Scalable Visible Light Catalysis for High-Purity β-Trifluoromethyl Alcohol Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to introduce trifluoromethyl groups into organic molecules, a modification known to significantly enhance metabolic stability and lipophilicity. Patent CN111205185A presents a groundbreaking preparation method for β-trifluoromethyl alcohol that leverages visible light catalysis to overcome the limitations of traditional thermal reactions. This innovative approach utilizes aliphatic alkenes and sodium trifluoromethanesulfonate as substrates, employing an organic manganese salt as a photocatalyst under mild room temperature conditions. By shifting from energy-intensive thermal processes to photon-driven chemistry, this technology aligns perfectly with the global trend towards green manufacturing and cost-effective intermediate production. The method not only broadens the substrate scope to include complex aliphatic chains but also ensures high selectivity and yield without the need for hazardous oxidants. For R&D directors and procurement specialists, this patent represents a viable route to high-purity pharmaceutical intermediates with a substantially reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β-trifluoromethyl alcohol has relied heavily on thermal reactions that demand harsh conditions and expensive reagents, creating significant bottlenecks in commercial production. Traditional methods often utilize noble metal catalysts such as iridium, which are not only cost-prohibitive for large-scale manufacturing but also pose challenges in terms of residual metal removal from the final API. Furthermore, many existing protocols require dangerous oxidants like t-butyl hydroperoxide in large excess, introducing severe safety risks and complicating waste disposal procedures. The substrate scope in these conventional thermal methods is frequently limited to aromatic olefins, failing to accommodate the diverse aliphatic structures required for modern drug discovery. Additionally, the poor selectivity of older methods often results in mixtures of alcohols and ketones, necessitating complex and yield-reducing purification steps. These cumulative inefficiencies drive up the cost of goods sold and extend lead times, making it difficult for supply chain managers to maintain consistent inventory levels.
The Novel Approach
In stark contrast, the visible light catalyzed method described in the patent offers a transformative solution by utilizing abundant and inexpensive organic manganese salts as photocatalysts. This novel approach operates at room temperature (23-25°C) using simple blue LED light sources, eliminating the need for energy-intensive heating systems and specialized high-pressure equipment. The reaction employs air or oxygen as a green oxidant, replacing hazardous peroxides and significantly enhancing the safety profile of the manufacturing process. Crucially, this method demonstrates excellent compatibility with aliphatic olefins, including those with long carbon chains, which were previously difficult to functionalize with high selectivity. The use of sodium trifluoromethanesulfonate as a stable and safe trifluoromethylating agent further simplifies the handling and storage requirements for production facilities. By achieving high yields through a radical addition mechanism under mild conditions, this technology provides a robust platform for the commercial scale-up of complex pharmaceutical intermediates.

Mechanistic Insights into Visible Light Photocatalysis
The core of this technological advancement lies in the efficient generation of trifluoromethyl radicals through the excitation of the manganese catalyst by visible light photons. Upon irradiation with blue light, the organic manganese salt enters an excited state that facilitates the single-electron transfer necessary to activate the sodium trifluoromethanesulfonate. This process generates the reactive trifluoromethyl radical species which then undergoes regioselective addition to the carbon-carbon double bond of the aliphatic alkene substrate. The resulting carbon-centered radical is subsequently trapped by molecular oxygen from the air, leading to the formation of the hydroperoxide intermediate that is reduced to the final alcohol product. This catalytic cycle is highly efficient because the manganese catalyst is regenerated at the end of the reaction, allowing for low catalyst loading while maintaining high turnover numbers. Understanding this mechanism is critical for R&D teams aiming to optimize reaction parameters for specific substrate derivatives.
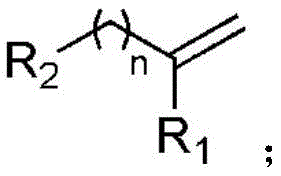
Controlling the impurity profile in this reaction is achieved through the precise modulation of the radical addition step and the exclusion of competing thermal pathways. Since the reaction proceeds at room temperature, there is minimal thermal degradation of sensitive functional groups often present in complex pharmaceutical intermediates. The selectivity for the β-trifluoromethyl alcohol over the corresponding ketone is enhanced by the specific redox potential of the manganese catalyst system, which favors the desired radical recombination pathway. Furthermore, the use of a mixed solvent system comprising 1,4-dioxane and acetone helps to stabilize the radical intermediates and ensures homogeneous reaction conditions. This level of control over the reaction trajectory means that downstream purification is simplified, resulting in a final product with high chemical purity and minimal byproduct formation. For quality control teams, this translates to more consistent batch-to-batch reproducibility and easier compliance with stringent regulatory standards.
