Advanced Aerobic Oxidation Technology for High-Purity 3-Nitro-2-Methylbenzoic Acid Manufacturing
Introduction to Next-Generation Intermediate Synthesis
The global demand for high-purity pharmaceutical and agrochemical intermediates necessitates a paradigm shift from hazardous, waste-intensive legacy processes to sustainable, catalytic technologies. Patent CN111362807B introduces a groundbreaking preparation method for 3-nitro-2-methylbenzoic acid, a critical building block for the antitumor drug lenalidomide and the pesticide methoxyfenozide. This technology replaces dangerous stoichiometric oxidants with molecular oxygen, utilizing a sophisticated cobalt-manganese catalytic system in an acidic organic solvent medium. By precisely controlling reaction parameters such as temperature, oxygen pressure, and solvent water content, this method achieves direct crystallization of the product, bypassing the environmentally damaging neutralization steps characteristic of older nitric acid or permanganate-based routes. For R&D directors and supply chain leaders, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering materials with purity exceeding 98% while adhering to stringent green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-nitro-2-methylbenzoic acid has relied heavily on oxidation using concentrated nitric acid or potassium permanganate, both of which present severe operational and environmental liabilities. Nitric acid oxidation requires high temperatures and pressures, generating substantial quantities of toxic nitrogen oxide gases and low-concentration acidic wastewater that demands complex neutralization treatments, resulting in massive amounts of nitrate salt waste. Alternatively, potassium permanganate oxidation poses significant safety risks due to the potential for combustion and explosion when mixed with organic matter, while simultaneously producing large volumes of manganese dioxide solid waste that is difficult to dispose of ecologically. Furthermore, existing aerobic methods using solvents like o-dichlorobenzene suffer from high boiling points and toxicity, making solvent removal and product purification energy-intensive and hazardous to occupational health. These legacy processes often yield products with residual color and impurities that require multiple recrystallization steps, driving up production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative process disclosed in the patent utilizes molecular oxygen as the sole oxidant in an acetic or propionic acid solvent system, fundamentally altering the reaction landscape towards sustainability and efficiency. By employing a catalyst system comprising cobalt acetate and manganese acetate alongside initiators like sodium bromide or N-hydroxyphthalimide, the reaction proceeds smoothly at moderate temperatures between 110-135°C and oxygen pressures of 1.0-3.0 MPa. A critical breakthrough of this method is the strategic management of water content within the acidic solvent; maintaining a specific mass fraction allows the reactants to dissolve fully during the exothermic oxidation phase while facilitating the direct precipitation of the product upon cooling. This eliminates the need for alkaline workups and subsequent acidification, thereby preventing the formation of inorganic salt by-products. The result is a streamlined workflow where the crude product can be filtered directly from the reaction mixture, significantly simplifying downstream processing and enhancing the overall yield and purity profile suitable for sensitive API applications.
Mechanistic Insights into Co/Mn-Catalyzed Aerobic Oxidation
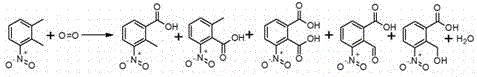
The core of this technological advancement lies in the radical-mediated oxidation mechanism facilitated by the cobalt-manganese-bromide catalyst system. In this cycle, the transition metal ions undergo redox changes to activate molecular oxygen, generating reactive radical species that selectively abstract hydrogen atoms from the methyl group of the 3-nitro-o-xylene substrate. The presence of the initiator is crucial for generating the initial radical flux, ensuring rapid onset of the reaction and minimizing induction periods that could lead to accumulation of unstable intermediates. The acidic environment provided by the acetic acid solvent stabilizes the high-valent metal-oxo species and prevents the precipitation of metal hydroxides, maintaining catalytic activity throughout the 4 to 16-hour reaction duration. Crucially, the solvent system acts not merely as a medium but as a dynamic participant in the crystallization thermodynamics; as the reaction progresses, water is generated as a by-product of oxidation, gradually altering the polarity of the solution. This in-situ generation of water, combined with the initial controlled water content, creates a supersaturated environment upon cooling that drives the selective nucleation of 3-nitro-2-methylbenzoic acid while keeping soluble impurities and unreacted starting materials in the mother liquor.
Impurity control is rigorously managed through the specific solvent-to-substrate ratios and the subsequent recycling strategy for the mother liquor. The patent highlights that unreacted 3-nitro-o-xylene and isomeric by-products like 2-methyl-6-nitrobenzoic acid remain soluble in the acidic mother liquor under the optimized crystallization conditions, preventing their co-precipitation with the target acid. Furthermore, the process includes a sophisticated valorization step for the concentrated mother liquor; instead of discarding this complex mixture, it is subjected to further oxidation with nitric acid and oxygen to convert residual organics into 3-nitrophthalic acid, a valuable chemical in its own right. This closed-loop approach not only maximizes atom economy but also ensures that the final product obtained after recrystallization with methanol or ethanol meets the stringent purity specifications required for oncology drug synthesis, with HPLC analysis confirming purity levels consistently above 98%.
How to Synthesize 3-Nitro-2-Methylbenzoic Acid Efficiently
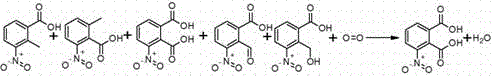
The synthesis protocol outlined in the patent provides a robust framework for scaling this reaction from laboratory benchtop to industrial manufacturing scales. The process begins with the precise charging of 3-nitro-o-xylene into an oxidation reactor containing glacial acetic acid with a controlled water content, followed by the addition of the dissolved catalyst package. Maintaining strict control over the oxygen partial pressure and temperature ramping is essential to manage the exotherm and ensure complete conversion of the starting material, typically monitored until the residual 3-nitro-o-xylene content drops below 10%. Following the reaction completion, the mixture is cooled to induce crystallization, filtered under inert atmosphere to remove oxygen, and the resulting cake is recrystallized to achieve final specification. For detailed operational parameters, safety interlocks, and specific workup procedures, please refer to the standardized synthesis guide below.
