Advanced Dynamic Kinetic Resolution for Commercial Scale-up of 2,6-Dimethyl-L-Tyrosine
The pharmaceutical industry's relentless pursuit of novel peptide therapeutics has placed unprecedented demand on the supply of specialized non-natural amino acids, particularly sterically hindered variants like 2,6-dimethyl-L-tyrosine. This specific building block is increasingly critical for stabilizing peptide structures and enhancing metabolic resistance in next-generation drug candidates. However, traditional synthetic routes have long been plagued by economic and technical inefficiencies, primarily relying on costly asymmetric hydrogenation protocols. A significant technological breakthrough is detailed in patent CN110467539B, which discloses a robust dynamic kinetic resolution method that fundamentally alters the production landscape for this valuable intermediate. By shifting away from transition metal catalysis toward an organic acid-mediated crystallization process, this innovation offers a pathway to substantially lower manufacturing costs while simplifying purification workflows. The core molecular architecture targeted by this process is illustrated below, highlighting the specific ester derivatives that serve as the pivotal intermediates in this streamlined synthesis.
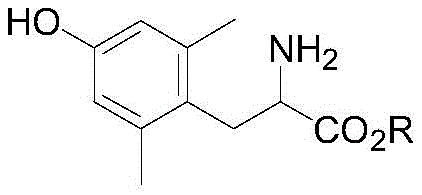
For R&D directors evaluating process viability, the structural integrity and purity profile of the final amino acid are paramount. The method described ensures that the steric bulk introduced by the ortho-methyl groups does not impede the formation of high-purity crystals, a common challenge in hindered amino acid synthesis. This patent provides a comprehensive framework for accessing the L-enantiomer with exceptional optical purity, bypassing the stringent metal clearance requirements that often bottleneck peptide API production. As we delve deeper into the technical specifics, it becomes evident that this approach represents a paradigm shift from catalytic dependency to thermodynamic control via crystallization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 2,6-dimethyl-tyrosine has been dominated by asymmetric hydrogenation strategies utilizing precious metal complexes. Literature references such as those by Dygos and Praquin describe routes employing rhodium or ruthenium catalysts paired with sophisticated chiral ligands to induce stereoselectivity. While chemically elegant, these methods present severe drawbacks for large-scale industrial application. The primary concern is the exorbitant cost associated with noble metals and the proprietary ligands required to achieve high enantioselectivity. Furthermore, the presence of heavy metals in the final product necessitates rigorous and expensive downstream purification steps to meet regulatory limits for pharmaceutical ingredients. The sensitivity of these catalytic systems to oxygen and moisture also imposes strict operational constraints, requiring specialized equipment and inert atmospheres that drive up capital expenditure. Additionally, the recovery and recycling of these expensive catalysts are often inefficient, leading to significant material loss and increased waste generation, which contradicts modern green chemistry principles.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110467539B introduces a dynamic kinetic resolution strategy that elegantly circumvents the need for transition metals. This novel approach leverages the differential solubility of diastereomeric salts formed between the racemic amino acid ester and a chiral resolving agent, specifically L-dibenzoyl tartaric acid. What sets this method apart is the integration of an in-situ racemization system within the crystallization vessel. By adding a racemizing agent such as salicylaldehyde in the presence of a mineral acid, the unwanted D-enantiomer remaining in the mother liquor is continuously converted back into a racemic mixture. This dynamic equilibrium allows the desired L-enantiomer to be continuously harvested as a precipitating salt, effectively breaking the theoretical 50% yield ceiling inherent to classical resolution techniques. The use of commodity chemicals like isopropanol, sulfuric acid, and salicylaldehyde ensures that the process is not only economically viable but also operationally simple, requiring standard glass-lined reactors rather than high-pressure hydrogenation vessels.
