimination of column chromatography, which is notoriously expensive due to the high cost of silica gel, the large volume of solvents required for elution, and the significant labor hours needed for operation. By replacing this with a crystallization-based purification, the process drastically reduces the consumption of organic solvents and hazardous waste generation. This simplification translates directly into lower operating expenditures (OPEX) and a reduced environmental compliance burden. Furthermore, the reliance on commodity chemicals like TBSCl and common solvents ensures a stable and reliable supply chain, mitigating the risks associated with specialized resin availability.
- Cost Reduction in Manufacturing: The patent explicitly notes that compared to traditional ion exchange resin schemes, this new method can reduce production costs significantly, with data suggesting a reduction range of 30-50%. This is primarily driven by the removal of the chromatography step, which eliminates the need for expensive stationary phases and the energy-intensive concentration of large solvent volumes. Additionally, the yield of qualified product is reported to improve by 30-50%, meaning more saleable API is generated from the same amount of raw materials, further driving down the cost per kilogram.
- Enhanced Supply Chain Reliability: The simplified process flow enhances supply continuity by reducing the number of potential failure points. Traditional chromatography is prone to column channeling, resin degradation, and batch-to-batch variability, which can lead to production delays. In contrast, crystallization is a robust, well-understood unit operation that is easier to control and scale. The use of widely available reagents like TBSCl and standard acids ensures that raw material sourcing remains stable, preventing bottlenecks that could disrupt the delivery of this critical nootropic intermediate to downstream formulation partners.
- Scalability and Environmental Compliance: From a sustainability perspective, this route is far superior for commercial scale-up. The reduction in solvent usage directly lowers the facility's VOC (Volatile Organic Compound) emissions and waste disposal costs. The ability to purify the intermediate via crystallization rather than chromatography makes the process inherently more scalable, as crystallization tanks can be easily enlarged, whereas scaling chromatography columns presents significant engineering challenges. This aligns perfectly with modern green chemistry principles, making the manufacturing process more attractive to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures within patent CN114621128A, providing clarity on the feasibility and advantages of the TBS-protection strategy for (S)-oxiracetam production.
Q: Why is column chromatography avoided in this new (S)-oxiracetam process?
A: Traditional methods require column chromatography because (S)-oxiracetam is highly hydrophilic and difficult to separate from salt impurities. This new method uses a TBS protecting group to temporarily reduce polarity, allowing impurities to be washed away and the intermediate to be purified via simple crystallization.
Q: What is the achievable purity of (S)-oxiracetam using this method?
A: According to patent CN114621128A, the final product achieves a chemical purity of over 99.9% and an optical purity (ee value) of over 99.9% after recrystallization.
Q: What protecting reagent is used in this synthesis?
A: The process utilizes tert-butyldimethylchlorosilane (TBSCl) as the protective reagent. It is chosen for its low cost, high conversion rate (>95%), and the excellent crystallinity it imparts to the intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN114621128A for the global nootropics market. As a leading CDMO partner, we possess the technical expertise to adapt and optimize such advanced protective group strategies for industrial-scale manufacturing. Our facilities are equipped to handle complex synthetic pathways, ensuring that we can deliver high-purity intermediates and APIs with consistent quality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing stringent purity specifications and rigorous QC labs to guarantee that every batch meets the >99.9% purity benchmarks required for pharmaceutical applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and high-yield technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply of (S)-oxiracetam is both economically optimized and technically secure for the long term.