How to Synthesize β-Trifluoromethyl Alcohol Efficiently
To implement this synthesis route effectively, manufacturers must adhere to specific operational parameters regarding light intensity and reagent stoichiometry as outlined in the patent examples. The process begins with the preparation of a reaction mixture containing the aliphatic olefin and sodium trifluoromethanesulfonate in a defined ratio, typically using a four-fold molar excess of the sulfonate to drive the reaction to completion. The detailed standardized synthesis steps, including specific solvent volumes and workup procedures, are provided in the technical guide below to ensure reproducibility. Operators should ensure that the reaction vessel is transparent to blue light and that the LED source provides uniform irradiation across the reaction mixture to maximize photon efficiency. Following the reaction period, standard extraction and column chromatography techniques are employed to isolate the target molecule with high purity. Adhering to these protocols allows for the safe and efficient production of valuable fluorinated intermediates.
- Prepare the reaction mixture by combining aliphatic olefin substrates with sodium trifluoromethanesulfonate in an organic solvent system such as 1,4-dioxane and acetone.
- Add the organic manganese salt catalyst, specifically manganese acetylacetonate (Mn(acac)3), to the reaction vessel under an oxygen-containing atmosphere like air.
- Irradiate the mixture with blue light LED (40W) at room temperature for 24 hours, followed by purification via column chromatography to isolate the target alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this visible light catalyzed process offers substantial advantages in terms of cost reduction and supply chain reliability for global chemical buyers. The replacement of expensive noble metal catalysts with commercially available manganese salts drastically lowers the raw material costs associated with the synthesis. Moreover, the elimination of hazardous oxidants reduces the need for specialized safety infrastructure and lowers the costs related to waste treatment and environmental compliance. The ability to run the reaction at room temperature significantly reduces energy consumption compared to thermal processes, contributing to lower operational expenditures over the lifecycle of the product. These factors combined create a more resilient supply chain that is less vulnerable to fluctuations in the price of precious metals or energy sources. Procurement managers can leverage this technology to secure more stable pricing agreements and reduce the total cost of ownership for critical intermediates.
- Cost Reduction in Manufacturing: The substitution of iridium catalysts with manganese salts represents a significant decrease in catalyst procurement costs, while the use of air as an oxidant eliminates the need for purchasing expensive chemical oxidants. Additionally, the mild reaction conditions reduce energy bills associated with heating and cooling, leading to substantial overall cost savings in the manufacturing process. The simplified workup procedure also reduces labor and solvent costs during the purification stage.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like aliphatic olefins and sodium trifluoromethanesulfonate, the risk of supply disruptions is minimized compared to methods relying on specialized or scarce reagents. The robustness of the reaction conditions ensures consistent production output, allowing supply chain heads to plan inventory with greater confidence. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing.
- Scalability and Environmental Compliance: The green nature of this process, characterized by the use of visible light and air, aligns with increasingly strict environmental regulations, reducing the risk of compliance-related shutdowns. The absence of toxic heavy metals and dangerous peroxides simplifies the scale-up process from pilot plant to commercial production volumes. This ease of scale-up ensures that the technology can meet growing market demand without significant re-engineering of the production line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new route for their specific product portfolios. The information covers catalyst loading, reaction times, and substrate compatibility to provide a comprehensive overview.
Q: What are the primary advantages of using visible light catalysis over traditional thermal methods for this synthesis?
A: Visible light catalysis eliminates the need for high-temperature thermal conditions, significantly reducing energy consumption and operational risks. Furthermore, it allows for the use of cheaper manganese catalysts instead of expensive noble metals like iridium, while utilizing air as a green oxidant.
Q: Is this method suitable for large-scale aliphatic olefin substrates?
A: Yes, the patent specifically highlights the method's applicability to aliphatic olefins with C9-C15 chains, overcoming the limitations of previous methods that were restricted mostly to aromatic olefins or required dangerous oxidants.
Q: How does the choice of oxidant impact the safety profile of the manufacturing process?
A: By using an oxygen-containing atmosphere such as air instead of dangerous organic peroxides like t-butyl hydroperoxide, the process drastically improves safety standards and simplifies waste treatment protocols for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable β-Trifluoromethyl Alcohol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies like visible light catalysis to deliver high-quality chemical intermediates to the global market. Our CDMO expertise allows us to translate complex patent methodologies into robust commercial processes, ensuring that clients receive materials that meet the highest standards of purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing stringent purity specifications and rigorous QC labs to guarantee product integrity. Our commitment to innovation means we can offer this cutting-edge β-trifluoromethyl alcohol synthesis as a viable option for your next project, combining technical excellence with commercial viability.
We invite you to discuss how this technology can optimize your supply chain and reduce your overall manufacturing costs through a Customized Cost-Saving Analysis. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your unique requirements. By partnering with us, you gain access to a reliable source of advanced intermediates that can accelerate your drug development timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
