Scalable Synthesis of 4-(4-Amino-3-Fluorophenoxy)-N-Methylpyridine-2-Carboxamide for Oncology Applications
The pharmaceutical landscape for oncology treatments continues to evolve, driven by the demand for robust and scalable supply chains for critical active pharmaceutical ingredients. Central to this ecosystem is the production of high-quality intermediates, such as 4-(4-amino-3-fluorophenoxy)-N-methylpyridine-2-carboxamide, a pivotal precursor in the synthesis of Regorafenib. Recent technological advancements, specifically detailed in patent CN105777625B, have introduced a transformative approach to manufacturing this compound. This innovation addresses long-standing inefficiencies in traditional synthetic routes by substituting hazardous organic bases with stable inorganic alternatives. For global procurement leaders and R&D directors, understanding this shift is crucial, as it represents a move towards safer, more cost-effective, and environmentally compliant manufacturing processes. By leveraging this patented methodology, stakeholders can secure a reliable pharmaceutical intermediates supplier partnership that prioritizes both chemical integrity and operational safety, ensuring a continuous flow of materials for life-saving therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key pyridine derivative relied heavily on the use of potassium tert-butoxide as the primary base catalyst. While chemically effective on a small laboratory scale, this reagent presents severe challenges when translated to industrial manufacturing environments. The inherent instability of potassium tert-butoxide introduces significant safety hazards, including the potential for exothermic runaways and explosion risks during large-scale handling. Furthermore, the downstream processing associated with these legacy methods is notoriously cumbersome, often requiring multiple steps of extraction, solvent concentration, and column chromatography to achieve acceptable purity levels. These additional unit operations not only inflate production costs through increased solvent usage and energy consumption but also generate substantial volumes of hazardous waste. The complexity of separating the product from the reaction matrix often leads to yield losses, with historical data indicating maximum efficiencies hovering around 77%, thereby constraining the overall economic viability of the supply chain.
The Novel Approach
In stark contrast to these legacy constraints, the innovative process outlined in the referenced patent utilizes inorganic bases, such as sodium hydroxide or potassium hydroxide, to drive the nucleophilic substitution reaction. This fundamental change in reagent selection eliminates the safety risks associated with volatile organic alkalis, creating a much more stable reaction environment suitable for tonnage-scale production. Moreover, the purification strategy has been radically simplified; instead of complex chromatographic separations, the product is isolated through a straightforward crystallization process induced by the addition of water. This modification not only streamlines the workflow but also significantly enhances the recovery rate of the final product. By removing the need for expensive purification columns and reducing solvent load, the new method offers a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing, allowing producers to deliver high-purity materials with greater operational efficiency and reduced environmental impact.
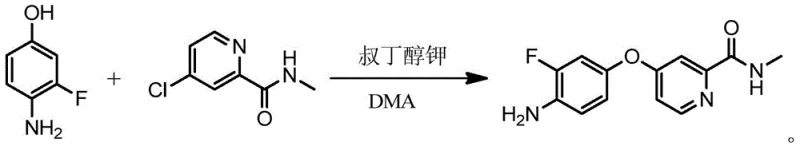
Mechanistic Insights into Inorganic Base Catalyzed Nucleophilic Substitution
The core of this technological breakthrough lies in the mechanistic efficiency of using inorganic hydroxides to facilitate the coupling of the pyridine and phenol moieties. In this reaction system, the inorganic base acts as a potent deprotonating agent, activating the phenolic hydroxyl group of 4-amino-3-fluorophenol to form a highly reactive phenoxide species. This nucleophile then attacks the electron-deficient carbon at the 4-position of the 4-chloro-N-methylpyridine-2-carboxamide ring, displacing the chloride leaving group through a classic nucleophilic aromatic substitution mechanism. The choice of solvent, such as N,N-dimethylacetamide (DMA), plays a critical role in stabilizing the transition state and ensuring the solubility of the inorganic salts throughout the reaction course. Unlike organic bases that may introduce steric hindrance or side reactions, the small ionic radius of sodium or potassium ions allows for tight ion pairing that enhances nucleophilicity without compromising the structural integrity of the sensitive amino and fluoro substituents on the aromatic ring.
Furthermore, the control of impurities is intrinsically linked to the simplicity of the inorganic base system. In traditional organic base routes, side reactions such as over-alkylation or decomposition of the base itself can lead to complex impurity profiles that are difficult to remove. The inorganic method minimizes these pathways by maintaining a cleaner reaction profile where the primary byproduct is simply an inorganic salt, which is easily removed during the aqueous workup. The crystallization step serves as a powerful purification tool, leveraging the differential solubility of the target molecule versus residual starting materials and byproducts in the water-solvent mixture. This ensures that the final high-purity pharmaceutical intermediates meet stringent quality specifications required for downstream drug synthesis, with HPLC purity levels consistently exceeding 98% without the need for further chromatographic refinement.
