Advanced Electrochemical Synthesis of p-Methylsulfonylbenzoic Acid for Global Pharma Supply Chains
Advanced Electrochemical Synthesis of p-Methylsulfonylbenzoic Acid for Global Pharma Supply Chains
The global demand for high-purity aromatic sulfone intermediates continues to surge, driven by the expanding pipelines of novel herbicides and antibacterial agents. In this context, the technological breakthrough detailed in patent CN103319383A represents a paradigm shift in the manufacturing of p-methylsulfonylbenzoic acid. Traditionally, the industry has relied on harsh nitric acid oxidation, a method plagued by safety hazards and environmental non-compliance. This new methodology introduces a sophisticated electrochemical regeneration loop that not only enhances selectivity but also aligns with the rigorous green chemistry standards demanded by modern regulatory bodies. By integrating selective chemical oxidation with in-situ oxidant recycling, this process offers a robust pathway for producing high-purity intermediates essential for complex API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-methylsulfonylbenzoic acid has been dominated by nitric acid oxidation techniques, which present severe operational and environmental challenges for large-scale manufacturers. The reaction degree in nitric acid systems is notoriously difficult to control, often leading to unpredictable selectivity, low conversion rates, and inconsistent yields that disrupt supply chain planning. Furthermore, the reaction mixture becomes exceedingly complex, generating a multitude of nitro-by-products and tarry residues that make downstream separation and refining an arduous, cost-intensive task. From a safety perspective, the use of concentrated nitric acid introduces significant risks of thermal runaway and the release of toxic nitrogen oxide gases, creating substantial liability and requiring expensive scrubbing infrastructure that erodes profit margins in competitive markets.
The Novel Approach
In stark contrast, the innovative process outlined in the patent utilizes a controlled liquid-liquid heterogeneous oxidation system powered by potassium dichromate in an acidic medium. This approach fundamentally alters the reaction landscape by enabling precise modulation of oxidizing power through temperature and acidity adjustments, ensuring high selectivity for the target carboxylic acid over partial oxidation products. A defining feature of this technology is the integration of an electrochemical regeneration unit that converts the reduced chromium species back into the active oxidant, effectively closing the material loop and drastically reducing reagent consumption. This closed-loop design not only mitigates the environmental burden of heavy metal waste but also simplifies the purification workflow, resulting in a cleaner product profile that requires less aggressive downstream processing to meet stringent pharmaceutical specifications.
Mechanistic Insights into Dichromate-Mediated Selective Oxidation
The core of this synthesis lies in the selective oxidation of the methyl group on the p-methylsulfonyltoluene ring to a carboxylic acid functionality using hexavalent chromium. The reaction proceeds through a liquid-liquid heterogeneous mechanism where molten organic substrate interacts with the aqueous acidic dichromate phase. As the oxidation progresses, the Cr(VI) species acts as the electron acceptor, systematically oxidizing the methyl group first to an aldehyde intermediate and finally to the carboxylic acid, while itself being reduced to trivalent chromium (Cr(III)). The thermodynamic conditions are carefully tuned such that the product, p-methylsulfonylbenzoic acid, along with the intermediate aldehyde, crystallizes out of the reaction matrix as it forms, driving the equilibrium forward and preventing over-oxidation or ring degradation that typically plagues homogeneous oxidation systems.
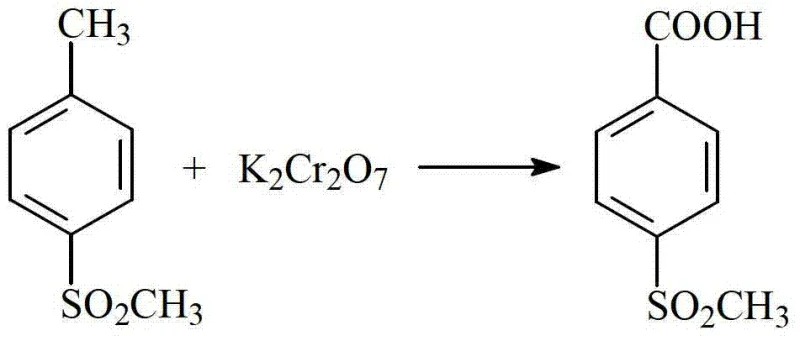
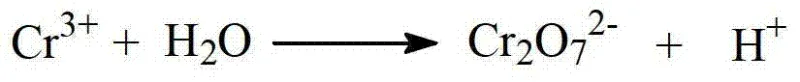
Following the oxidation phase, the process employs a clever separation strategy based on solubility differences to isolate the product from unreacted starting materials. The crude solid mixture is treated with a neutralizing agent, converting the acidic product into a water-soluble salt while leaving the neutral unreacted toluene derivative and aldehyde impurities in the solid phase for recycling. This salt solution is then subjected to adsorption purification to remove trace colored impurities and residual organics before being re-acidified to precipitate the final high-purity acid. Crucially, the spent oxidant stream containing Cr(III) is not discarded; instead, it undergoes electrochemical oxidation at the anode to regenerate Cr(VI), which is then fed back into the preheating stage, creating a sustainable catalytic cycle that minimizes raw material costs and waste generation.
