Revolutionizing Organoboron Synthesis: Metal-Free Electrocatalysis for Commercial Scale Production
The landscape of organoboron chemistry is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing processes. A groundbreaking development detailed in patent CN114622226A introduces a novel method for synthesizing alkyl borate compounds through electrocatalysis, marking a departure from traditional transition metal-dependent pathways. This technology leverages electricity as a clean energy source to drive the hydroboration of aryl alkene compounds with pinacol borane, offering a robust alternative for producing high-purity alkyl borates. By utilizing the unique properties of acetonitrile solvent electrolysis in the presence of N,N-diisopropylethylamine, this method efficiently releases hydrogen protons to facilitate the reaction without external oxidants or reducing agents. For global procurement teams and R&D directors, this represents a pivotal shift towards greener chemistry that maintains high selectivity while drastically simplifying the downstream processing requirements typically associated with organometallic catalysis.
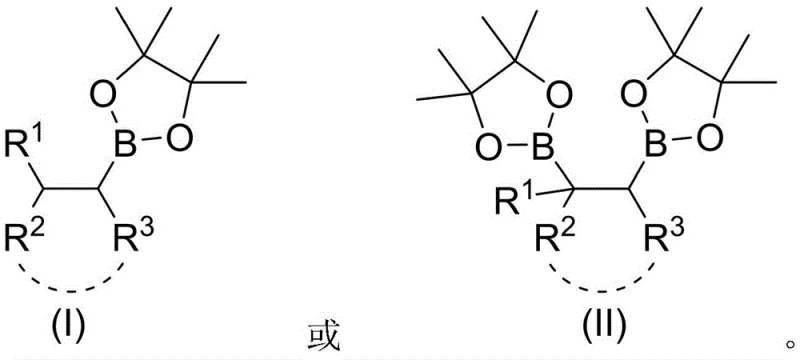
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the preparation of alkyl boronate esters has relied heavily on transition metal-catalyzed olefin hydroboration reactions, which involve the addition of a B-H bond across unsaturated double bonds. Since the development of Wilkinson's catalyst, numerous noble metal systems involving ruthenium, rhodium, and iridium, as well as non-noble metals like iron and cobalt, have been employed to achieve these transformations. While these catalysts often exhibit high chemo-selectivity and regio-selectivity, they impose severe limitations on large-scale manufacturing due to their inherent complexity and cost. The requirement for expensive and difficult-to-synthesize organic ligands, along with the necessity for specific metal complexes with single configurations, creates a significant barrier to entry for cost-sensitive production. Furthermore, the post-reaction removal and recovery of these metal catalysts present a major challenge, often necessitating additional purification steps that increase waste generation and extend production lead times, thereby impacting the overall supply chain efficiency for critical pharmaceutical intermediates.
The Novel Approach
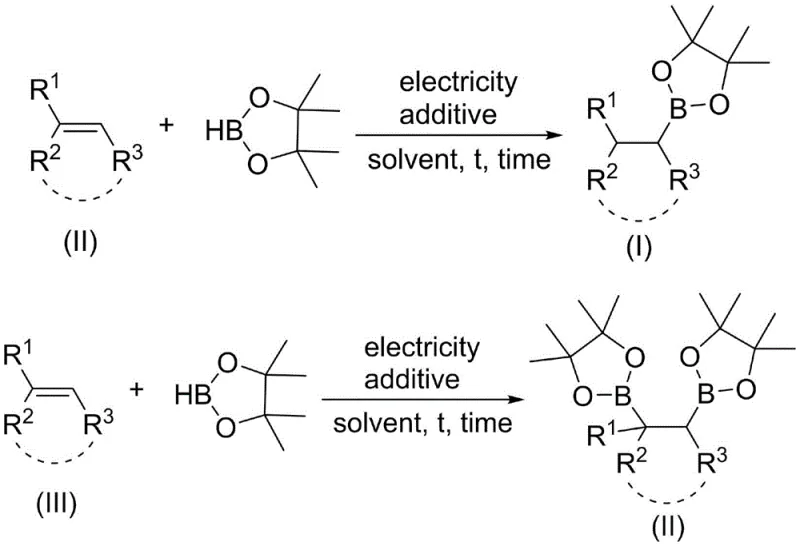
In stark contrast to conventional methodologies, the electrocatalytic approach described in the patent data offers a streamlined pathway that bypasses the need for transition metal catalysts entirely. This novel method utilizes cheap and easily obtained electricity as the primary energy source to drive the reaction, fundamentally altering the economic and environmental profile of alkyl borate synthesis. By employing a system where the solvent itself participates in the electrolysis process to release hydrogen protons, the reaction achieves high selectivity in converting aryl alkene compounds and pinacol borane into valuable alkyl borate esters. This metal-free strategy not only eliminates the cost associated with precious metal catalysts but also removes the regulatory and safety burdens linked to heavy metal residues in final products. The ability to selectively obtain different alkyl borate structures by adjusting reaction parameters provides a versatile platform for synthesizing complex intermediates required in drug discovery and material science, positioning this technology as a superior choice for modern chemical manufacturing.