Mechanistic Insights into Dynamic Kinetic Chiral Resolution
The success of this synthesis hinges on the precise orchestration of crystallization kinetics and chemical racemization. The mechanism begins with the formation of a less soluble diastereomeric salt between the L-enantiomer of the 2,6-dimethyl-tyrosine ester and the chiral resolving agent, L-dibenzoyl tartaric acid. As this salt precipitates out of the alcoholic solvent, typically isopropanol, the concentration of the L-isomer in the solution decreases. Simultaneously, the D-isomer, which remains soluble, undergoes acid-catalyzed imine formation with the added salicylaldehyde. This transient imine intermediate facilitates the epimerization at the alpha-carbon, effectively scrambling the stereochemistry and regenerating the racemic mixture in the solution phase. As the L-isomer is replenished through this racemization loop, it continues to crystallize out as the stable salt. This continuous cycle drives the reaction towards near-quantitative conversion of the racemic starting material into the single L-enantiomer salt, a process visually summarized in the reaction scheme below.
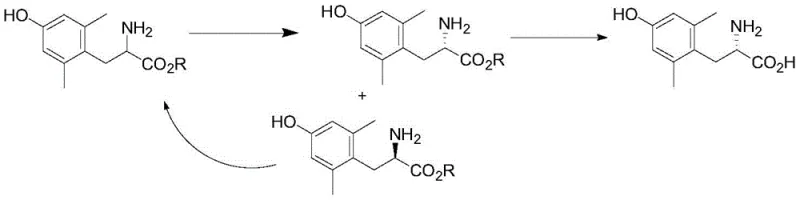
From an impurity control perspective, this mechanism offers distinct advantages over catalytic hydrogenation. Since no metal catalysts are involved, the risk of metal-induced side reactions or residual metal contamination is entirely eliminated. The primary impurities are derived from the organic reagents themselves, such as unreacted aldehyde or resolving agent, which are generally easier to remove via aqueous workups or recrystallization than trace metals bound to organic matrices. The patent data indicates that optimizing the molar ratio of the resolving agent to the substrate, preferably around 1.0:1.5, is critical for maximizing both yield and enantiomeric excess. Furthermore, the choice of solvent plays a vital role; isopropanol provides the ideal balance of solubility for the racemate and insolubility for the target diastereomeric salt, ensuring efficient crystal growth and high purity. The ability to tune the temperature between 30°C and 80°C allows process engineers to fine-tune the nucleation rate, preventing the occlusion of mother liquor and ensuring the formation of well-defined crystals that filter easily.
How to Synthesize 2,6-Dimethyl-L-Tyrosine Efficiently
The practical implementation of this technology involves a logical three-stage sequence designed for maximum throughput and minimal waste. Initially, the readily available racemic 2,6-dimethyl-tyrosine undergoes esterification, typically using ethanol and thionyl chloride, to generate the reactive ester substrate required for resolution. This esterification step is crucial as it modifies the solubility profile of the amino acid, making it amenable to the subsequent organic-phase resolution. Following esterification, the core dynamic kinetic resolution takes place in an alcoholic medium, where the interplay between the resolving agent and the racemization catalyst drives the enrichment of the L-isomer. Finally, the isolated double salt is subjected to acidic hydrolysis to cleave the ester group and liberate the free amino acid, yielding the target 2,6-dimethyl-L-tyrosine in high purity. For a detailed breakdown of the specific operating parameters, stoichiometry, and workup procedures validated by experimental data, please refer to the standardized synthesis guide below.
- Esterify racemic 2,6-dimethyl-tyrosine with alcohol (e.g., ethanol) using thionyl chloride to form the corresponding ester.
- Perform dynamic kinetic resolution in isopropanol using L-dibenzoyl tartaric acid as the resolving agent and salicylaldehyde with sulfuric acid as the racemization system to precipitate the L-isomer double salt.