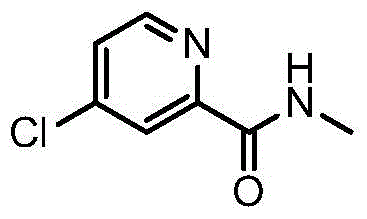
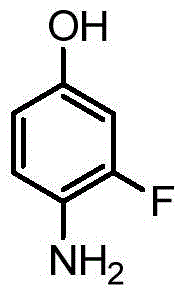
How to Synthesize 4-(4-Amino-3-Fluorophenoxy)-N-Methylpyridine-2-Carboxamide Efficiently
Implementing this optimized synthetic route requires precise control over reaction parameters to maximize yield and safety. The process begins with the dissolution of the reactants in a polar aprotic solvent under an inert atmosphere, followed by the controlled addition of the inorganic base. Temperature management is critical, with the reaction typically maintained between 80°C and 140°C to ensure complete conversion while preventing thermal degradation. Once the reaction is complete, the addition of water triggers the precipitation of the product, which is then filtered and dried. This streamlined approach eliminates the need for complex equipment setups associated with distillation or chromatography.
- React 4-chloro-N-methylpyridine-2-formamide with 4-amino-3-fluorophenol using an inorganic base like sodium hydroxide in an organic solvent.
- Maintain reaction temperature between 80-140°C under inert gas protection to ensure complete conversion.
- Add water to the reaction mixture and cool to induce crystallization, filtering the solid product to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this inorganic base methodology translates into tangible strategic advantages beyond mere chemical efficiency. The elimination of hazardous organic bases like potassium tert-butoxide significantly reduces the regulatory burden and insurance costs associated with storing and handling dangerous goods. This shift enhances the overall resilience of the supply chain by minimizing the risk of production stoppages due to safety incidents or regulatory compliance issues. Additionally, the simplified purification process reduces the dependency on specialized chromatography resins and large volumes of organic solvents, which are subject to market volatility and supply constraints. By adopting a process that relies on commodity chemicals like sodium hydroxide and water for isolation, manufacturers can achieve a more predictable and stable cost structure, ensuring long-term availability of this critical oncology intermediate.
- Cost Reduction in Manufacturing: The transition to inorganic bases and crystallization-based purification fundamentally alters the cost dynamics of production. By removing the requirement for expensive organic bases and eliminating energy-intensive concentration and column chromatography steps, the overall operational expenditure is drastically lowered. The reduction in solvent usage directly correlates to lower waste disposal costs and reduced raw material procurement expenses. Furthermore, the higher yield achieved through this method means that less starting material is required to produce the same amount of final product, effectively amplifying the cost savings across the entire production batch. This economic efficiency allows for more competitive pricing models without compromising on the quality or purity of the supplied intermediates.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical cancer drug intermediates is paramount, and this process enhances stability by utilizing widely available and non-hazardous reagents. The reliance on common inorganic chemicals mitigates the risk of supply disruptions that often plague specialty organic reagents. The robustness of the reaction conditions also allows for greater flexibility in manufacturing scheduling, as the process is less sensitive to minor variations in environmental conditions compared to moisture-sensitive organic base reactions. This stability ensures that production timelines are met consistently, reducing lead time for high-purity pharmaceutical intermediates and providing downstream drug manufacturers with the confidence of a secure and uninterrupted material flow.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden bottlenecks, particularly regarding heat transfer and waste management. This novel method is inherently designed for scalability, as the exothermic profile is manageable and the workup involves simple phase separation and crystallization. From an environmental perspective, the reduction in organic solvent waste and the avoidance of toxic byproducts align with increasingly stringent global environmental regulations. The ability to process larger batches with a smaller environmental footprint facilitates the commercial scale-up of complex pharmaceutical intermediates, enabling manufacturers to meet growing global demand for Regorafenib while adhering to green chemistry principles and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this intermediate. These insights are derived directly from the patent data and practical manufacturing considerations, aiming to clarify the benefits of the new inorganic base route for potential partners. Understanding these details is essential for making informed sourcing decisions.
Q: Why is the inorganic base method safer than traditional potassium tert-butoxide routes?
A: Traditional methods utilize potassium tert-butoxide, which poses significant explosion risks during large-scale industrial production. The novel method replaces this hazardous organic base with stable inorganic bases like sodium hydroxide, drastically improving operational safety and eliminating thermal runaway hazards associated with strong organic alkalis.
Q: How does the new purification process impact environmental compliance?
A: Conventional processes rely on complex extraction, concentration, and column chromatography, generating substantial wastewater and organic solvent waste. The improved protocol employs a direct crystallization method by adding water, which simplifies post-reaction processing, significantly reduces solvent consumption, and minimizes the environmental footprint of the manufacturing cycle.
Q: What yields can be expected from this optimized synthetic route?
A: While prior art methods typically reported yields around 77% to 78%, the optimized inorganic base protocol demonstrates superior efficiency. Experimental data indicates that yields can be consistently improved to approximately 88%, providing a more material-efficient pathway for the production of this critical anticancer intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Amino-3-Fluorophenoxy)-N-Methylpyridine-2-Carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs depends on the reliability and quality of the supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-(4-amino-3-fluorophenoxy)-N-methylpyridine-2-carboxamide meets the highest international standards. We are committed to implementing advanced synthetic technologies, such as the inorganic base method described, to deliver superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain for Regorafenib production. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and reliability in your pharmaceutical manufacturing operations.
