Advanced Enzymatic Resolution for High-Purity Beta-Thioamino Acid Derivatives and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to synthesize optically active amino acids, particularly those with functional side chains essential for advanced peptide therapeutics. Patent CN105392891B introduces a groundbreaking methodology for producing D-or L-amino acid derivatives possessing a thiol group at the beta-position with exceptional yield and simplicity. This technology addresses the critical bottleneck of stereoisomer control during chemical synthesis by leveraging a unique enzymatic resolution strategy. Instead of relying on expensive chiral starting materials that limit scalability, this process allows for the use of racemic mixtures, which are subsequently resolved using highly selective hydrolases. For R&D Directors and Procurement Managers, this represents a significant shift towards more cost-effective and robust supply chains for complex peptide building blocks. The ability to produce both D-form and L-form derivatives from a common intermediate streamlines inventory management and reduces the complexity of sourcing rare chiral precursors. Furthermore, the mild reaction conditions preserve the integrity of sensitive functional groups, ensuring high purity profiles required for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing optically active amino acids with thiol groups often suffer from severe limitations regarding yield and operational complexity. Conventional routes typically require the use of optically pure starting materials to prevent racemization, which drastically increases raw material costs and limits availability. Moreover, the introduction of a thiol group at the beta-position usually necessitates harsh chemical conditions, such as strong acids or bases, which can compromise the stereochemical integrity of the alpha-carbon atom. This risk of isomerization forces manufacturers to operate under restricted temperature and pH ranges, leading to slower reaction rates and lower throughput. Additionally, the purification of intermediates in these traditional pathways is often cumbersome, requiring multiple chromatography steps that reduce overall mass balance. For Supply Chain Heads, these inefficiencies translate into longer lead times and higher vulnerability to supply disruptions. The reliance on specific chiral pools also creates a dependency on limited suppliers, posing a strategic risk for long-term project planning. Consequently, the industry has long needed a method that decouples the introduction of the functional group from the maintenance of chirality.
The Novel Approach
The innovative process described in the patent overcomes these hurdles by intentionally producing a racemic intermediate composition containing both D-and L-forms of the amino acid derivative with a thiol group at the beta-position. This strategic pivot allows chemists to utilize robust chemical reactions that would otherwise be forbidden due to racemization risks, as the stereochemistry is resolved at a later stage. By employing a D-or L-amino acid-selective hydrolase, specifically an aminoacylase, the process achieves high stereoselectivity under mild aqueous conditions. This enzymatic step selectively hydrolyzes one enantiomer, creating a distinct difference in hydrophobicity between the hydrolyzed product and the unreacted substrate. This physical property difference facilitates easy separation via extraction or crystallization, significantly simplifying downstream processing. 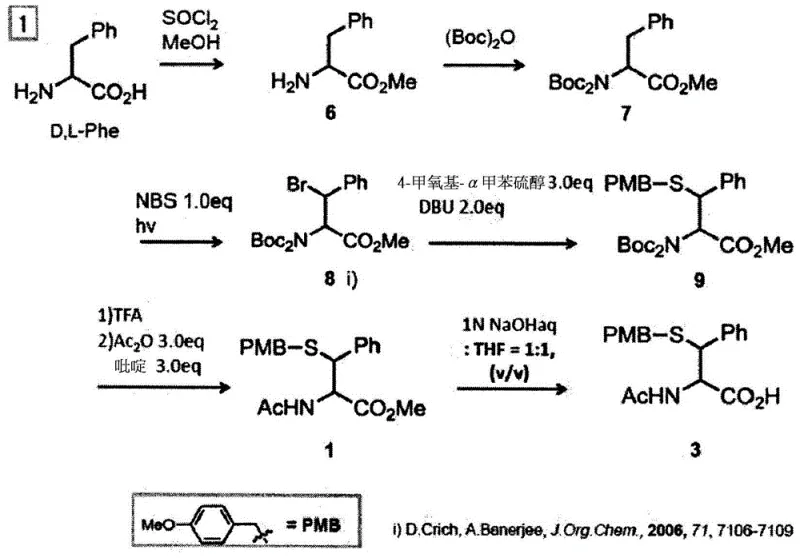 The ability to start from readily available racemic aromatic amino acids or glycine further enhances the economic viability of this route. It eliminates the need for costly chiral catalysts or resolving agents in the early stages, thereby reducing the overall cost of goods sold. This approach not only improves yield but also aligns with green chemistry principles by reducing solvent usage and waste generation associated with repetitive purification cycles.
The ability to start from readily available racemic aromatic amino acids or glycine further enhances the economic viability of this route. It eliminates the need for costly chiral catalysts or resolving agents in the early stages, thereby reducing the overall cost of goods sold. This approach not only improves yield but also aligns with green chemistry principles by reducing solvent usage and waste generation associated with repetitive purification cycles.
