Advanced Rhodium-Catalyzed Synthesis of Functionalized Pyrrole Ethylene Derivatives for Pharmaceutical Applications
The landscape of organic synthesis for nitrogen-containing heterocycles is continuously evolving, driven by the demand for more efficient and sustainable manufacturing processes in the pharmaceutical and material science sectors. Patent CN113045549A introduces a significant advancement in this field by disclosing a novel preparation method for pyrrole ethylene derivatives. These compounds are pivotal structural motifs found in various bioactive molecules, including non-steroidal anti-inflammatory drugs like Tolmetin, and functional materials such as conductive polypyrroles. The disclosed technology leverages a transition metal-catalyzed direct C-H olefination strategy, offering a streamlined pathway to access structurally diverse pyrrole derivatives that were previously challenging to synthesize with high efficiency. This innovation addresses critical bottlenecks in traditional synthetic routes, providing a robust platform for the development of next-generation therapeutic agents and advanced electronic materials.
Historically, the functionalization of pyrrole rings relied heavily on classical electrophilic substitution reactions, such as nitration, sulfonation, or Friedel-Crafts acylation. While these methods are well-established, they often suffer from significant limitations, including poor regioselectivity, harsh reaction conditions requiring strong acids or Lewis acids, and the generation of substantial hazardous waste. Furthermore, constructing specific vinyl-substituted pyrrole scaffolds typically necessitates multi-step sequences involving pre-halogenation followed by palladium-catalyzed cross-coupling, which increases both the cost and the environmental footprint of the process. In contrast, the novel approach detailed in the patent utilizes a rhodium-catalyzed system that enables the direct coupling of pyrrole substrates with alkenyl boronates. This strategy bypasses the need for pre-functionalized halogenated intermediates, thereby simplifying the synthetic route and enhancing the overall atom economy of the transformation.
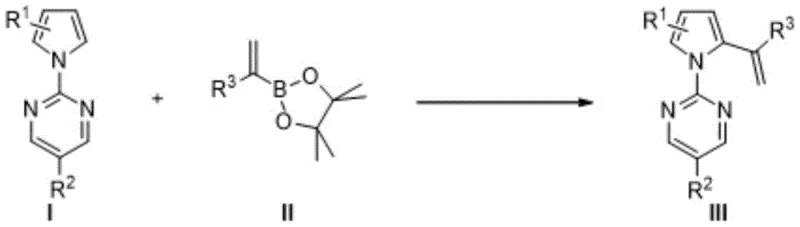
The mechanistic underpinnings of this Rhodium(III)-catalyzed C-H activation process represent a sophisticated interplay between organometallic chemistry and substrate design. The reaction initiates with the coordination of the cationic Rhodium species to the nitrogen atoms of the pyrimidine directing group attached to the pyrrole ring. This coordination facilitates the cleavage of the proximal C-H bond on the pyrrole ring through a concerted metalation-deprotonation (CMD) pathway, forming a stable five-membered rhodacycle intermediate. Subsequent transmetallation with the alkenyl boronate reagent, activated by the silver salt additive, transfers the vinyl group to the metal center. The catalytic cycle concludes with a reductive elimination step that releases the desired pyrrole ethylene derivative and regenerates the active Rhodium catalyst. This precise mechanistic control ensures that substitution occurs exclusively at the desired position, minimizing the formation of regioisomeric impurities that are difficult to separate and can compromise the quality of the final pharmaceutical intermediate.
Implementing this synthesis protocol requires careful attention to reaction parameters to maximize yield and purity. The process involves reacting the pyrrole substrate with an excess of the alkenyl boronate reagent in a polar protic solvent, typically methanol, which aids in solubilizing the inorganic additives and stabilizing the catalytic species. The use of silver acetate as an oxidant and halide scavenger is critical for maintaining the catalytic turnover. Following the reaction period, the workup procedure is straightforward, involving concentration and purification via standard silica gel chromatography. For detailed operational parameters and specific stoichiometric ratios optimized for different substrate classes, please refer to the standardized synthesis guide below.