- Oxidize 3-nitro-o-xylene in acetic acid solvent using oxygen pressure (1.0-3.0 MPa) and a Co/Mn catalyst system at 110-135°C.
- Cool the reaction mixture to 10-30°C to induce crystallization of the crude product, followed by filtration and oxygen removal.
- Recrystallize the crude solid using methanol or ethanol to achieve final purity exceeding 98%, while recovering mother liquor for byproduct synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aerobic oxidation technology translates into tangible strategic advantages regarding cost stability and regulatory compliance. The elimination of stoichiometric oxidants like nitric acid and permanganate removes the volatility associated with the sourcing and handling of these hazardous bulk chemicals, thereby stabilizing the raw material supply chain. Moreover, the drastic reduction in waste generation—specifically the avoidance of tons of nitrate salts and manganese sludge per ton of product—significantly lowers the operational expenditure related to waste disposal and environmental remediation fees. The ability to recover and reuse the acetic acid solvent further compounds these savings, reducing the overall consumption of organic solvents and insulating the production cost from fluctuations in solvent market prices. This process inherently supports cost reduction in pharmaceutical intermediate manufacturing by simplifying the unit operations; fewer steps mean less equipment downtime, lower labor requirements, and reduced energy consumption for heating and cooling cycles.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the neutralization and acidification steps traditionally required to isolate carboxylic acids from reaction mixtures. By leveraging the solubility properties of the acetic acid-water system to precipitate the product directly, the method eliminates the need for large quantities of caustic soda and mineral acids, which are significant cost centers in conventional batch processing. Additionally, the catalyst loading is minimal, representing less than 1% of the substrate weight, and the efficient recovery of the solvent means that the variable cost per kilogram of product is substantially lower than legacy methods. The valorization of the mother liquor into 3-nitrophthalic acid creates an additional revenue stream that offsets the production costs of the primary intermediate, effectively subsidizing the manufacturing of the main API precursor.
- Enhanced Supply Chain Reliability: Relying on molecular oxygen as the oxidant removes the dependency on external suppliers for specialized oxidizing agents, which can be subject to logistical bottlenecks and regulatory transport restrictions. Oxygen is readily available on-site through generation units or bulk supply, ensuring uninterrupted production continuity even during global supply chain disruptions. The robustness of the catalyst system and the tolerance for varying water content in the solvent provide a wide operating window, reducing the risk of batch failures due to minor raw material specification deviations. This reliability is critical for maintaining just-in-time delivery schedules for downstream pharmaceutical manufacturers who require consistent quality and volume to support their own clinical or commercial production timelines.
- Scalability and Environmental Compliance: The process is designed with inherent scalability, utilizing standard stainless steel pressure reactors common in the fine chemical industry, which facilitates rapid technology transfer from pilot plant to commercial scale. From an environmental perspective, the substitution of toxic chlorinated solvents like o-dichlorobenzene with acetic acid aligns with increasingly stringent global environmental regulations and corporate sustainability goals. The reduction in E-factor (mass of waste per mass of product) makes this facility easier to permit and operate in regions with strict effluent limitations, future-proofing the supply chain against tightening environmental legislation. This green profile enhances the brand reputation of the supply chain partners and reduces the liability associated with hazardous waste storage and transportation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced oxidation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is essential for making informed decisions about supplier qualification and process integration.
Q: How does this aerobic oxidation method improve environmental compliance compared to nitric acid oxidation?
A: Unlike traditional nitric acid oxidation which generates large volumes of nitrogen oxide gases and acidic wastewater requiring neutralization, this method uses molecular oxygen as the oxidant. The primary by-product is water, significantly reducing the load on wastewater treatment facilities and eliminating the need for extensive acid-base neutralization steps that produce solid salt waste.
Q: What is the role of water content control in the acetic acid solvent system?
A: Controlling the water mass fraction between 0-17% in the initial solvent is critical. It ensures sufficient solubility for the catalyst and reactants during the high-temperature reaction phase while allowing the product to crystallize directly upon cooling. This eliminates the need for complex extraction or neutralization processes typically required to isolate the product from organic solvents.
Q: Can the mother liquor from this process be utilized to generate additional value?
A: Yes, the concentrated mother liquor containing unreacted raw materials and by-products can be further oxidized using nitric acid and oxygen to produce 3-nitrophthalic acid. This valorization strategy turns potential waste streams into a secondary commercially valuable chemical intermediate, enhancing the overall economic efficiency of the production line.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitro-2-Methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is vital for the long-term competitiveness of the pharmaceutical and agrochemical industries. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111362807B are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped with high-pressure oxidation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-nitro-2-methylbenzoic acid delivered meets the exacting standards required for oncology and crop protection formulations. We are committed to providing a secure, compliant, and cost-effective supply of this critical intermediate to support your global development programs.
We invite you to engage with our technical procurement team to discuss how this innovative oxidation technology can be integrated into your supply chain to drive efficiency and reduce total landed costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this aerobic process compared to your current sourcing model. We encourage you to contact us today to obtain specific COA data from our recent pilot batches and to receive comprehensive route feasibility assessments tailored to your project timelines and volume requirements.
