Advanced Iodide-Catalyzed Synthesis of N-Hydroxy Esters for Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing N-hydroxy ester motifs, which serve as critical structural units in bioactive natural products and drug molecules. Patent CN102617441B introduces a transformative approach to this challenge by utilizing aldehyde derivatives and hydroxylamine derivatives as reaction substrates. This innovative protocol employs iodide species as catalysts and tert-butyl hydrogen peroxide (TBHP) as an oxidant to drive a double free radical cross-coupling reaction. Unlike traditional pathways that often rely on harsh conditions or unstable intermediates, this method operates under remarkably mild parameters, typically between 60°C and 100°C. The strategic selection of readily available iodide salts, such as tetrabutylammonium iodide, ensures that the process remains economically viable while maintaining high atom economy. This technological breakthrough addresses long-standing inefficiencies in the synthesis of high-purity pharmaceutical intermediates, offering a streamlined route that aligns with modern green chemistry principles and industrial scalability requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of N-hydroxy esters has been plagued by significant operational and economic hurdles that hinder efficient commercial manufacturing. Conventional strategies frequently depend on the coupling of carboxylic acids or their derivatives with hydroxylamine, processes that often necessitate the use of expensive and toxic transition metal catalysts such as ruthenium, rhodium, or palladium. Furthermore, alternative routes involving acid chlorides pose severe safety risks due to their susceptibility to hydrolysis and the generation of corrosive byproducts. Some methods require the use of carbon monoxide, a hazardous gas that demands specialized equipment and rigorous safety protocols, thereby inflating capital expenditure. Additionally, these traditional approaches often suffer from narrow substrate scopes, failing to accommodate diverse functional groups without extensive protection and deprotection sequences. The cumulative effect of these limitations is a complex, costly, and environmentally burdensome workflow that struggles to meet the demands of modern supply chains for reliable agrochemical intermediate and pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast, the novel iodide-catalyzed methodology presented in the patent data offers a paradigm shift towards simplicity and efficiency. By leveraging the unique redox properties of iodide ions in conjunction with TBHP, this system generates reactive radical species in situ without the need for precious metals. The reaction proceeds smoothly in common organic solvents like 1,2-dichloroethane or toluene, utilizing molecular oxygen from the air as a benign component of the oxidation cycle. This eliminates the dependency on sensitive anhydrous conditions or inert atmospheres, significantly lowering the barrier to entry for scale-up. The use of aldehyde derivatives as starting materials is particularly advantageous, as aldehydes are widely available and often cheaper than their carboxylic acid counterparts. This approach not only simplifies the synthetic sequence but also enhances the overall safety profile of the manufacturing process, making it an ideal candidate for cost reduction in electronic chemical manufacturing and other high-value sectors where purity and safety are paramount.
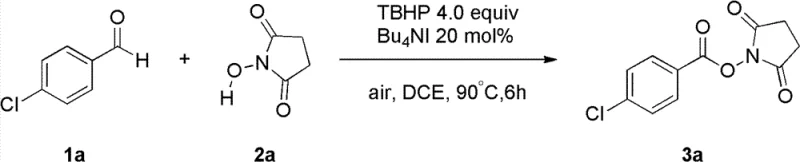
Mechanistically, the success of this transformation hinges on the efficient generation and propagation of free radical species mediated by the iodide catalyst. The reaction initiates with the oxidation of the iodide anion by TBHP to form an active iodine species, which subsequently abstracts a hydrogen atom from the aldehyde substrate. This step generates an acyl radical that is poised for cross-coupling with the nitrogen-centered radical derived from the hydroxylamine derivative. The double free radical cross-coupling mechanism ensures high selectivity for the desired N-O bond formation while minimizing side reactions such as over-oxidation to carboxylic acids. The catalytic cycle is regenerated through the interaction of the iodine species with the oxidant, sustaining the reaction without the accumulation of stoichiometric waste. This precise control over radical intermediates is crucial for maintaining high purity specifications, as it prevents the formation of complex impurity profiles that are difficult to separate during downstream processing.
Impurity control is a critical consideration for R&D directors evaluating the feasibility of this route for GMP manufacturing. The mild reaction conditions inherently suppress thermal degradation pathways that often lead to polymeric byproducts or decomposition of sensitive functional groups. Moreover, the absence of heavy metal residues eliminates the need for costly and time-consuming scavenging steps, which are mandatory when using palladium or copper catalysts. The patent data indicates that the reaction tolerates a wide array of substituents on the aromatic ring of the aldehyde, including electron-withdrawing groups like halogens and nitro groups, as well as electron-donating groups like methoxy and alkyl chains. This broad functional group compatibility ensures that the impurity spectrum remains predictable and manageable across different substrate analogs. Consequently, the resulting N-hydroxy esters can be isolated with high purity through straightforward work-up procedures, facilitating their direct use in subsequent synthetic steps without extensive purification.
