Advanced Atmospheric Synthesis of Benzomelamine for High-Purity Industrial Applications
Advanced Atmospheric Synthesis of Benzomelamine for High-Purity Industrial Applications
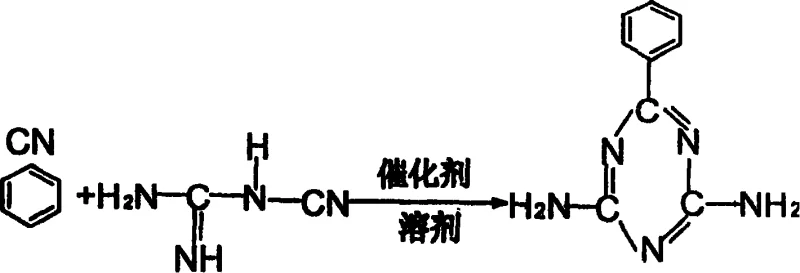
The global demand for high-performance heterocyclic compounds continues to surge, driven by the expanding needs of the coatings, plastics, and dye industries. A pivotal advancement in this sector is detailed in patent CN100391948C, which outlines a robust and economically viable production method for benzomelamine. This technology represents a significant departure from legacy processes by utilizing high-purity dicyandiamide and benzonitrile in the presence of highly active catalysts within low-toxicity solvents. Operating under atmospheric pressure at temperatures between 80-125°C, this method achieves a remarkable yield of approximately 92% and a product purity of 99.8%. For procurement leaders and technical directors seeking a reliable benzomelamine supplier, understanding the nuances of this atmospheric cyclization process is essential for securing a stable supply of this critical fine chemical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzomelamine has been fraught with significant operational hazards and efficiency bottlenecks. Prominent international manufacturers have relied on divergent pathways that introduce substantial complexity; for instance, some legacy routes utilize guanidine carbonate and formaldehyde, which involve handling volatile aldehydes and generate difficult-to-manage byproducts. Other established methods, such as those employing ethylene glycol methyl ether as a solvent, pose severe toxicity risks to personnel and complicate waste stream management due to the solvent's hazardous nature. Furthermore, certain high-pressure processes necessitate the use of methanol under elevated pressures, requiring expensive autoclave reactors and stringent safety interlocks that drastically increase capital expenditure and limit the flexibility of batch scheduling in multi-purpose plants.
The Novel Approach
The methodology described in CN100391948C effectively circumvents these historical constraints by optimizing the reaction environment for both safety and yield. By shifting to solvents such as isobutanol, butanol, or ethanol, the process significantly reduces toxicity profiles while maintaining excellent solubility for the reactants. The decision to operate at atmospheric pressure rather than high pressure simplifies the reactor design, allowing for the use of standard glass-lined or stainless steel vessels that are more readily available and easier to maintain. This novel approach not only streamlines the cost reduction in fine chemical intermediates manufacturing but also ensures a consistent crystal morphology through the strategic addition of seeding crystals, resulting in a product that exhibits superior dissolution characteristics in downstream formaldehyde applications.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this synthesis lies in the base-catalyzed condensation and cyclization of dicyandiamide with benzonitrile. The reaction mechanism involves the nucleophilic attack of the amino groups of dicyandiamide on the nitrile carbon of benzonitrile, facilitated by catalysts such as sodium hydroxide, potassium hydroxide, or aluminum hydroxide. These basic catalysts activate the nitrile group, lowering the energy barrier for the formation of the triazine ring structure. The precise control of the molar ratio between dicyandiamide and benzonitrile, maintained between 1:1 and 1:2, is critical to driving the equilibrium toward the desired product while minimizing the formation of oligomeric byproducts. The use of high-purity starting materials (≥99.5% for dicyandiamide and ≥99.7% for benzonitrile) further suppresses side reactions, ensuring that the final impurity spectrum is dominated by easily removable unreacted starting materials rather than complex structural analogs.
Following the cyclization, the purification mechanism relies on differential solubility and thermal shock. The process involves distilling off a portion of the solvent to concentrate the reaction mixture, followed by the rapid addition of near-freezing water. This sharp temperature drop induces supersaturation, causing the benzomelamine to precipitate rapidly as fine crystals. The subsequent washing steps, which include neutralization with glacial acetic acid, are designed to remove residual alkaline catalysts and any unreacted dicyandiamide that may have co-precipitated. This rigorous washing protocol is what enables the final product to achieve a free base content of less than 0.05% and an ash content as low as 0.009%, meeting the stringent specifications required for high-end fluorescent dye production and specialized polymer additives.