Mechanistic Insights into Electrocatalytic Hydroboration
The core innovation of this technology lies in its unique mechanistic pathway where electrical energy replaces chemical reagents to drive the hydroboration process. In this system, the reaction is conducted in an anhydrous and oxygen-free environment using a combination of electrodes, such as platinum sheets, and a specific electrolyte system. The key mechanistic step involves the efficient electrolysis of the acetonitrile solvent under the auxiliary action of additives like N,N-diisopropylethylamine, which facilitates the release of hydrogen protons necessary for the reaction. These protons then interact with the pinacol borane and the aryl olefin substrate to form the carbon-boron bond with high precision. This electrochemical activation allows for fine-tuned control over the reaction kinetics by simply adjusting the rated current, typically ranging from 1mA to 100mA, providing a level of operational flexibility that is difficult to achieve with thermal or chemical activation methods. The absence of metal catalysts means the reaction mechanism avoids common deactivation pathways associated with metal poisoning, ensuring consistent performance across diverse substrate classes.
Impurity control in this electrocatalytic system is inherently superior due to the absence of metal species that often contribute to complex impurity profiles in traditional catalysis. Without the need for metal-ligand complexes, the risk of metal-induced side reactions or decomposition products is significantly minimized, leading to cleaner reaction mixtures. The selectivity of the process is further enhanced by the specific interaction between the electrolyte, the solvent, and the applied current, which directs the formation of the desired alkyl borate esters shown in structural formulas I and II. By carefully selecting the electrode material and optimizing the electrolyte concentration, manufacturers can suppress unwanted by-products and ensure high purity of the final organoboron compounds. This high level of purity is critical for downstream applications in pharmaceutical synthesis, where strict regulatory standards mandate minimal levels of elemental impurities, thus reducing the burden on quality control laboratories and accelerating the release of batches for clinical or commercial use.
How to Synthesize Alkyl Borate Efficiently
To implement this cutting-edge electrocatalytic synthesis route effectively, precise adherence to the patented reaction conditions is essential for achieving optimal yields and selectivity. The process begins with the preparation of a reaction vessel equipped with suitable electrodes, such as nickel, copper, or platinum sheets, ensuring a controlled environment free from moisture and oxygen which could interfere with the electrochemical process. Substrates including aryl olefin compounds and pinacol borane are introduced into a solvent system comprising acetonitrile and potentially other co-solvents like tetrahydrofuran, along with a supporting electrolyte such as tetrabutylammonium tetrafluoroborate. The addition of an amine additive is crucial for facilitating the proton release mechanism that drives the hydroboration. Once the mixture is prepared, a rated current is applied to initiate the reaction, which proceeds at temperatures ranging from 0 to 85 degrees Celsius depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Prepare an anhydrous and oxygen-free reaction environment using a three-neck flask equipped with appropriate electrodes such as platinum sheets.
- Combine aryl olefin compounds and pinacol borane in a solvent system containing acetonitrile and an electrolyte like tetrabutylammonium tetrafluoroborate.
