Advanced Liquid-Phase Synthesis Strategy for High-Purity Synthetic Pentapeptide Intermediates
Advanced Liquid-Phase Synthesis Strategy for High-Purity Synthetic Pentapeptide Intermediates
The pharmaceutical industry continuously seeks robust manufacturing routes for complex peptide therapeutics, particularly those targeting specific receptor subtypes like the peripheral kappa opioid receptor. Patent CN106459150B introduces a groundbreaking methodology for the industrial production of a specific synthetic pentapeptide, designated as Compound (A), which serves as a potent kappa opioid receptor agonist. Unlike traditional approaches that struggle with scalability and purity, this invention leverages a sophisticated liquid-phase peptide synthesis strategy. By focusing on the strategic isolation of a novel intermediate, Compound (1), the process circumvents the notorious purification bottlenecks associated with solid-phase synthesis. This technical breakthrough not only ensures the delivery of high-purity active pharmaceutical ingredients but also establishes a new benchmark for efficiency in peptide manufacturing, making it a critical development for any reliable pharmaceutical intermediate supplier aiming to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of complex pentapeptides like Compound (A) has relied heavily on solid-phase peptide synthesis (SPPS). While SPPS is effective for small-scale library generation, it presents severe limitations when transitioning to industrial manufacturing. The primary bottleneck lies in the final purification stage; after cleavage from the resin and global deprotection, the crude product contains a complex mixture of deletion sequences and diastereomers that are structurally similar to the target molecule. Separating these impurities typically necessitates the use of large-scale preparative High-Performance Liquid Chromatography (HPLC). This requirement imposes a massive capital expenditure on equipment and consumes vast quantities of organic solvents, drastically inflating the cost of goods. Furthermore, the reliance on specialized resins and limited solvent compatibility in SPPS restricts the flexibility needed for large-scale optimization, often resulting in yields that are economically unviable for commercial supply chains.
The Novel Approach
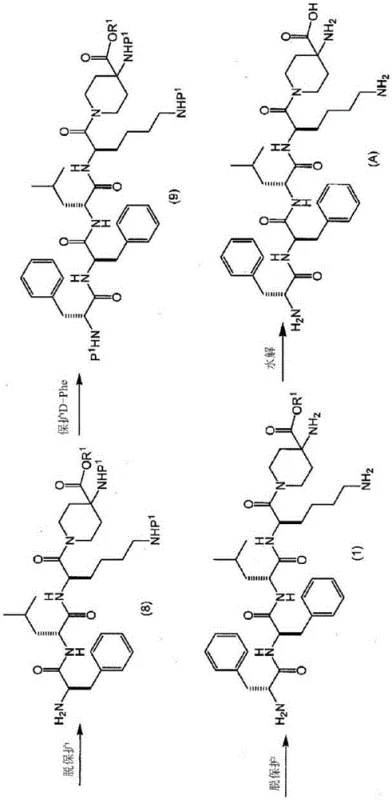
The methodology disclosed in the patent fundamentally shifts the paradigm by employing a solution-phase (liquid-phase) synthesis route that prioritizes intermediate purification. Instead of attempting to purify the final deprotected peptide directly, the process isolates a fully assembled but partially protected intermediate, specifically the methyl ester derivative known as Compound (1). This intermediate is designed with orthogonal protecting groups that allow for the selective removal of the N-terminal protection while retaining the C-terminal ester. This chemical design enables the intermediate to be converted into a stable acid addition salt, such as a hydrochloride salt. Consequently, purification can be achieved through simple, scalable unit operations like slurry processing and recrystallization. This approach eliminates the dependency on preparative HPLC, significantly reducing solvent consumption and equipment costs while achieving purity levels exceeding 99%, which is difficult to attain consistently with conventional solid-phase methods.
Mechanistic Insights into Orthogonal Protection and Intermediate Isolation
The success of this liquid-phase strategy hinges on a meticulously planned orthogonal protection scheme involving two distinct N-protecting groups, denoted as P1 and P2. In the preferred embodiment, P1 is an acid-labile group such as tert-butoxycarbonyl (Boc), while P2 is a group removable by hydrogenolysis, such as benzyloxycarbonyl (Cbz). The synthesis proceeds by sequentially condensing protected amino acid fragments—starting from 4-aminopiperidine-4-carboxylic acid derivatives and extending through D-Lysine, D-Leucine, and D-Phenylalanine residues. The use of carbodiimide coupling agents like EDC·HCl in conjunction with additives like HOBt ensures high coupling efficiency and minimizes racemization. The critical mechanistic step occurs after the full pentapeptide backbone is assembled. By selectively cleaving the P1 group using acidic conditions, the N-terminus is exposed without disturbing the P2 group or the C-terminal ester. This specific chemical state allows the molecule to form a crystalline salt, facilitating the removal of soluble impurities through filtration and washing, a physical separation mechanism that is far more robust than chromatographic methods.
Furthermore, the control of impurities is inherently built into the stepwise nature of the liquid-phase synthesis. In solid-phase synthesis, incomplete reactions at any step lead to deletion peptides that accumulate and are only removed at the very end. In contrast, the liquid-phase approach allows for the purification of intermediates at multiple stages if necessary, although the patent highlights the efficiency of purifying specifically at the Compound (1) stage. The final conversion to Compound (A) involves a mild hydrolysis of the methyl ester under basic conditions, followed by neutralization. Because the precursor Compound (1) is already of exceptionally high purity (often greater than 98% prior to hydrolysis), the final product inherits this purity profile. This mechanistic advantage ensures that the final API intermediate meets stringent regulatory specifications for impurity profiles, including limits on diastereomers and related substances, without requiring aggressive downstream processing.