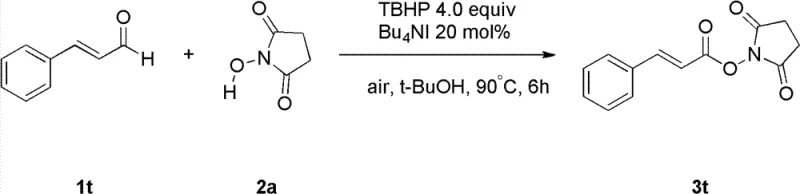
How to Synthesize N-Hydroxy Ester Efficiently
To implement this synthesis effectively, operators must adhere to specific procedural guidelines that maximize yield and reproducibility. The process begins with the careful charging of the reactor with the iodide catalyst, typically at a loading of 10 to 30 mol%, alongside the aldehyde and hydroxylamine substrates. The choice of oxidant equivalents is critical, with 4 to 5 equivalents of TBHP generally providing optimal conversion rates. The reaction mixture is then heated to a temperature range of 60°C to 100°C, with 90°C identified as the preferred setpoint for balancing reaction rate and selectivity. Monitoring the reaction progress is essential to determine the precise endpoint, usually achieved within 4 to 8 hours depending on the specific substrate electronics. Upon completion, the reaction is quenched with saturated sodium sulfite to neutralize excess oxidant, followed by extraction with ethyl acetate or dichloromethane. The detailed standardized synthesis steps see the guide below.
- Charge the reaction vessel with iodide catalyst (e.g., Bu4NI), aldehyde derivative, hydroxylamine derivative, and oxidant (TBHP) in a suitable solvent like 1,2-dichloroethane.
- Heat the reaction mixture in air at temperatures between 60°C to 100°C for a duration of 4 to 8 hours to facilitate the double free radical cross-coupling.
- Quench the reaction with saturated sodium sulfite, extract with ethyl acetate, and purify the crude product via simple column chromatography to obtain the final N-hydroxy ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodide-catalyzed technology presents compelling opportunities for optimizing operational expenditures and securing supply continuity. The primary driver of value lies in the substantial cost savings achieved by replacing expensive noble metal catalysts with inexpensive and abundant iodide salts. This shift drastically reduces the raw material costs associated with catalysis, which is a significant portion of the overall manufacturing budget for complex intermediates. Furthermore, the simplified post-treatment process reduces the consumption of solvents and adsorbents required for metal scavenging, leading to additional savings in waste disposal and material handling. The robustness of the reaction conditions also minimizes the risk of batch failures due to sensitivity to moisture or oxygen, thereby enhancing overall production reliability and reducing the cost of quality control measures.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium, rhodium, or ruthenium removes a major cost center from the production budget. Iodide salts are commodity chemicals with stable pricing and widespread availability, ensuring that the cost of goods sold remains low even during periods of market volatility. Additionally, the avoidance of specialized reagents like acid chlorides or carbon monoxide further reduces the financial burden associated with hazardous material storage and handling. The overall process efficiency translates into a leaner manufacturing model where resources are allocated more effectively, driving down the unit cost of the final N-hydroxy ester product significantly.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as all key reagents, including the aldehyde substrates, hydroxylamine derivatives, and iodide catalysts, are commercially available off-the-shelf products. This reduces dependency on single-source suppliers for exotic catalysts that may face supply disruptions. The mild reaction conditions also allow for flexibility in manufacturing locations, as the process does not require specialized high-pressure or cryogenic equipment. This decentralization potential strengthens the supply chain against geopolitical or logistical shocks, ensuring consistent delivery schedules for downstream customers who rely on timely access to high-purity pharmaceutical intermediates for their own production lines.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than traditional methods, aligning with increasingly stringent global regulations on industrial emissions and waste. The absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with metal discharge limits. Scalability is facilitated by the homogeneous nature of the reaction and the use of standard heating and stirring equipment, allowing for seamless transfer from laboratory bench scale to multi-ton commercial production. This ease of scale-up ensures that supply can be rapidly ramped up to meet surging demand without the need for prolonged process re-engineering or capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodide-catalyzed synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these aspects is vital for stakeholders evaluating the integration of this technology into existing manufacturing portfolios. The answers reflect a commitment to transparency and technical accuracy, ensuring that decision-makers have the necessary information to assess feasibility.
Q: What are the primary advantages of using iodide catalysts over traditional metal catalysts for N-hydroxy ester synthesis?
A: Iodide catalysts offer significant cost reductions by eliminating the need for expensive transition metals like palladium or rhodium. They also operate under milder conditions, reducing energy consumption and simplifying safety protocols regarding toxic metal residue removal.
Q: How does the substrate scope of this iodide-catalyzed method compare to conventional carboxylic acid coupling?
A: This method utilizes aldehyde derivatives directly, bypassing the need for pre-activated carboxylic acids or acid chlorides. It demonstrates high functional group compatibility, tolerating halogens, ethers, and heterocycles, which broadens the range of accessible pharmaceutical intermediates.
Q: Is the post-treatment process for this synthesis suitable for large-scale manufacturing?
A: Yes, the work-up involves standard quenching with sodium sulfite and extraction, followed by simple column chromatography. The absence of heavy metal contaminants streamlines the purification process, making it highly scalable for industrial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Hydroxy Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic routes like the iodide-catalyzed preparation of N-hydroxy esters to maintain competitive advantage. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence allows us to navigate the complexities of radical chemistry and deliver high-quality intermediates consistently.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to reach out to request specific COA data and route feasibility assessments that demonstrate how our capabilities can optimize your supply chain. By partnering with us, you gain access to a reliable source of complex intermediates backed by deep chemical expertise and a dedication to sustainable manufacturing practices.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