- Apply a rated current between 1mA and 100mA in the presence of an additive like N,N-diisopropylethylamine to initiate the hydroboration reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrocatalytic technology translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The most significant benefit arises from the elimination of expensive transition metal catalysts, which are subject to volatile market pricing and complex supply chains. By replacing these costly reagents with electricity, manufacturers can achieve substantial cost savings in raw material procurement while simultaneously simplifying the inventory management of hazardous chemicals. Furthermore, the removal of metal catalysts obviates the need for specialized scavenging resins or complex filtration processes typically required to meet strict residual metal specifications, thereby reducing the consumption of auxiliary materials and shortening the overall production cycle time. This streamlining of the manufacturing process enhances the agility of the supply chain, allowing for faster response times to market demands and reducing the risk of production delays associated with catalyst availability or purification bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of switching to an electrocatalytic process is profound, primarily driven by the complete removal of noble metal catalysts such as rhodium or iridium from the bill of materials. These metals represent a significant portion of the variable costs in traditional organoboron synthesis, and their elimination results in direct material cost reductions. Additionally, the simplified workup procedure, which does not require extensive metal removal steps, reduces the consumption of solvents and purification media, leading to lower waste disposal costs and reduced utility consumption. The use of electricity as a reagent is inherently more efficient and scalable than stoichiometric chemical oxidants or reductants, offering a pathway to lower energy intensity per unit of product produced. These cumulative efficiencies contribute to a more competitive cost structure for high-purity alkyl borates, enabling better margin protection in price-sensitive markets.
- Enhanced Supply Chain Reliability: Relying on electricity as a primary driver for chemical transformation significantly de-risks the supply chain by reducing dependency on specialized chemical reagents that may face sourcing constraints. Transition metal catalysts often have long lead times and are sourced from geographically concentrated regions, creating vulnerability to geopolitical disruptions; replacing them with universally available electrical power enhances supply security. Moreover, the robustness of the electrochemical system against variations in raw material quality ensures consistent output, minimizing the risk of batch failures that can disrupt downstream production schedules. This reliability is crucial for maintaining continuous supply to pharmaceutical customers who require just-in-time delivery of critical intermediates, thereby strengthening the partnership between suppliers and end-users through improved service levels and predictability.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this metal-free methodology aligns perfectly with modern green chemistry principles and increasingly stringent regulatory requirements. The absence of heavy metals simplifies the treatment of effluent streams, reducing the environmental footprint of the manufacturing facility and lowering compliance costs associated with wastewater treatment. The process is inherently scalable, as demonstrated by gram-scale amplification experiments in the patent data, suggesting that transitioning from laboratory to commercial scale can be achieved with minimal re-engineering of the core reaction physics. This ease of scale-up supports the commercial production of complex organoboron compounds needed for emerging therapeutic areas, ensuring that supply can grow in tandem with demand without compromising on sustainability goals or regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows or for sourcing intermediates produced via this advanced route. The insights provided here aim to clarify the operational benefits and technical capabilities of the process for decision-makers in R&D and procurement.
Q: What are the primary advantages of electrocatalytic hydroboration over traditional metal-catalyzed methods?
A: The primary advantage is the elimination of expensive and toxic transition metal catalysts such as rhodium or iridium. This removes the need for complex ligand synthesis and costly metal removal steps, significantly simplifying the purification process and reducing environmental impact.
Q: Can this electrocatalytic method be scaled for industrial production of alkyl borates?
A: Yes, the patent data indicates successful gram-scale amplification experiments. The use of electricity as a reagent allows for precise control over reaction kinetics, which is highly favorable for scaling up complex organoboron compound manufacturing without the variability associated with chemical oxidants.
Q: What types of substrates are compatible with this electrocatalytic synthesis route?
A: The method demonstrates broad substrate scope, accommodating various aryl olefin compounds including styrenes with electron-donating or electron-withdrawing groups, as well as heterocyclic and polycyclic aromatic substrates, yielding both mono- and diboronated products selectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Borate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in delivering high-value chemical intermediates to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory technologies are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We are uniquely positioned to leverage this metal-free technology to provide our clients with a reliable alkyl borate supplier solution that balances cost efficiency with exceptional product quality, supporting your drug development pipelines with uninterrupted supply continuity.
We invite you to explore how this advanced manufacturing capability can optimize your supply chain and reduce overall project costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our electrocatalytic expertise can serve as a strategic asset for your organization, ensuring you stay ahead in the competitive landscape of pharmaceutical and fine chemical manufacturing.
