Advanced Nicotine Racemization: Transforming Chiral Resolution Waste into High-Purity API Intermediates
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of nicotine has relied heavily on extraction from tobacco plants or complex total synthesis routes, both of which present significant logistical and chemical challenges for modern pharmaceutical supply chains. Traditional total synthesis often involves harsh conditions and expensive reagents; for instance, early methodologies utilized flammable organolithium metals like butyllithium and required cryogenic temperatures as low as -78°C to maintain reaction control, as illustrated in classical synthetic pathways. 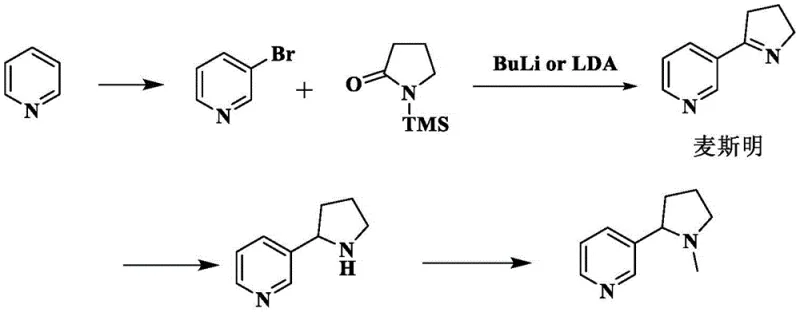 Furthermore, these synthetic routes frequently employ nitrogen protection strategies using trimethylsilyl groups, which adds unnecessary steps and material costs to the manufacturing process. Another prevalent method involves the chiral resolution of racemic nicotine to isolate the pharmacologically active levorotatory form, a process that inherently generates a massive amount of waste in the form of the unwanted dextrorotatory enantiomer salt. This "mother liquor" waste represents a significant loss of valuable chemical potential, traditionally accounting for nearly half of the input material being discarded or requiring costly disposal, thereby inflating the overall cost of goods sold (COGS) and creating substantial environmental burdens through three-waste generation.
Furthermore, these synthetic routes frequently employ nitrogen protection strategies using trimethylsilyl groups, which adds unnecessary steps and material costs to the manufacturing process. Another prevalent method involves the chiral resolution of racemic nicotine to isolate the pharmacologically active levorotatory form, a process that inherently generates a massive amount of waste in the form of the unwanted dextrorotatory enantiomer salt. This "mother liquor" waste represents a significant loss of valuable chemical potential, traditionally accounting for nearly half of the input material being discarded or requiring costly disposal, thereby inflating the overall cost of goods sold (COGS) and creating substantial environmental burdens through three-waste generation.
The Novel Approach
In a groundbreaking shift towards sustainable chemical manufacturing, Patent CN115246815A introduces a circular economy approach that transforms this liability into an asset by utilizing nicotine split waste solids as the primary raw material. Instead of discarding the dextronicotine salts remaining after chiral resolution, this novel method employs a strategic acid-base treatment followed by a thermal racemization reaction to regenerate high-purity racemic nicotine. 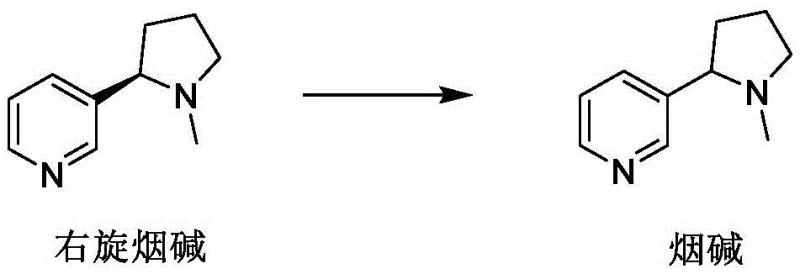 The core innovation lies in the efficient dissociation of the chiral acid (such as L-tartaric acid) from the waste solid, allowing for its recovery and reuse, while the nicotine base is liberated and subsequently subjected to racemization conditions. This process effectively closes the loop on chiral resolution waste, turning what was previously a disposal cost into a valuable feedstock for producing additional batches of racemic nicotine, which can then be re-resolved or used directly depending on market demand. By operating at moderate to high temperatures (50-300°C) with common alkali reagents, this method eliminates the need for cryogenic conditions and exotic catalysts, drastically simplifying the operational complexity and enhancing the safety profile for large-scale commercial production facilities.