Mechanistic Insights into Enzymatic Resolution of Beta-Thioamino Acids
The core of this technology lies in the precise molecular recognition capabilities of the selected aminoacylase enzymes. The intermediate substrate is designed with an acylated amino group at the alpha-position, making it a specific target for the hydrolase. The enzyme distinguishes between the D-and L-configurations at the alpha-carbon atom, catalyzing the hydrolysis of the amide bond for only one stereoisomer. This reaction converts the acylamino group into a free amino group, fundamentally altering the polarity and solubility of that specific enantiomer. 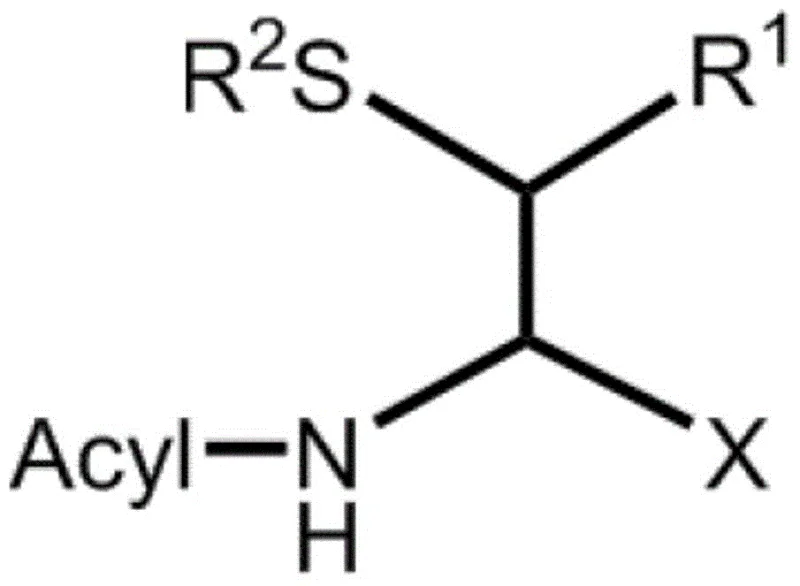 The presence of the thiol group at the beta-position, whether protected or unprotected, does not hinder the enzymatic activity, demonstrating the broad substrate tolerance of the system. Protecting groups such as PMB or Acm can be utilized to prevent side reactions during the chemical synthesis phase, ensuring that the thiol functionality remains intact until the final deprotection step. The mechanistic pathway ensures that the stereochemical information at the beta-carbon does not interfere with the resolution at the alpha-carbon, allowing for the production of diastereomerically pure compounds if needed. This level of control is paramount for R&D teams developing complex peptide drugs where even minor impurities can affect biological activity or regulatory approval.
The presence of the thiol group at the beta-position, whether protected or unprotected, does not hinder the enzymatic activity, demonstrating the broad substrate tolerance of the system. Protecting groups such as PMB or Acm can be utilized to prevent side reactions during the chemical synthesis phase, ensuring that the thiol functionality remains intact until the final deprotection step. The mechanistic pathway ensures that the stereochemical information at the beta-carbon does not interfere with the resolution at the alpha-carbon, allowing for the production of diastereomerically pure compounds if needed. This level of control is paramount for R&D teams developing complex peptide drugs where even minor impurities can affect biological activity or regulatory approval.
Impurity control is inherently built into this process through the specificity of the enzymatic reaction. Unlike chemical resolution methods that might co-crystallize impurities, the enzyme acts as a biological filter, ignoring structurally similar byproducts that do not fit the active site. Following hydrolysis, the separation is driven by the introduction of fat-soluble protecting groups, such as Boc or Fmoc, onto the newly freed amino group. This step amplifies the hydrophobicity difference between the desired product and the unreacted enantiomer, enabling high-efficiency separation via silica gel column chromatography or liquid-liquid extraction. 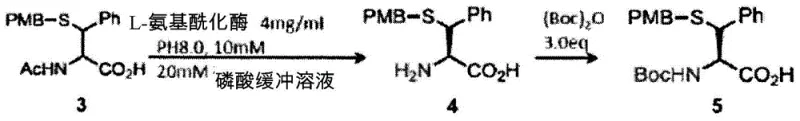 The result is a high-purity optically active amino acid derivative suitable for direct use in Native Chemical Ligation (NCL). This mechanism supports the synthesis of both D-proteins and L-proteins, expanding the toolkit available for structural biology and therapeutic development. The robustness of the enzymatic step ensures consistent quality across batches, a critical factor for commercial scale-up and regulatory compliance.
The result is a high-purity optically active amino acid derivative suitable for direct use in Native Chemical Ligation (NCL). This mechanism supports the synthesis of both D-proteins and L-proteins, expanding the toolkit available for structural biology and therapeutic development. The robustness of the enzymatic step ensures consistent quality across batches, a critical factor for commercial scale-up and regulatory compliance.
