Advanced Synthetic Route for Dorzolamide Intermediates Enhancing Safety and Yield
Advanced Synthetic Route for Dorzolamide Intermediates Enhancing Safety and Yield
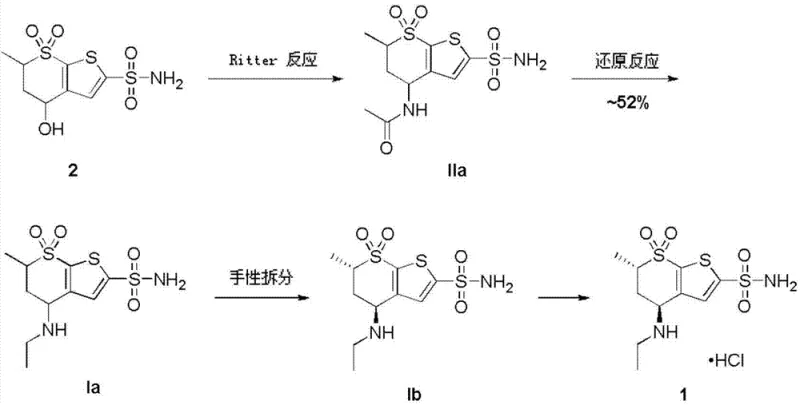
The pharmaceutical industry continuously seeks robust manufacturing processes that balance high purity with operational safety, particularly for critical ophthalmic medications like dorzolamide hydrochloride. Patent CN103497202A introduces a transformative synthetic methodology for producing key intermediates Ia and Ib, which are essential precursors in the manufacture of this carbonic anhydrase inhibitor. This innovation addresses long-standing safety concerns associated with traditional reduction techniques by replacing hazardous borane complexes with a controlled sodium borohydride and iodine system. By shifting away from toxic reagents and complex purification steps, this technology offers a streamlined pathway that aligns with modern green chemistry principles while maintaining the rigorous quality standards required for glaucoma treatment APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dorzolamide intermediates has relied on chemically aggressive and logistically challenging reagents that pose significant risks to both personnel and the environment. As illustrated in earlier patents such as US4797413, the conventional Route 1 utilizes borane dimethyl sulfide complexes for the critical reduction step, a substance known for its extreme toxicity, unpleasant odor, and high procurement costs. Furthermore, alternative pathways like Route 2, disclosed in US5688968, employ boron trifluoride diethyl etherate to generate borane in situ, which introduces severe moisture sensitivity and generates fluorine-containing wastewater that is notoriously difficult and expensive to treat. These legacy methods often require energy-intensive chiral column chromatography for purification, creating bottlenecks that hinder efficient commercial scale-up and increase the overall cost of goods sold.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the novel approach detailed in the patent utilizes a sodium borohydride and iodine system to effect the reduction of the acetamido group to the ethylamino group under remarkably mild conditions. This method completely eliminates the need for handling free borane gas or toxic borane-sulfide complexes, thereby removing the associated risks of violent heat release or acute gas emissions during post-processing workups. The reaction proceeds smoothly in common organic solvents such as tetrahydrofuran or diethyl ether at temperatures controlled below 10°C, ensuring a stable thermal profile that is ideal for large-scale reactor operations. By simplifying the workup procedure to standard acid-base extraction and distillation, this technology drastically reduces processing time and waste generation, offering a commercially superior alternative for the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Sodium Borohydride Iodine Reduction
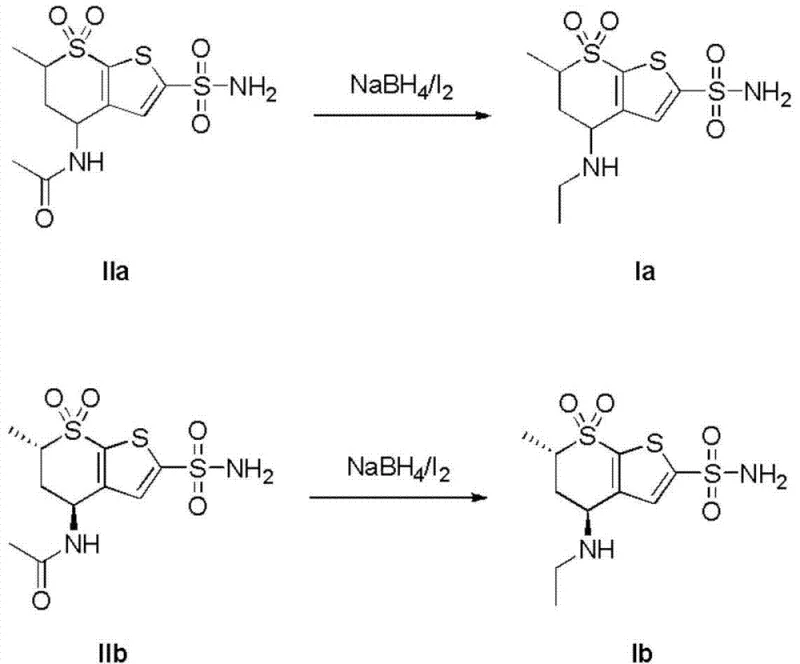
The core chemical innovation lies in the in situ generation of a reactive borane species through the interaction of sodium borohydride and iodine, which serves as the active reducing agent for the acetamido functionality. Unlike direct addition of unstable borane sources, this controlled generation allows for precise stoichiometric management, typically utilizing a molar ratio of sodium borohydride to iodine between 1.5:1 and 3:1, with 2:1 being optimal for maximizing conversion efficiency. The mechanism involves the formation of borane-ether complexes that selectively reduce the amide bond to the corresponding amine without affecting other sensitive functional groups on the thienothiopyran scaffold, such as the sulfonamide moiety. This selectivity is crucial for maintaining the structural integrity of the intermediate and preventing the formation of difficult-to-remove impurities that could compromise the final API quality.
