Advanced Synthesis of 8-Hydroxy Octanal for High-Purity Royal Jelly Acid Production
Introduction to High-Efficiency 8-Hydroxy Octanal Synthesis
The global demand for Royal Jelly Acid, chemically known as trans-10-hydroxy-2-decenoic acid (10-HDA), has surged due to its potent immunological and anti-tumor properties found in royal jelly. As the quality indicator for royal jelly products, the efficient synthesis of its key precursor, 8-hydroxy octanal, represents a critical bottleneck in the nutraceutical and pharmaceutical supply chains. Traditional extraction methods from natural sources are plagued by low yields and prohibitive costs, necessitating a shift toward robust chemical synthesis. Patent CN102010308B introduces a groundbreaking preparation method that utilizes 1,8-octanediol as a starting material, employing a unique carrier-adsorption technique combined with TEMPO-catalyzed oxidation. This innovation not only streamlines the synthetic route but also achieves remarkable selectivity, positioning it as a superior alternative for reliable pharmaceutical intermediate supplier networks seeking to optimize their production of high-value bioactive compounds.
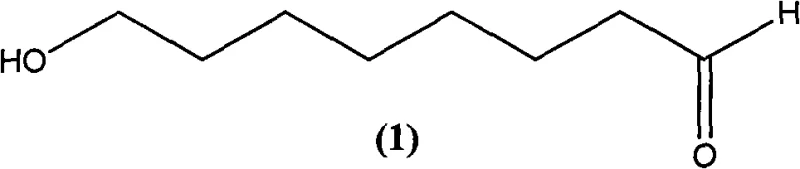
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-hydroxy octanal has been hindered by complex multi-step sequences that are ill-suited for modern industrial scale-up. Prior art, such as the method reported in the Indian Journal of Chemistry, relies on the coupling of magnesium metal reagents with propenal to extend the carbon chain. This Grignard-based approach is inherently fraught with operational difficulties, including the requirement for strictly anhydrous conditions, the handling of pyrophoric reagents, and the generation of substantial hazardous waste. Furthermore, the linked reaction steps often suffer from mediocre yields, and the purification of the final product is complicated by the presence of various organometallic byproducts. These factors collectively result in a high cost of goods sold (COGS) and significant safety risks, limiting the suitability of such traditional routes for the cost reduction in pharmaceutical intermediates manufacturing that today's market demands.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data revolutionizes the production landscape by utilizing a direct, selective oxidation strategy. By employing 1,8-octanediol as a readily available feedstock, the process bypasses the need for carbon-chain extension entirely. The core innovation lies in the physical adsorption of the diol onto a solid carrier, such as silica gel or diatomite, prior to the oxidation step. This heterogeneous-like environment, combined with the catalytic power of 2,2,6,6-tetramethylpiperidine-1-oxide (TEMPO) and a hypochlorite oxidant, facilitates a highly efficient transformation. The reaction proceeds under mild conditions with reaction times ranging from 0.1 to 12 hours, delivering the target aldehyde with exceptional purity. This novel approach effectively eliminates the cumbersome protection and deprotection steps typical of older syntheses, offering a streamlined pathway that is both environmentally friendlier and economically superior.

Mechanistic Insights into TEMPO-Catalyzed Selective Oxidation
The success of this synthesis hinges on the precise mechanistic action of the TEMPO radical catalyst in conjunction with the hypochlorite co-oxidant. TEMPO acts as a stable nitroxyl radical that mediates the oxidation of the primary alcohol group of 1,8-octanediol to the corresponding aldehyde. In the presence of a base and hypochlorite (such as sodium hypochlorite), the TEMPO is oxidized to an oxoammonium cation, which is the active species responsible for abstracting a hydride from the alcohol substrate. This mechanism is highly specific for primary alcohols, which is crucial for differentiating between the two hydroxyl groups or preventing over-oxidation to the carboxylic acid, although the patent notes high selectivity for the aldehyde form under these specific carrier-assisted conditions. The regeneration of the catalyst ensures that only catalytic amounts are required, typically in a molar ratio of 1:10 to 1:100 relative to the substrate, driving down the cost of catalyst consumption significantly.
Furthermore, the role of the solid carrier cannot be overstated in controlling the reaction microenvironment. By adsorbing the 1,8-octanediol onto materials like silica gel or alkaline alumina, the local concentration of the substrate is modulated, which appears to suppress side reactions and enhance the chemo-selectivity of the oxidation. This physical confinement likely restricts the conformational freedom of the long-chain diol, favoring the exposure of the terminal primary alcohol to the catalytic cycle while protecting the internal chain or the other hydroxyl group from aggressive oxidation. This synergy between the homogeneous catalyst and the heterogeneous support creates a unique reaction system that achieves yields of 98% to 100%, a level of efficiency that is rarely seen in traditional liquid-phase oxidations of long-chain diols without extensive protecting group chemistry.
