Advanced Anhydrous Synthesis of Diallyl Bisphenol S Ether for High-Purity Polymer Applications
The chemical industry is currently witnessing a paradigm shift towards greener, more efficient synthesis routes, particularly for high-value intermediates used in advanced polymer applications. Patent CN112079756B introduces a groundbreaking preparation method for diallyl bisphenol S ether, a critical monomer for heat-resistant polymers and thermal recording materials. This innovation addresses the longstanding challenges of traditional aqueous alkylation processes, specifically the generation of hazardous salt-laden wastewater and the formation of difficult-to-remove impurities like bisphenol S triallyl ether. By transitioning to a strictly anhydrous system utilizing diethylene glycol monomethyl ether as a solvent and a non-ionic surfactant as a catalyst, this technology achieves a one-time conversion rate exceeding 99% and a product purity of over 99.6%. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier or polymer additive partner, this patent represents a significant leap forward in process intensification and environmental compliance, offering a robust pathway for commercial scale-up of complex fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for diallyl bisphenol S ether have historically relied on aqueous or alcohol-water mixed solvent systems, often employing liquid alkalis like sodium hydroxide solution. These conventional methods suffer from inherent thermodynamic and kinetic limitations that severely impact both product quality and environmental sustainability. The presence of water in the reaction medium inevitably leads to hydrolysis side reactions and promotes over-alkylation, resulting in the formation of bisphenol S triallyl ether, a persistent impurity that complicates downstream purification and lowers the overall yield to around 92%. Furthermore, the use of liquid alkali and aqueous solvents generates massive quantities of high-salinity wastewater containing dissolved organic compounds, posing a severe burden on wastewater treatment facilities and increasing the total cost of ownership. The necessity to distill and recover solvents from these aqueous mixtures further exacerbates energy consumption, making the process economically inefficient and environmentally unsustainable for modern large-scale manufacturing operations.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes an anhydrous aprotic strong polar solvent, specifically diethylene glycol monomethyl ether, which creates a homogeneous reaction phase with superior solubility for the sodium phenolate intermediate. This strategic shift eliminates water from the reaction environment entirely, thereby suppressing the formation of triallyl ether impurities and driving the reaction equilibrium towards the desired diallyl product with exceptional selectivity. The process employs solid alkali instead of liquid caustic soda, which facilitates the easy separation of inorganic salt byproducts as a solid filter cake rather than a dissolved waste stream. Crucially, the filtrate containing the solvent and the non-ionic surfactant catalyst can be directly recycled into subsequent batches without the need for energy-intensive distillation or purification steps. This closed-loop system not only drastically simplifies the operational workflow but also fundamentally solves the problem of industrial wastewater discharge, aligning perfectly with the stringent environmental regulations faced by a reliable specialty chemical supplier today.
Mechanistic Insights into Non-Ionic Surfactant Catalyzed Alkylation
The core of this technological breakthrough lies in the sophisticated interplay between the anhydrous polar solvent and the non-ionic surfactant catalyst, typically a fatty alcohol polyoxyethylene ether such as Peregal O. In this homogeneous system, the solvent acts not merely as a medium but as a critical promoter of nucleophilicity, effectively solvating the phenoxide anions generated from bisphenol S and solid sodium hydroxide. The non-ionic surfactant, added at a specific loading of at least 5% by weight relative to bisphenol S, functions through a mechanism akin to phase transfer catalysis but within a single liquid phase, enhancing the accessibility of the allyl chloride electrophile to the phenolic oxygen centers. This unique catalytic environment ensures that the alkylation proceeds rapidly and selectively at the phenolic hydroxyl groups without attacking other potential sites on the aromatic ring, thus maintaining the structural integrity of the bisphenol S backbone. The absence of water prevents the hydrolysis of allyl chloride to allyl alcohol, a common side reaction in aqueous systems that consumes reagents and generates additional waste, thereby maximizing atom economy.
Impurity control is another critical aspect where the mechanistic design excels, particularly in preventing the formation of bisphenol S triallyl ether. In conventional aqueous processes, the equilibrium dynamics often favor over-alkylation due to the solvation effects of water on the transition states. However, in this anhydrous system, the specific solvation shell provided by the glycol ether solvent stabilizes the mono- and di-alkylated intermediates in a way that kinetically hinders the third alkylation step. Experimental data confirms that even after multiple recycles of the reaction mother liquor, the content of the triallyl impurity remains undetectable (0%), while the purity of the target diallyl bisphenol S ether consistently stays above 99.6%. This high level of selectivity reduces the need for complex recrystallization or chromatographic purification steps, allowing for a streamlined isolation process where the product is simply dissolved in a weak polar solvent like toluene to separate it from the solid inorganic salts.
