Revolutionizing Octanoyl Hydroxamic Acid Production via Heterogeneous Catalysis for Global Supply Chains
Revolutionizing Octanoyl Hydroxamic Acid Production via Heterogeneous Catalysis for Global Supply Chains
The global demand for high-purity preservatives and tyrosinase inhibitors in the cosmetic and pharmaceutical sectors has necessitated a shift towards greener, more efficient synthetic routes. Patent CN115197097A introduces a groundbreaking methodology for the preparation of safe antibacterial Octanoyl Hydroxamic Acid, addressing critical bottlenecks in traditional manufacturing. This technology replaces hazardous homogeneous catalysts with robust heterogeneous systems, fundamentally altering the economic and environmental landscape of producing this vital intermediate. By leveraging solid acid and solid base catalysts, the process not only enhances reaction kinetics but also ensures exceptional product consistency, making it an ideal candidate for large-scale industrial adoption by forward-thinking chemical enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Octanoyl Hydroxamic Acid has relied heavily on liquid strong acids like concentrated sulfuric acid for esterification and subsequent neutralization with strong bases. This conventional approach presents severe operational challenges, including significant equipment corrosion which leads to frequent maintenance shutdowns and increased capital expenditure for specialized alloy reactors. Furthermore, the use of homogeneous catalysts complicates downstream processing, as extensive washing and neutralization steps are required to remove residual acidic or basic species, generating substantial volumes of saline wastewater. These inefficiencies not only inflate production costs but also pose significant environmental compliance risks, making traditional routes increasingly untenable for modern sustainable manufacturing facilities aiming for zero-liquid discharge goals.
The Novel Approach
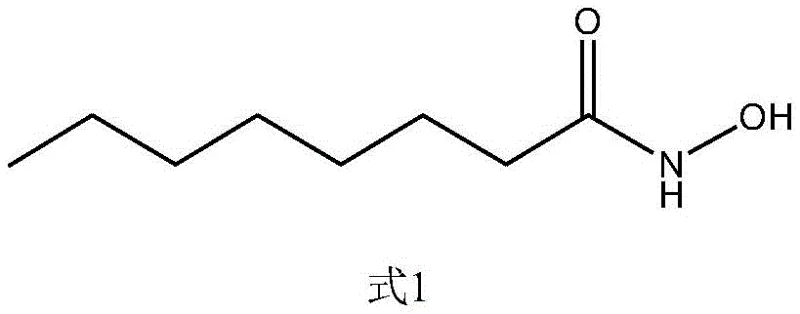
The innovative pathway disclosed in the patent circumvents these issues by employing a dual solid-catalyst system that streamlines the entire production workflow. In the first stage, n-octanoic acid undergoes esterification with low-carbon alcohols using a sulfonic acid-loaded solid phase catalyst, eliminating the need for corrosive liquid acids. This is followed by a hydroxamation reaction utilizing a two-dimensional layered double metal hydroxide as a solid base catalyst. This heterogenized approach allows for simple filtration to recover and reuse catalysts, drastically reducing waste generation. The transition from liquid to solid catalysis represents a paradigm shift, offering a cleaner, safer, and more economically viable route that aligns perfectly with the rigorous quality and sustainability standards demanded by top-tier reliable cosmetic preservative supplier networks globally.
Mechanistic Insights into Solid Acid and Base Catalyzed Synthesis
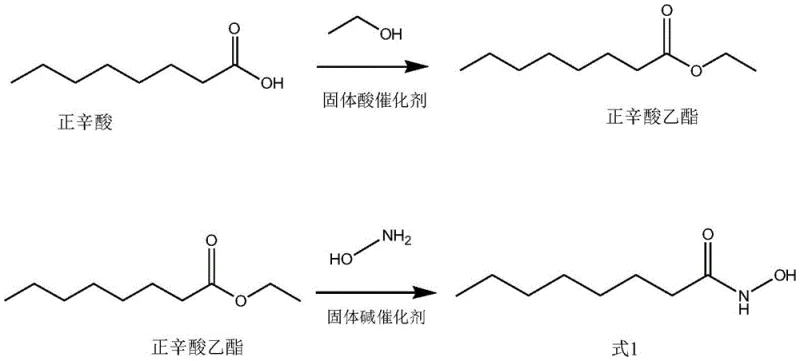
The core of this technological advancement lies in the precise engineering of the catalytic active sites. The solid acid catalyst, specifically a selenoviral acid solid phase loaded with sulfonic groups (such as model SC18143), provides strong Brønsted acidity necessary for driving the esterification equilibrium forward without leaching into the reaction medium. This structural stability ensures that the catalyst maintains high activity over repeated cycles, preventing the formation of side products often associated with harsh liquid acid conditions. Simultaneously, the use of a layered double metal hydroxide (LDH), such as MgAl-LDH, in the second step offers tunable basicity that facilitates the nucleophilic attack of hydroxylamine on the ester carbonyl. The layered structure of the LDH catalyst provides a high surface area and accessible active sites, promoting efficient conversion while maintaining the integrity of the sensitive hydroxamic acid functionality against degradation.