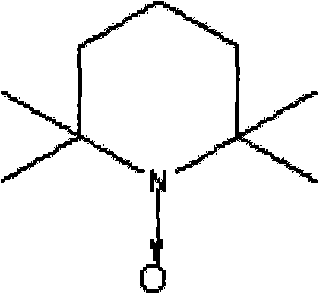
How to Synthesize 8-Hydroxy Octanal Efficiently
Implementing this advanced synthesis protocol requires careful attention to the adsorption step and the controlled addition of the oxidant to maintain thermal stability. The process begins by dissolving the 1,8-octanediol in a volatile solvent like ethyl acetate or ether and mixing it with the chosen carrier, followed by solvent removal to create a free-flowing solid mixture. This solid is then suspended in a reaction solvent such as methylene chloride or petroleum ether, where the TEMPO catalyst and a bromide promoter are introduced. The detailed standardized synthesis steps, including specific stoichiometric ratios, temperature profiles, and workup procedures involving potassium iodide quenching and magnesium sulfate drying, are outlined below to ensure reproducibility and safety in a pilot or production setting.
- Adsorb 1,8-octanediol onto a solid carrier such as silica gel, alumina, or diatomite by dissolving in solvent and removing the solvent under reduced pressure.
- Suspend the adsorbed material in an organic solvent and add TEMPO catalyst followed by a hypochlorite oxidant solution at controlled temperatures between 0°C and 80°C.
- Upon completion, separate the organic phase, wash with reducing agents to remove excess oxidant, dry over magnesium sulfate, and concentrate to isolate the pure aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this TEMPO-mediated oxidation process offers transformative advantages over legacy synthetic routes. The shift from complex organometallic couplings to a simple oxidation of a commodity diol fundamentally alters the cost structure of the supply chain. By eliminating the need for expensive and hazardous Grignard reagents, the process drastically reduces raw material costs and the associated safety infrastructure investments. Moreover, the use of inexpensive bleach (sodium hypochlorite) as the terminal oxidant ensures that the variable costs remain low and stable, shielding the supply chain from the volatility often seen with specialized fine chemical reagents. This economic efficiency is compounded by the high yields reported, which minimize waste disposal costs and maximize the throughput of existing reactor volumes.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and pyrophoric reagents removes the need for expensive heavy metal scavenging steps and specialized inert atmosphere equipment. This simplification of the unit operations leads to substantial cost savings in both capital expenditure (CAPEX) and operational expenditure (OPEX). The ability to use common solvents and recoverable carriers further drives down the cost per kilogram, making the commercial scale-up of complex pharmaceutical intermediates financially attractive.
- Enhanced Supply Chain Reliability: The starting material, 1,8-octanediol, is a widely available bulk chemical, reducing the risk of supply bottlenecks associated with custom-synthesized precursors. The robustness of the reaction conditions, which tolerate a range of temperatures from 0°C to 80°C, allows for flexible scheduling and reduces the risk of batch failures due to minor process deviations. This reliability ensures consistent delivery schedules, a critical factor for downstream manufacturers of Royal Jelly Acid who operate on tight production timelines.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods, as the primary byproduct is salt water from the hypochlorite reduction. The use of solid carriers facilitates easier filtration and separation, reducing the volume of solvent required for extraction and purification. This aligns with increasingly stringent environmental regulations, allowing manufacturers to scale production to 100 MT levels without incurring prohibitive waste treatment costs, thereby future-proofing the supply chain against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of using a solid carrier in this oxidation process?
A: According to patent CN102010308B, adsorbing the substrate on carriers like silica gel or diatomite significantly enhances chemo-selectivity, preventing over-oxidation and simplifying the subsequent purification steps compared to homogeneous reactions.
Q: What yields can be expected from this TEMPO-catalyzed method?
A: The patent data indicates exceptionally high isolated yields ranging from 97% to 99% across multiple embodiments, demonstrating the robustness and efficiency of this catalytic system for industrial application.
Q: Is this process suitable for large-scale manufacturing of Royal Jelly Acid intermediates?
A: Yes, the process utilizes inexpensive reagents like sodium hypochlorite and operates under mild conditions (0-80°C), making it highly scalable and economically viable for the commercial production of high-purity 8-hydroxy octanal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Hydroxy Octanal Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity intermediates like 8-hydroxy octanal in the production of valuable nutraceuticals and pharmaceuticals. Our technical team has extensively analyzed the pathway described in CN102010308B and possesses the expertise to optimize this TEMPO-catalyzed process for GMP-compliant manufacturing. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of residual metals and chlorinated impurities, guaranteeing a product profile that supports the highest standards of downstream synthesis.
We invite global partners to collaborate with us to leverage this cost-effective and scalable technology for their Royal Jelly Acid projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your competitive edge in the global market for bioactive ingredients.