The core innovation lies in the efficient dissociation of the chiral acid (such as L-tartaric acid) from the waste solid, allowing for its recovery and reuse, while the nicotine base is liberated and subsequently subjected to racemization conditions. This process effectively closes the loop on chiral resolution waste, turning what was previously a disposal cost into a valuable feedstock for producing additional batches of racemic nicotine, which can then be re-resolved or used directly depending on market demand. By operating at moderate to high temperatures (50-300°C) with common alkali reagents, this method eliminates the need for cryogenic conditions and exotic catalysts, drastically simplifying the operational complexity and enhancing the safety profile for large-scale commercial production facilities.
Mechanistic Insights into Alkali-Catalyzed Thermal Racemization
The chemical mechanism underpinning this invention is a robust base-catalyzed epimerization at the chiral center of the pyrrolidine ring, driven by thermodynamic equilibrium. The process begins with the treatment of the nicotine resolution waste solid (typically a dextronicotine tartrate salt) with a strong mineral acid such as hydrochloric acid or phosphoric acid. This acidification step protonates the carboxylate anions of the chiral resolving agent, causing the chiral acid to precipitate or become soluble in the aqueous phase depending on the specific acid used, thereby allowing for its physical separation via filtration. Once the chiral acid is removed, the remaining acidic aqueous phase contains the protonated nicotine cations, which are then liberated by adjusting the pH to a strongly basic range of 9 to 13 using reagents like sodium hydroxide or sodium ethoxide. This basification converts the nicotine salts into their free base form, which is then extracted into an organic solvent such as dichloromethane or ethyl acetate, concentrating the substrate for the critical racemization step.
The subsequent racemization reaction relies on the abstraction of the acidic proton at the C2 position of the pyrrolidine ring by a strong base, such as potassium tert-butoxide or sodium hydride, generating a planar carbanion or enamine-like intermediate. 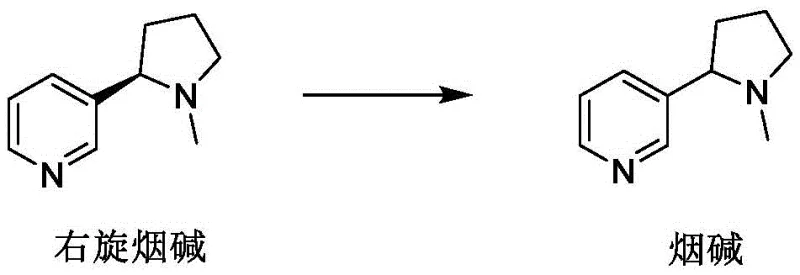 Upon heating the system to temperatures between 80°C and 200°C, this intermediate loses its stereochemical memory, and upon reprotonation, it forms a statistical mixture of both levorotatory and dextrorotatory enantiomers, effectively resetting the optical purity to a racemic state (0% ee). This thermal equilibration is highly efficient, with the patent reporting racemization yields ranging from 85% to 95%, indicating minimal degradation of the pyridine or pyrrolidine rings under these conditions. The final workup involves cooling the reaction mixture, phase separation with water, and vacuum distillation, which not only isolates the pure nicotine product with 99.8% chemical purity but also ensures that any residual base or solvent is removed, resulting in a pharmaceutical-grade intermediate suitable for downstream formulation or further chiral resolution.
Upon heating the system to temperatures between 80°C and 200°C, this intermediate loses its stereochemical memory, and upon reprotonation, it forms a statistical mixture of both levorotatory and dextrorotatory enantiomers, effectively resetting the optical purity to a racemic state (0% ee). This thermal equilibration is highly efficient, with the patent reporting racemization yields ranging from 85% to 95%, indicating minimal degradation of the pyridine or pyrrolidine rings under these conditions. The final workup involves cooling the reaction mixture, phase separation with water, and vacuum distillation, which not only isolates the pure nicotine product with 99.8% chemical purity but also ensures that any residual base or solvent is removed, resulting in a pharmaceutical-grade intermediate suitable for downstream formulation or further chiral resolution.
How to Synthesize Racemic Nicotine Efficiently
The synthesis of racemic nicotine from resolution waste represents a paradigm shift in process chemistry, moving from linear consumption to circular regeneration. This method allows manufacturers to maximize atom economy by recovering both the resolving agent and the substrate, significantly lowering the barrier to entry for high-volume production. The following guide outlines the standardized operational procedure derived from the patent examples, detailing the precise stoichiometry and conditions required to achieve optimal yields and purity.