How to Synthesize Benzomelamine Efficiently
Implementing this synthesis route requires careful attention to the sequence of addition and thermal management to maximize the commercial scale-up of complex heterocyclic intermediates. The process begins with charging the reactor with the selected alcohol solvent and reactants, followed by heating to reflux conditions specific to the solvent boiling point. Detailed operational parameters, including the precise timing for solvent recovery and the temperature gradients required for crystallization, are critical for reproducibility. For R&D teams looking to replicate or license this technology, the following standardized synthesis steps outline the proven workflow derived from the patent examples.
- Charge reactor with alcohol solvent (butanol/isobutanol/ethanol), high-purity benzonitrile, dicyandiamide, and a base catalyst (NaOH/KOH/Al(OH)3). Optional: Add seeding crystals.
- Heat mixture to reflux (80-125°C) under atmospheric pressure for 3-6 hours to complete the cyclization reaction.
- Distill off partial solvent, shock cool with ice water, recover remaining solvent via reflux, and wash solid product with water and acetic acid to neutral pH before drying.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition to this atmospheric production method offers tangible strategic benefits beyond mere technical specifications. The elimination of high-pressure requirements fundamentally alters the risk profile of the manufacturing site, reducing insurance premiums and allowing for higher batch frequencies due to shorter turnaround times between runs. Additionally, the use of common alcohol solvents instead of specialized ethers or high-pressure methanol systems simplifies raw material sourcing, ensuring that production is not bottlenecked by the availability of niche chemicals. This robustness in the supply chain is crucial for maintaining continuity in the face of global logistical disruptions, providing buyers with a reliable benzomelamine supplier capable of meeting just-in-time delivery schedules.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of equipment and utility requirements. By operating at atmospheric pressure, manufacturers avoid the substantial capital costs associated with high-pressure autoclaves and the associated safety infrastructure. Furthermore, the catalyst system utilizes inexpensive inorganic bases like sodium hydroxide rather than costly transition metals, which eliminates the need for expensive metal scavenging resins or complex filtration steps to meet heavy metal limits. The ability to recover and recycle the alcohol solvent through simple distillation further contributes to substantial cost savings by minimizing raw material waste and reducing the volume of hazardous waste requiring disposal.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as butanol, ethanol, and benzonitrile ensures that the production of benzomelamine is resilient to supply shocks. Unlike processes dependent on proprietary or tightly controlled reagents, this method leverages a global market for its inputs, allowing for flexible sourcing strategies across different geographic regions. The stability of the reaction conditions also means that production campaigns can be extended without significant degradation in equipment performance, leading to more predictable lead times for high-purity benzomelamine and allowing customers to optimize their own inventory levels with confidence.
- Scalability and Environmental Compliance: From an environmental perspective, the substitution of toxic ethylene glycol methyl ether with lower-toxicity alcohols significantly eases the burden on wastewater treatment facilities and reduces the regulatory compliance overhead. The aqueous wash streams generated during the purification phase are easier to treat biologically compared to ether-containing effluents, facilitating faster permitting for capacity expansions. The process generates a white crystalline powder with low moisture and ash content, reducing the energy load required for final drying and packaging, thereby supporting broader corporate sustainability goals related to energy efficiency and carbon footprint reduction in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of benzomelamine synthesized via this advanced atmospheric method. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on quality benchmarks and process safety. Understanding these details is vital for technical buyers evaluating potential partners for long-term supply agreements.
Q: What are the safety advantages of this benzomelamine production method compared to traditional high-pressure processes?
A: Unlike the high-pressure methods employed by companies like Degussa which require specialized autoclaves and rigorous safety protocols, this patent utilizes an atmospheric pressure reaction. This significantly reduces equipment capital expenditure and operational risk, while the substitution of toxic ethylene glycol methyl ether with lower-toxicity alcohols like butanol or ethanol enhances workplace safety and environmental compliance.
Q: How does the process ensure high purity (99.8%) and optimal crystallization for dye applications?
A: The process achieves 99.8% purity through a multi-stage purification strategy involving the distillation of excess solvent followed by a sharp temperature drop using ice water to precipitate the product. Furthermore, the addition of seeding crystals during the initial charge refines the crystal lattice, resulting in finer particles that dissolve rapidly in formaldehyde, a critical parameter for fluorescent dye manufacturing.
Q: Can this synthesis route be scaled for commercial production without complex transition metal removal?
A: Yes, the route uses inexpensive inorganic base catalysts such as sodium hydroxide or aluminum hydroxide rather than expensive transition metals. This eliminates the need for complex and costly heavy metal scavenging steps during downstream processing, simplifying the scale-up from pilot to commercial tonnage while maintaining a clean impurity profile suitable for sensitive polymer and coating applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzomelamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into consistent, ton-scale reality to truly serve the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in laboratory settings are maintained throughout full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of benzomelamine meets the exacting standards required for fluorescent dyes, coatings, and plastic additives, regardless of the complexity of the synthesis route.
We invite forward-thinking organizations to collaborate with us to leverage this efficient production technology for their specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this atmospheric process can optimize your total cost of ownership. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
