Scalable Synthesis of Ansa-Metallocene Precursors for Advanced Olefin Polymerization Catalysts
Scalable Synthesis of Ansa-Metallocene Precursors for Advanced Olefin Polymerization Catalysts
The development of high-performance olefin polymerization catalysts relies heavily on the precise synthesis of ansa-metallocene compounds, particularly those with specific substitution patterns that control steric hindrance during polymerization. Patent CN1753853A introduces a groundbreaking methodology for preparing 1,4,6-substituted, 1,4-substituted, 1,6-substituted, or 1-substituted methylenecyclopentadiene compounds, which serve as critical precursors for these advanced catalysts. This innovation addresses long-standing challenges in organometallic chemistry, specifically the difficulty of introducing substituents exclusively at the position adjacent to the bridging point of cyclopentadienyl ligands. By streamlining the synthetic pathway and eliminating hazardous high-pressure steps, this technology offers a robust solution for producing high-purity intermediates essential for copolymerizing monomers with high steric hindrance, such as norbornene, with ethylene.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass traditional bottlenecks associated with metallocene ligand synthesis. The described method utilizes a novel sequence involving nucleophilic addition to cyclopentenones, followed by lithiation, etherification, and dehydration. This approach not only simplifies the molecular construction but also ensures exceptional regioselectivity, a parameter that is often difficult to control in conventional routes. The resulting ansa-metallocene compounds exhibit superior catalytic properties due to their unique structural configuration, making this patent a cornerstone for developing next-generation polyolefin materials with tailored physical properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ansa-metallocene catalysts with substituents adjacent to the bridging point has been plagued by inefficient and hazardous reaction conditions. As illustrated in the prior art Reaction Scheme I, traditional methods often rely on the Pauson-Khand reaction, which necessitates the use of toxic and expensive cobalt carbonyl catalysts under high carbon monoxide pressure (e.g., 10 bar) and elevated temperatures. These severe conditions pose significant safety risks and engineering challenges for scale-up, requiring specialized high-pressure reactors that increase capital expenditure. Furthermore, the conventional pathway involves a Diels-Alder reverse reaction and multiple steps that generate complex mixtures of intermediates, necessitating rigorous separation and purification via column chromatography.
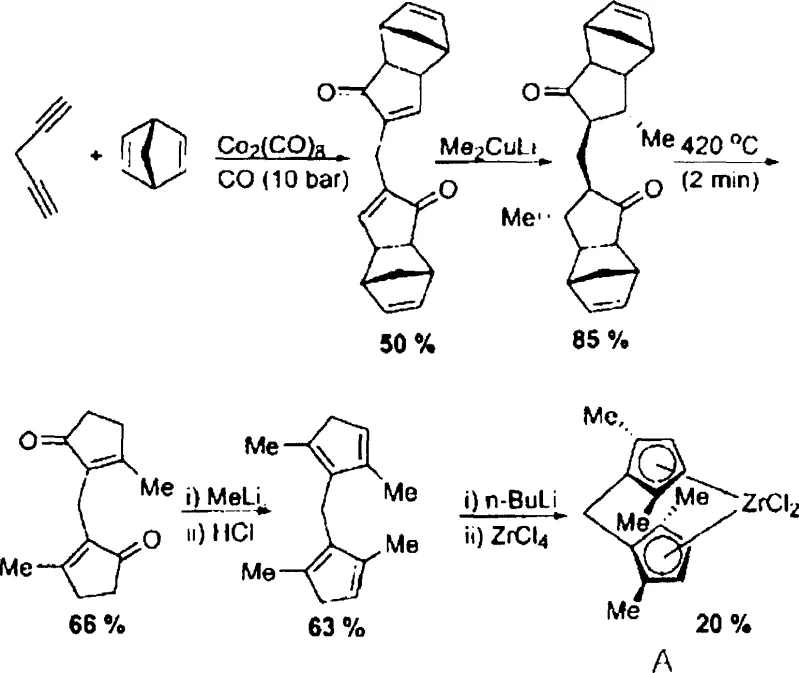
In addition to safety and equipment concerns, the conventional route suffers from poor atom economy and reliance on non-commercially available starting materials, such as 1,4-pentadiyne. The requirement for excess amounts of dangerous reagents like methyllithium further complicates the process, increasing both the cost of goods sold and the environmental footprint due to hazardous waste generation. The cumulative effect of these factors is a synthesis route that is fundamentally unsuitable for mass production, limiting the commercial availability of high-performance ansa-metallocene catalysts. The inability to easily purify intermediates without chromatography creates a bottleneck that prevents the consistent supply of high-purity materials required for sensitive polymerization applications.