- Treat nicotine resolution waste solids with mineral acid (e.g., HCl or H3PO4) to dissociate and recover the chiral acid (e.g., L-tartaric acid) via filtration.
- Adjust the pH of the acidic aqueous phase to 9-13 using alkali (e.g., NaOH) and extract the free base nicotine into an organic solvent.
- Subject the concentrated nicotine mother liquor to thermal racemization using a strong base (e.g., t-BuOK) at elevated temperatures (80-200°C) to obtain racemic nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this waste-recycling technology offers profound strategic advantages that extend far beyond simple chemical synthesis. By valorizing a waste stream that is abundantly available from existing chiral resolution operations, companies can drastically reduce their dependency on volatile agricultural markets for tobacco extraction or the complex global supply chains associated with total synthesis precursors. This decoupling from raw plant materials mitigates risks related to climate change, crop failures, and seasonal fluctuations, ensuring a consistent and reliable supply of high-purity nicotine intermediates year-round. Furthermore, the ability to recycle the chiral resolving acid (such as L-tartaric acid) means that one of the most significant consumable costs in chiral manufacturing is effectively neutralized, leading to substantial reductions in variable production costs without compromising on quality or throughput.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of raw material waste and the recovery of high-value reagents. By converting the dextronicotine byproduct—typically a disposal cost—into a saleable or reusable racemic product, the effective yield of the overall resolution process is nearly doubled. Additionally, the recovery of the chiral acid resolves a major cost center, as these specialized acids are often expensive and constitute a significant portion of the bill of materials. The use of commodity chemicals like potassium hydroxide and hydrochloric acid for the transformation further ensures that reagent costs remain low and predictable, avoiding the price volatility associated with specialized catalysts or organometallic reagents used in alternative synthetic routes.
- Enhanced Supply Chain Reliability: Implementing this technology enhances supply chain resilience by creating an internal loop for material flow that is less susceptible to external disruptions. Since the feedstock is a byproduct of the company's own resolution processes or can be sourced from partners performing similar separations, the lead time for acquiring raw materials is significantly reduced compared to importing botanical extracts or synthesizing complex heterocycles from scratch. This localized sourcing capability allows for more agile production scheduling and inventory management, enabling manufacturers to respond rapidly to spikes in demand for smoking cessation therapies or vaping products without the long lag times inherent in agricultural supply chains.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a clear pathway to greener manufacturing credentials, which is increasingly critical for maintaining social licenses to operate and meeting corporate sustainability goals. The patent explicitly notes a reduction in three-waste generation by more than 50%, a qualitative improvement that translates to lower wastewater treatment costs and reduced hazardous waste disposal fees. The process avoids the use of heavy metals or toxic organolithium reagents, simplifying the effluent profile and making it easier to comply with stringent environmental regulations in major pharmaceutical markets. This cleaner production profile not only reduces operational risk but also enhances the brand value of the final product as a sustainably manufactured pharmaceutical ingredient.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nicotine racemization technology. These answers are derived directly from the experimental data and claims presented in Patent CN115246815A, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is crucial for R&D teams assessing process transfer and procurement teams evaluating cost structures.
Q: What is the primary advantage of this nicotine preparation method?
A: The primary advantage is the conversion of chiral resolution waste solids (specifically dextronicotine salts) back into usable racemic nicotine, significantly reducing raw material costs and chemical waste generation compared to traditional disposal or total synthesis methods.
Q: What is the chemical purity of the nicotine produced via this patent?
A: According to the patent data, the racemization process yields nicotine with a chemical purity of up to 99.8%, meeting stringent requirements for pharmaceutical intermediates and smoking cessation products.
Q: Which bases are suitable for the racemization step?
A: The patent specifies several effective bases for the racemization reaction, including potassium hydroxide, sodium hydroxide, potassium tert-butoxide, sodium methoxide, and sodium hydride, allowing flexibility based on available industrial reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the future of fine chemical manufacturing lies in sustainable, efficient, and scalable processes like the one described in this patent analysis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries such as thermal racemization and chiral resolution are executed with precision and safety. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the 99.8% chemical purity benchmark achieved in this novel nicotine synthesis route, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how integrating waste-recycling protocols can optimize your bottom line. Contact us today to discuss route feasibility assessments and to obtain specific COA data for our high-purity nicotine intermediates, securing a competitive edge in the rapidly evolving pharmaceutical and consumer health markets.