From a commercial procurement and supply chain perspective, the adoption of this Rh-catalyzed C-H activation technology offers compelling advantages over legacy manufacturing methods. The elimination of pre-halogenation steps significantly reduces the raw material costs and the number of unit operations required, leading to a more cost-effective production model. By utilizing commercially available alkenyl boronates and avoiding expensive palladium catalysts often associated with traditional cross-coupling, manufacturers can achieve substantial cost reduction in pharmaceutical intermediates manufacturing without compromising on quality. Furthermore, the mild reaction conditions (60°C) reduce energy consumption compared to high-temperature processes, aligning with modern green chemistry principles and lowering the overall carbon footprint of the supply chain.
- Cost Reduction in Manufacturing: The direct C-H functionalization strategy eliminates the need for synthesizing and handling hazardous halogenated precursors, which are often costly and require specialized disposal protocols. This streamlining of the synthetic route reduces the consumption of reagents and solvents, directly translating to lower variable costs per kilogram of product. Additionally, the high atom economy of the reaction minimizes waste generation, reducing the burden and expense associated with waste treatment and environmental compliance.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially abundant starting materials, such as substituted pyrroles and pinacol boronates, mitigates the risk of supply disruptions often associated with custom-synthesized specialty reagents. The robustness of the catalytic system allows for consistent batch-to-batch reproducibility, ensuring a steady flow of high-purity intermediates to downstream customers. This reliability is crucial for maintaining uninterrupted production schedules in the fast-paced pharmaceutical industry where delays can have significant financial implications.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from laboratory to pilot and commercial production scales. The use of methanol as a primary solvent simplifies solvent recovery and recycling processes. Moreover, the reduction in heavy metal waste and hazardous by-products facilitates easier adherence to stringent environmental regulations, positioning this method as a sustainable choice for long-term manufacturing partnerships.
Understanding the technical nuances and commercial implications of this synthesis method is essential for stakeholders involved in the sourcing and development of complex organic intermediates. The following frequently asked questions address common inquiries regarding the practical application, scalability, and quality control aspects of this technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what partners can expect when integrating this methodology into their supply chains.
- Prepare the reaction mixture by combining the pyrrole substrate (Compound I), alkenyl boronate (Compound II), AgOAc, and [RhCp*Cl2]2 catalyst in methanol solvent.
- Heat the reaction mixture to 60°C and maintain stirring for 24 hours to ensure complete conversion monitored by TLC.
- Cool the reaction to room temperature, concentrate under reduced pressure, and purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate.
Frequently Asked Questions (FAQ)
Q: What are the key advantages of this Rh-catalyzed method over traditional electrophilic substitution?
A: This method utilizes direct C-H activation, eliminating the need for pre-functionalized halogenated precursors required in traditional cross-coupling. It offers superior atom economy, milder reaction conditions (60°C), and excellent regioselectivity directed by the pyrimidine moiety, resulting in fewer by-products and simplified purification.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process employs commercially available reagents and standard organic solvents like methanol. The moderate temperature (60°C) and ambient pressure conditions make it highly amenable to scale-up, reducing energy consumption and safety risks associated with high-pressure or cryogenic processes often found in alternative synthetic routes.
Q: What is the role of the pyrimidine ring in the reaction mechanism?
A: The pyrimidine ring attached to the pyrrole nitrogen acts as a crucial directing group. It coordinates with the Rhodium catalyst to facilitate selective C-H bond activation at the adjacent position on the pyrrole ring, ensuring high regioselectivity and preventing random substitution patterns that complicate downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrole Ethylene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the Rh-catalyzed C-H activation described in CN113045549A can be successfully translated into robust industrial manufacturing. We are committed to delivering high-purity pyrrole ethylene derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite pharmaceutical and chemical companies to collaborate with us to leverage this cutting-edge technology for their specific project needs. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce time-to-market for your critical projects.