How to Synthesize Beta-Thioamino Acid Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical sequence designed to maximize yield while minimizing operational risks. The process begins with the preparation of a racemic amino acid derivative, where a thiol group is introduced at the beta-position using standard organic transformations such as nucleophilic substitution or condensation reactions. Subsequently, the amino group is acylated to create the necessary substrate for the enzymatic resolution. The critical step involves the incubation of this intermediate with a specific aminoacylase in a buffered aqueous solution at controlled pH and temperature. Detailed standardized synthesis steps see the guide below.
- Produce a racemic amino acid derivative containing a protected or unprotected thiol group at the beta-position via chemical synthesis.
- Convert the amino or carboxyl group at the alpha-carbon into a substrate suitable for D-or L-amino acid-selective hydrolase.
- Perform enzymatic hydrolysis using a selective aminoacylase to separate the desired D-or L-enantiomer based on hydrophobicity differences.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this enzymatic resolution technology offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply base. By enabling the use of racemic starting materials, manufacturers are no longer constrained by the availability and pricing volatility of expensive chiral pools. This flexibility allows for dynamic sourcing strategies that can adapt to market fluctuations without compromising production schedules. Furthermore, the mild conditions of the enzymatic step reduce energy consumption and equipment wear, contributing to lower operational expenditures. The high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, reducing the burden on quality control laboratories and accelerating release times. These factors collectively enhance the reliability of the supply chain, ensuring consistent delivery of high-quality intermediates to downstream customers.
- Cost Reduction in Manufacturing: The elimination of chiral starting materials represents a fundamental shift in cost structure for amino acid manufacturing. Traditional methods often incur high costs due to the low atom economy of chiral resolution or the premium pricing of enantiopure reagents. By resolving the mixture enzymatically at a later stage, the process maximizes the utility of every gram of raw material purchased. Additionally, the ability to recover and recycle the unreacted enantiomer further improves the overall mass balance, effectively doubling the potential yield from a single batch of racemic precursor. This efficiency translates into substantial cost savings that can be passed down the value chain, making advanced peptide therapeutics more economically viable. The reduction in solvent usage and purification steps also lowers waste disposal costs, aligning with sustainability goals.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for specialized chiral chemicals poses a significant risk to production continuity. This technology mitigates that risk by allowing the use of commodity-grade racemic amino acids, which are widely available from multiple global vendors. The robustness of the enzymatic process ensures that production is less susceptible to variations in raw material quality, providing a stable foundation for long-term supply agreements. Moreover, the scalability of the enzymatic reaction means that production capacity can be increased rapidly to meet surges in demand without the need for extensive capital investment in new reactor types. This agility is crucial for responding to the fast-paced requirements of the pharmaceutical industry, where timelines for drug development are constantly compressing.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and mixing, particularly for exothermic reactions. The enzymatic nature of the resolution step occurs under mild physiological conditions, eliminating the safety hazards associated with high-pressure or high-temperature operations. This makes the transition from laboratory bench to pilot plant and finally to commercial production much smoother and predictable. From an environmental perspective, the use of biocatalysts reduces the need for heavy metal catalysts and toxic solvents, simplifying wastewater treatment and regulatory reporting. The process generates less hazardous waste, facilitating compliance with increasingly stringent environmental regulations across different jurisdictions. This eco-friendly profile enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of beta-thioamino acid derivatives using this patented enzymatic resolution method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the scope and applicability of the technology. Understanding these details helps stakeholders evaluate the feasibility of integrating these intermediates into their existing development pipelines. The answers reflect the current state of the art as described in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: How does this process improve yield compared to traditional chiral synthesis?
A: By utilizing racemic starting materials and resolving them enzymatically, the process avoids the yield losses associated with maintaining chirality during harsh chemical steps, allowing for higher overall efficiency.
Q: What types of amino acids can be produced using this method?
A: The method is versatile and supports the production of various natural and unnatural amino acids with thiol groups at the beta-position, including phenylalanine and tyrosine derivatives.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of mild enzymatic conditions and robust chemical intermediates makes the process highly scalable and compliant with environmental standards for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Thioamino Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic resolution technology for the next generation of peptide-based therapeutics. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to market. Our facilities are equipped with state-of-the-art fermentation and purification units capable of handling sensitive enzymatic reactions with precision. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of beta-thioamino acid derivative meets the highest international standards. Our team of experts is dedicated to optimizing the reaction parameters to maximize yield and minimize impurities, ensuring a reliable supply for your critical projects.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a secure, scalable, and cost-effective supply chain for high-purity amino acid intermediates, empowering your R&D efforts to move faster and more efficiently.