From an impurity control perspective, the mild nature of this reduction system significantly minimizes side reactions such as over-reduction or ring-opening, which are common pitfalls in harsher reducing environments. The post-reaction quenching process involves the careful addition of ketones or alcohols to consume excess reducing agents, followed by acid hydrolysis to break N-B bonds, ensuring that the final product is free from boron-containing residues. The subsequent pH adjustment to a neutral range of 6-8 prior to extraction further purifies the organic phase, resulting in intermediates with HPLC purity levels consistently exceeding 98%. This high level of intrinsic purity reduces the burden on downstream crystallization steps, ensuring that the final dorzolamide hydrochloride meets stringent pharmacopeial specifications with minimal additional processing.
How to Synthesize Dorzolamide Intermediate Efficiently
The synthesis of these critical intermediates involves a carefully orchestrated sequence of reagent addition and temperature control to ensure safety and reproducibility. Operators must dissolve the acetamido precursor and sodium borohydride in an anhydrous solvent under inert atmosphere before slowly introducing the iodine solution to manage the exotherm effectively. Following the reaction period, the mixture is quenched with alcohol and treated with dilute acid to liberate the free amine, which is then isolated via solvent extraction and concentration. For a comprehensive understanding of the specific operating parameters and safety precautions, please refer to the detailed standardized synthesis steps provided in the guide below.
- Dissolve the acetamido precursor (Compound IIa) and sodium borohydride in an anhydrous solvent such as tetrahydrofuran or diethyl ether, cooling the mixture to below 10°C.
- Slowly add a solution of iodine in the same solvent dropwise while maintaining the temperature below 10°C to generate the active reducing species in situ.
- After reaction completion, quench excess reagent with alcohol, acidify with dilute sulfuric or hydrochloric acid, adjust pH to 6-8, and extract the product with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route represents a strategic opportunity to mitigate risk and optimize the cost structure of dorzolamide manufacturing. By transitioning away from highly regulated and hazardous raw materials like borane dimethyl sulfide, companies can significantly reduce the administrative and logistical burdens associated with storing and transporting dangerous goods. The elimination of fluorine-containing reagents also simplifies wastewater treatment protocols, leading to substantial cost savings in environmental compliance and waste disposal fees. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed batches or extensive rework.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic borane complexes with commodity chemicals like sodium borohydride and iodine drives down raw material costs significantly. Additionally, the simplified workup procedure eliminates the need for costly chiral column chromatography in the racemic route, replacing it with standard extraction and distillation techniques that are far more economical at scale. The high yields reported in the patent embodiments, often exceeding 90%, further contribute to cost efficiency by maximizing the output per unit of starting material and minimizing solvent consumption.
- Enhanced Supply Chain Reliability: Relying on widely available and stable reagents such as sodium borohydride ensures a secure supply chain that is less susceptible to market volatility compared to specialized borane complexes. The mild reaction conditions reduce the risk of equipment corrosion or safety incidents, thereby enhancing plant uptime and ensuring continuous production schedules. This reliability is critical for meeting the demanding delivery timelines of global pharmaceutical clients who require uninterrupted supply of key intermediates for their finished dosage forms.
- Scalability and Environmental Compliance: The absence of violent exotherms or gas evolution during the quenching phase makes this process inherently safer and easier to scale from pilot plant to commercial production volumes. The reduction in hazardous waste generation, particularly the avoidance of fluorine and sulfur-based toxic byproducts, aligns with increasingly strict environmental regulations, facilitating smoother regulatory approvals and permitting processes. This environmental stewardship not only protects the ecosystem but also enhances the corporate reputation of manufacturers as responsible partners in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthetic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is essential for making informed decisions about process adoption and supplier qualification.
Q: Why is the NaBH4/I2 system preferred over borane dimethyl sulfide for dorzolamide synthesis?
A: The NaBH4/I2 system eliminates the need for highly toxic and malodorous borane dimethyl sulfide complexes. It offers milder reaction conditions without violent heat release or gas emission during post-processing, significantly enhancing operational safety and environmental compliance.
Q: What yields can be expected from this novel reduction process?
A: Experimental data from the patent indicates consistently high yields ranging from 90% to 97% for the racemic intermediate Ia and 92% to 95% for the chiral intermediate Ib, demonstrating superior efficiency compared to traditional chromatographic separation methods.
Q: Which solvents are compatible with this reduction protocol?
A: The process is versatile regarding solvent choice, successfully utilizing tetrahydrofuran (THF), diethyl ether, methyl tert-butyl ether (MTBE), and isopropyl ether, allowing manufacturers to optimize based on cost and recovery capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dorzolamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for high-value pharmaceutical intermediates like dorzolamide precursors. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and compliant. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your operational efficiency and product quality.