Impurity control is inherently built into this mechanistic design through the physical separation capabilities of heterogeneous catalysis. Unlike homogeneous systems where catalyst residues are difficult to separate, the solid catalysts in this process can be removed via simple filtration, significantly reducing the burden on purification units. Furthermore, the patent highlights the critical importance of stoichiometric control, specifically maintaining the molar ratio of n-octanoate ester to hydroxylamine between 1:1.01 and 1:1.02. This narrow window ensures complete consumption of the ester while minimizing excess hydroxylamine, which could otherwise lead to complex impurity profiles requiring costly recrystallization steps. The result is a process capable of delivering high-purity Octanoyl Hydroxamic Acid with purity levels consistently exceeding 99.8%, satisfying the most stringent specifications for pharmaceutical and personal care applications.
How to Synthesize Octanoyl Hydroxamic Acid Efficiently
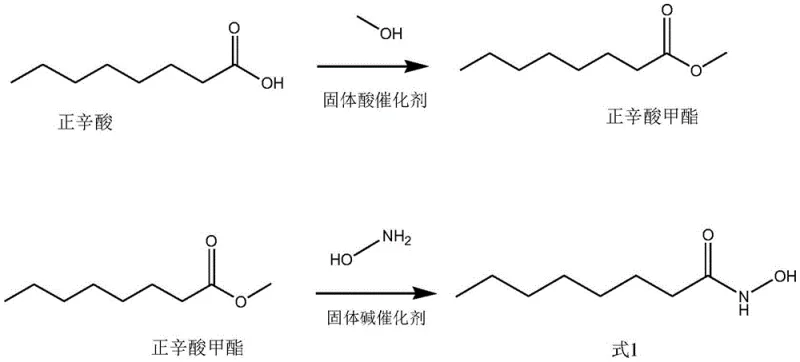
Implementing this synthesis route requires careful attention to catalyst loading ratios and solvent systems to maximize the benefits of the heterogeneous design. The process begins with the reflux of n-octanoic acid and ethanol in the presence of the solid acid catalyst, followed by a distinct hydroxamation step in an ethanol-water mixture using the solid base catalyst. The synergy between the specific catalyst types and the controlled reaction parameters allows for a streamlined operation that minimizes unit operations. For detailed operational parameters, temperature controls, and workup procedures that ensure optimal yield and catalyst longevity, please refer to the standardized synthesis guide below which outlines the critical process controls derived from the patent examples.
- Step 1: Esterification of n-Octanoic Acid with low-carbon alcohol using a sulfonic acid-loaded solid catalyst at reflux.
- Step 2: Reaction of the resulting ester with hydroxylamine in an ethanol-water solvent system using a layered double metal hydroxide catalyst.
- Step 3: Acidification with citric acid solution, followed by filtration, distillation, and crystallization to isolate high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this solid-catalyst technology translates directly into enhanced operational resilience and cost predictability. By eliminating the reliance on corrosive liquid acids and bases, manufacturers can significantly extend the lifecycle of their reactor vessels and piping infrastructure, thereby deferring major capital replacement costs. The ability to recycle catalysts multiple times without significant loss of activity reduces the recurring expense of raw material procurement for catalysts, creating a more stable cost structure that is less susceptible to market volatility. This stability is crucial for long-term supply contracts where price consistency is a key differentiator in the competitive landscape of cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of neutralization steps and the reduction in wastewater treatment requirements lead to substantial savings in utility and disposal costs. Since the catalysts are heterogeneous, they do not contaminate the product stream, reducing the need for expensive purification solvents and energy-intensive distillation processes. This leaner manufacturing footprint allows for a more competitive pricing model while maintaining healthy margins, providing a distinct advantage in tender negotiations for bulk chemical supplies.
- Enhanced Supply Chain Reliability: The robustness of the solid catalysts ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by off-spec batches. The simplified workup procedure, involving mere filtration rather than complex extraction and washing sequences, shortens the overall cycle time per batch. This efficiency gain enhances throughput capacity, allowing suppliers to respond more agilely to fluctuating market demands and ensuring uninterrupted supply continuity for downstream formulators who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as atom economy and waste prevention, facilitate easier regulatory approval and compliance with increasingly strict environmental regulations. The reduced generation of saline effluent simplifies the permitting process for plant expansions, enabling seamless commercial scale-up of complex cosmetic intermediates. This environmental stewardship not only mitigates regulatory risk but also aligns with the corporate sustainability goals of major multinational clients, strengthening business partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms legacy processes in terms of efficiency and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement officers assessing the long-term value proposition of suppliers utilizing this technology.
Q: How does the solid catalyst method improve equipment longevity compared to traditional sulfuric acid processes?
A: By replacing corrosive liquid sulfuric acid and sodium hydroxide with recyclable solid acid and base catalysts, the process significantly reduces equipment corrosion and maintenance downtime, ensuring longer reactor lifespans.
Q: What is the typical purity achievable with this novel heterogeneous catalysis route?
A: The patented method consistently achieves product purity levels exceeding 99.8%, with yields maintained above 94% even after multiple catalyst recycling cycles, meeting stringent pharmaceutical standards.
Q: Why is the molar ratio of ester to hydroxylamine critical in this synthesis?
A: Controlling the molar ratio between 1:1.01 and 1:1.02 is essential to maximize conversion efficiency while minimizing unreacted starting materials, thereby optimizing both yield and final product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octanoyl Hydroxamic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this solid-catalyst technology in delivering superior value to the global marketplace. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Octanoyl Hydroxamic Acid meets the exacting standards required for sensitive cosmetic and pharmaceutical formulations.
We invite industry leaders to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your partnership with us drives both innovation and profitability in your respective markets.