- Hydrolyze the isolated L-isomer double salt under acidic conditions to release the final 2,6-dimethyl-L-tyrosine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from noble metal catalysis to this organic resolution process represents a significant opportunity for cost optimization and risk mitigation. The elimination of expensive rhodium or ruthenium catalysts removes a major volatile cost component from the bill of materials, stabilizing the cost structure against fluctuations in precious metal markets. Furthermore, the reliance on bulk commodity chemicals such as sulfuric acid, salicylaldehyde, and isopropanol ensures a robust and diversified supply base, reducing the risk of single-source bottlenecks. The simplicity of the unit operations—primarily stirring, heating, and filtration—means that the process can be easily scaled in existing multipurpose facilities without the need for specialized high-pressure infrastructure. This flexibility enhances supply chain resilience, allowing for rapid ramp-up of production volumes to meet fluctuating market demands for peptide intermediates.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete avoidance of precious metal catalysts and complex chiral ligands, which traditionally account for a substantial portion of raw material costs in asymmetric synthesis. By replacing these high-value inputs with inexpensive organic acids and aldehydes, the direct material cost is drastically reduced. Additionally, the simplified downstream processing eliminates the need for specialized metal scavenging resins or extensive chromatographic purification, further lowering operational expenditures. The ability to recover and recycle the resolving agent from the aqueous phase, as demonstrated in the patent examples, adds another layer of economic efficiency by minimizing reagent consumption over multiple batches.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including L-dibenzoyl tartaric acid and salicylaldehyde, are widely produced commodity chemicals with stable global supply chains. This contrasts sharply with proprietary chiral ligands that may be sourced from a single supplier, creating potential vulnerability. The use of standard solvents like isopropanol and ethanol ensures that solvent availability is never a constraint, even during periods of global supply tightness. Moreover, the ambient pressure and moderate temperature conditions reduce the safety risks associated with high-pressure hydrogenation, simplifying logistics and storage requirements for hazardous materials and ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this resolution method aligns perfectly with green chemistry initiatives. The absence of heavy metals significantly reduces the toxicity of the waste stream, simplifying wastewater treatment and disposal compliance. The process generates primarily organic waste that can often be incinerated for energy recovery or treated biologically, unlike metal-contaminated sludge which requires hazardous waste handling. The crystallization-based isolation is inherently scalable; filtration and drying are unit operations that translate linearly from pilot plant to commercial tonnage, ensuring that the high yields and purity observed in the lab can be reliably reproduced in 100 MT annual production campaigns without loss of efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a realistic overview of what manufacturers can expect when adopting this route. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: Why is dynamic kinetic resolution preferred over asymmetric hydrogenation for 2,6-dimethyl-L-tyrosine?
A: Traditional asymmetric hydrogenation relies on expensive noble metal catalysts like rhodium or ruthenium and complex chiral ligands, which significantly increase production costs and introduce risks of heavy metal contamination. The dynamic kinetic resolution method described in patent CN110467539B utilizes inexpensive organic acids and aldehydes, avoiding metal residues entirely while achieving yields theoretically impossible for standard resolution.
Q: How does the racemization agent improve the overall yield in this process?
A: In a standard resolution, the maximum theoretical yield is limited to 50% because only one enantiomer is harvested. By introducing a racemization agent like salicylaldehyde alongside an acid catalyst, the unwanted D-isomer remaining in the mother liquor is continuously converted back into a racemic mixture. This allows the desired L-isomer to be continuously crystallized out, pushing the total yield significantly beyond the 50% barrier.
Q: What are the critical parameters for ensuring high optical purity during the resolution step?
A: The selection of the resolving agent is paramount, with L-dibenzoyl tartaric acid demonstrating superior performance compared to camphorsulfonic acid or simple tartaric acid. Additionally, maintaining the reaction temperature between 30°C and 80°C and optimizing the molar ratio of the resolving agent to the substrate (preferably around 1.5:1) are essential to ensure the selective precipitation of the L-isomer double salt with high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dimethyl-L-Tyrosine Supplier
The technological advancements detailed in patent CN110467539B underscore the immense potential for producing high-quality 2,6-dimethyl-L-tyrosine through efficient, metal-free resolution. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale experiments to industrial manufacturing is seamless and reliable. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify enantiomeric excess and impurity profiles, guaranteeing that every batch meets the exacting standards required for peptide drug synthesis.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. By leveraging our optimized resolution protocol, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free route can improve your bottom line. We encourage you to contact our technical procurement team to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your specific project needs. Let us partner to secure a sustainable and cost-effective supply of high-purity 2,6-dimethyl-L-tyrosine for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