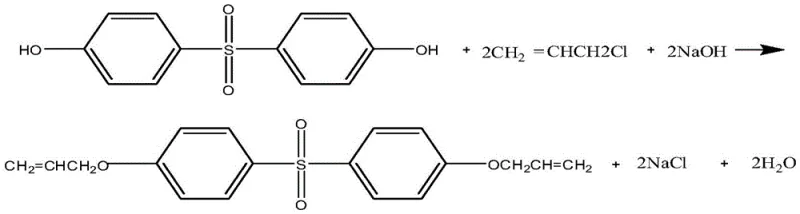
How to Synthesize Diallyl Bisphenol S Ether Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize the benefits of the anhydrous system. The process begins with the preparation of the phenoxide salt in situ by reacting bisphenol S with solid sodium hydroxide in the glycol ether solvent, followed by the controlled addition of allyl chloride under a nitrogen atmosphere to prevent oxidation. Temperature management is crucial, with the reaction initiated at 60°C and gradually ramped to 100°C to ensure complete conversion while minimizing thermal degradation of the sensitive allyl groups. The following section outlines the standardized operational protocol derived from the patent examples, providing a clear roadmap for laboratory validation and pilot plant trials.
- Prepare the reaction system by adding solid alkali (NaOH/KOH) and anhydrous diethylene glycol monomethyl ether into a reactor under stirring.
- Introduce Bisphenol S and a non-ionic surfactant catalyst (at least 5% wt of Bisphenol S), followed by the addition of chloropropene.
- Heat the mixture to 55-100°C for 7-12 hours under nitrogen, then perform solid-liquid separation to isolate the product and recycle the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this anhydrous synthesis technology translates into tangible strategic advantages that go beyond simple yield improvements. The ability to recycle the reaction mother liquor directly without distillation represents a massive reduction in utility costs, specifically steam and cooling water consumption, which are major cost drivers in batch chemical processing. By eliminating the need for solvent recovery units and wastewater treatment for high-salt effluents, the capital expenditure (CAPEX) for new production lines is significantly lowered, and the operational expenditure (OPEX) for existing facilities is optimized. This efficiency gain allows for cost reduction in polymer additive manufacturing, making the final product more competitive in price-sensitive markets such as thermal paper coatings and flame-retardant resins. Furthermore, the simplified post-treatment process, which involves mere filtration and crystallization, shortens the batch cycle time, thereby increasing the overall throughput of the manufacturing facility without requiring additional reactor volume.
- Cost Reduction in Manufacturing: The elimination of solvent distillation and wastewater treatment steps drastically reduces energy consumption and waste disposal fees. Since the catalyst and solvent are retained in the liquid phase and reused, the consumption of fresh raw materials per kilogram of product is minimized, leading to substantial variable cost savings. The high selectivity of the reaction means less raw material is wasted on byproducts, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of the anhydrous process ensures consistent product quality across multiple batches, reducing the risk of off-spec material that could disrupt downstream customer operations. The use of readily available commodity chemicals like solid caustic soda and allyl chloride, combined with a stable catalyst system, mitigates supply risks associated with specialized reagents. This reliability is crucial for maintaining long-term contracts with major polymer manufacturers who require uninterrupted supply of high-purity intermediates.
- Scalability and Environmental Compliance: The generation of solid inorganic salts instead of liquid brine simplifies waste handling and allows for the potential sale of high-purity sodium chloride as a byproduct, turning a waste liability into a minor revenue stream. This zero-liquid-discharge characteristic makes the process highly scalable in regions with strict environmental regulations, ensuring long-term operational continuity without the threat of regulatory shutdowns due to effluent violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this anhydrous alkylation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on scalability, impurity profiles, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the anhydrous process prevent the formation of bisphenol S triallyl ether?
A: The absence of water eliminates hydrolysis side reactions and suppresses over-alkylation, ensuring the product purity remains above 99.6% without triallyl impurities.
Q: Can the reaction solvent and catalyst be recycled without purification?
A: Yes, the patented method allows for the direct reuse of the filtrate containing the solvent and catalyst for multiple batches, significantly reducing waste and operational costs.
Q: What is the environmental advantage of this synthesis method?
A: By utilizing solid alkali and anhydrous conditions, the process generates solid inorganic salts instead of salt-containing wastewater, fundamentally solving industrial effluent treatment issues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diallyl Bisphenol S Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, eco-friendly synthesis routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the anhydrous process are fully realized in a practical industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of diallyl bisphenol S ether meets the exacting standards required for high-performance polymer applications. Our commitment to quality assurance means that customers receive a product with consistent impurity profiles, free from the triallyl contaminants that plague conventionally produced materials.
We invite global partners to collaborate with us to leverage this innovative technology for their specific application needs. Whether you are developing next-generation thermal recording materials or high-temperature resistant resins, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line. Together, we can drive the industry towards a more sustainable and efficient future in fine chemical manufacturing.