How to Synthesize D-Phe-D-Phe-D-Leu-D-Lys-Pic Efficiently
The synthesis of this high-value pentapeptide requires precise control over reaction stoichiometry, temperature, and solvent selection to maximize yield and optical purity. The process begins with the activation of carboxylic acid components and their subsequent coupling to amine partners in solvents such as ethyl acetate, which offers a favorable balance of solubility and environmental safety compared to traditional chlorinated solvents. The key to operational success lies in the workup procedures following each coupling step, where aqueous washes effectively remove urea byproducts and excess reagents. The pivotal moment in the workflow is the deprotection of the fully protected pentapeptide ester to generate the key intermediate salt. This step must be monitored carefully to ensure complete removal of the acid-labile protecting group without inducing side reactions. Once the salt is isolated, the purification via slurry or recrystallization provides a polished intermediate ready for the final hydrolysis. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized synthesis guide below.
- Sequentially condense protected amino acids (4-aminopiperidine-4-carboxylic acid, D-Lys, D-Leu, D-Phe) using carbodiimide coupling agents in organic solvents.
- Selectively remove the N-terminal protecting group (P1) to isolate the key intermediate compound (1) as a stable salt.
- Purify intermediate (1) via slurry or recrystallization methods, followed by final hydrolysis to yield the high-purity target compound (A).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from solid-phase to this novel liquid-phase manufacturing process represents a significant opportunity for cost reduction in peptide manufacturing. The most immediate financial benefit stems from the elimination of preparative HPLC purification. Preparative chromatography is not only capital-intensive regarding equipment but also operationally expensive due to the high cost of chromatography-grade solvents and column media. By replacing this with crystallization and filtration, the variable costs per kilogram of product are drastically reduced. Additionally, the solvents utilized in this liquid-phase route, such as ethyl acetate and isopropanol, are commodity chemicals with stable pricing and widespread availability, unlike some specialized solvents often required for resin swelling and washing in SPPS. This shift translates directly into a more competitive pricing structure for the final intermediate, allowing downstream drug manufacturers to improve their margins or invest more in clinical development.
From a supply chain reliability perspective, this process offers enhanced scalability and continuity. Solid-phase synthesis is often constrained by reactor volume relative to resin capacity, making the jump from kilogram to multi-ton scale challenging and risky. Liquid-phase synthesis, however, utilizes standard stirred-tank reactors that are ubiquitous in the fine chemical industry. This compatibility means that production can be scaled up linearly with much lower technical risk, ensuring that supply can meet demand spikes without long lead times for specialized equipment installation. Furthermore, the robustness of the crystallization purification step ensures consistent quality batch-after-batch, reducing the risk of supply disruptions caused by failed purification runs. The ability to produce high-purity synthetic pentapeptide intermediates with such reliability makes this route highly attractive for long-term supply agreements and strategic partnerships.
Environmental compliance and waste management are also significantly improved, contributing to a more sustainable supply chain. Solid-phase peptide synthesis generates substantial amounts of solid waste in the form of spent resin, along with large volumes of solvent waste from repeated washing cycles. The liquid-phase method described in the patent minimizes solid waste entirely and reduces overall solvent consumption through efficient crystallization processes. This reduction in waste volume simplifies disposal logistics and lowers the environmental footprint of the manufacturing site. For global pharmaceutical companies with strict sustainability goals, sourcing intermediates produced via this greener chemistry pathway aligns with corporate responsibility initiatives. The combination of economic efficiency, scalable infrastructure, and environmental stewardship positions this manufacturing method as a superior choice for the commercial scale-up of complex peptides.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this synthetic pentapeptide. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific pipeline candidates. The focus is on clarifying the purity advantages, the nature of the intermediates, and the scalability of the process to ensure all stakeholders have a clear understanding of the value proposition offered by this technology.
Q: Why is the liquid-phase method superior to solid-phase synthesis for this pentapeptide?
A: The liquid-phase method allows for the isolation and purification of intermediates using cost-effective crystallization techniques, avoiding the expensive preparative HPLC required in solid-phase synthesis to remove difficult impurities.
Q: What is the critical intermediate in this new manufacturing process?
A: The critical intermediate is compound (1), a partially deprotected pentapeptide ester. Its ability to form stable salts enables high-purity purification via simple slurry or recrystallization methods before final hydrolysis.
Q: How does this process impact supply chain reliability for opioid agonist intermediates?
A: By utilizing standard organic solvents and avoiding specialized resin-based equipment, the process is significantly easier to scale from laboratory to commercial production, ensuring consistent supply continuity and reduced lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Synthetic Pentapeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced manufacturing technologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the liquid-phase synthesis route described in CN106459150B and is fully prepared to implement this methodology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with state-of-the-art reaction vessels and rigorous QC labs capable of meeting stringent purity specifications required for opioid receptor agonists and other potent therapeutic intermediates. We are committed to delivering high-purity synthetic pentapeptide intermediates that adhere to the highest quality standards.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective and scalable synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and preliminary quotations. Let us help you secure a reliable supply chain for your next-generation pain management therapeutics while optimizing your manufacturing costs and timelines.