How to Synthesize p-Methylsulfonylbenzoic Acid Efficiently
The synthesis protocol described in the patent provides a comprehensive roadmap for transitioning from batch nitric acid methods to this continuous-flow compatible electrochemical process. It involves a sequence of unit operations including melting, preheating, heterogeneous reaction, solid-liquid separation, neutralization, adsorption, and electrochemical regeneration. Each step is optimized to maximize yield and purity while ensuring operator safety and environmental compliance. For R&D teams looking to implement this technology, understanding the interplay between the electrochemical current density and the chemical oxidation rate is vital for scaling. The detailed standardized synthesis steps see the guide below.
- Melt p-methylsulfonyltoluene at 90°C-150°C and preheat a sulfuric acid solution containing potassium dichromate to 90°C-120°C.
- Perform liquid-liquid heterogeneous oxidation where Cr(VI) selectively oxidizes the methyl group, precipitating the product while reducing to Cr(III).
- Separate the solid product, neutralize to form soluble salts for purification, then acidify to crystallize high-purity p-methylsulfonylbenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical oxidation technology translates into tangible strategic advantages regarding cost stability and supply continuity. By eliminating the reliance on stoichiometric quantities of nitric acid and the associated waste treatment costs, the overall manufacturing cost structure is significantly optimized. The ability to recycle the expensive chromium oxidant internally reduces exposure to volatile raw material pricing, providing a more predictable cost base for long-term contracts. Furthermore, the simplified purification train reduces the need for extensive solvent usage and energy-intensive distillation steps, contributing to substantial operational expenditure savings that can be passed down the supply chain.
- Cost Reduction in Manufacturing: The implementation of the electrochemical regeneration loop fundamentally changes the cost dynamics of oxidant consumption. Instead of purchasing fresh oxidant for every batch, the system regenerates the active species in situ, leading to a drastic reduction in raw material procurement costs. Additionally, the recovery and recycling of unreacted p-methylsulfonyltoluene and intermediate aldehydes ensure that atom economy is maximized, meaning less feedstock is wasted as by-product. This efficiency gain allows for a more competitive pricing model without compromising on the quality of the final intermediate, offering significant value to cost-sensitive agrochemical and pharmaceutical manufacturers.
- Enhanced Supply Chain Reliability: Safety and regulatory compliance are critical bottlenecks in the supply of hazardous intermediates. By replacing the dangerous nitric acid oxidation process with a safer, controlled dichromate system, the risk of production shutdowns due to safety incidents or environmental violations is markedly reduced. The process operates under milder conditions compared to high-pressure nitration, enhancing equipment longevity and reducing maintenance downtime. This reliability ensures a consistent flow of high-purity p-methylsulfonylbenzoic acid, securing the production schedules of downstream API manufacturers who depend on just-in-time delivery for their own formulation lines.
- Scalability and Environmental Compliance: The modular nature of the electrochemical reactors and solid-liquid separation units facilitates easy scale-up from pilot to commercial production volumes. The process generates significantly less hazardous waste, particularly nitrogen oxides and acidic wastewater, simplifying the permitting process and reducing the burden on effluent treatment plants. This alignment with green chemistry principles future-proofs the supply chain against tightening environmental regulations, ensuring that the manufacturing site remains operational and compliant in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of p-methylsulfonylbenzoic acid using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your specific synthesis pathways.
Q: How does the electrochemical regeneration step improve sustainability?
A: The process electrochemically oxidizes spent Cr(III) back to Cr(VI), allowing the expensive oxidant to be recycled continuously, significantly reducing heavy metal waste discharge compared to traditional stoichiometric oxidation.
Q: What are the purity advantages over nitric acid oxidation?
A: Unlike nitric acid oxidation which produces complex by-products and nitrogen oxides, this selective dichromate method combined with adsorption purification yields a cleaner impurity profile suitable for sensitive pharmaceutical applications.
Q: Can unreacted raw materials be recovered in this process?
A: Yes, the solid-liquid separation after neutralization isolates unreacted p-methylsulfonyltoluene and intermediate aldehydes in the solid phase, which are returned to the melting stage for recycling, maximizing atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Methylsulfonylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the long-term viability of the fine chemical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this electrochemical process are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of p-methylsulfonylbenzoic acid meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and high-quality supply of this critical intermediate.