The Novel Approach
The methodology disclosed in CN1753853A represents a paradigm shift by introducing a linear, protection-group-free synthesis that drastically reduces operational complexity. Instead of relying on cyclization reactions under extreme conditions, the novel approach builds the carbon skeleton through controlled nucleophilic attacks on readily available cyclopentenone derivatives. This strategy allows for the precise installation of substituents at the desired positions without the steric clashes that typically lead to unwanted isomers. By avoiding the need for ketal protection and deprotection steps, which are common in alternative schemes, the process minimizes the number of unit operations and reduces solvent consumption. The elimination of chromatographic purification in favor of vacuum distillation or simple extraction marks a critical advancement for industrial viability.
Moreover, the new route leverages commercially accessible starting materials, such as 2-bromo-3-methyl-2-cyclopenten-1-one, ensuring a stable and cost-effective supply chain. The reaction conditions are significantly milder, typically operating at low temperatures for lithiation steps followed by ambient or moderate heating for dehydration, thereby reducing energy consumption. This streamlined workflow not only accelerates the timeline from raw materials to final product but also enhances the overall yield and purity profile. The ability to produce the target methylenecyclopentadiene compounds in fewer steps with higher reliability makes this approach uniquely suited for meeting the demanding specifications of the specialty chemicals market.
Mechanistic Insights into Nucleophilic Construction of Methylenecyclopentadiene
The core of this technological breakthrough lies in the sophisticated manipulation of organometallic intermediates to achieve specific regiochemistry. The process initiates with the reaction of a halogenated cyclopentenone (Formula I) with an organolithium or Grignard reagent (Formula II), resulting in a tertiary alcohol intermediate (Formula III). This step is crucial as it establishes the initial carbon framework and introduces the first substituent. Subsequent lithiation of this intermediate generates a reactive species that can be trapped with various electrophiles (Formula IV), such as formaldehyde or acetone, to extend the carbon chain or introduce functional handles. The precision of this lithiation step is vital, as it determines the positioning of the future bridge and substituents on the cyclopentadiene ring.
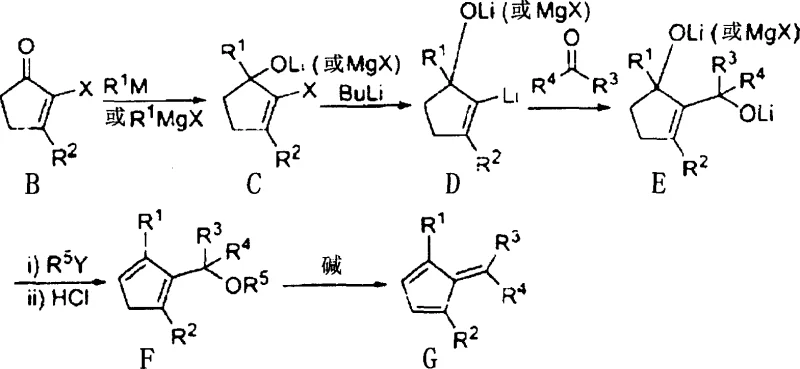
Following the electrophilic capture, the intermediate undergoes etherification with an alkyl halide or sulfonate (Formula VI) to protect the hydroxyl groups as ethers. This protection is transient and strategic; it stabilizes the molecule during subsequent transformations while setting the stage for the final elimination. The dehydration step, catalyzed by acid, removes the alkoxy group along with an adjacent hydrogen atom to form the exocyclic double bond characteristic of methylenecyclopentadienes (Formula VII to Formula VIII). This elimination is highly selective, driven by the stability of the resulting conjugated diene system. The final treatment with a strong base facilitates the complete aromatization or formation of the diene system, yielding the target ligand precursor. This mechanistic pathway effectively circumvents the steric hindrance issues observed in direct nucleophilic attacks on pre-formed methylenecyclopentadienes, ensuring that substituents are located exclusively at the alpha-position relative to the bridge.
Impurity control is inherently built into this mechanism through the use of crystalline or distillable intermediates. Unlike the messy mixtures generated by the Pauson-Khand reaction, the intermediates in this novel route (such as Formula III and Formula VII) possess distinct physical properties that allow for efficient separation via extraction or distillation. The avoidance of transition metal catalysts in the main synthetic sequence eliminates the risk of heavy metal contamination, a critical quality attribute for catalysts used in producing polymers for medical or food-contact applications. The robustness of the lithiation and etherification steps ensures that side reactions are minimized, leading to a cleaner reaction profile and higher overall process efficiency.
How to Synthesize Substituted Methylenecyclopentadiene Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize yield and selectivity. The patent outlines a reproducible protocol that begins with the preparation of the tertiary alcohol intermediate at cryogenic temperatures to prevent side reactions. Detailed standard operating procedures for the lithiation, electrophilic quench, and dehydration steps are essential for maintaining batch-to-batch consistency. For a comprehensive guide on executing this synthesis with optimal safety and efficiency, please refer to the standardized protocol below.
- React a halogenated cyclopentenone (Formula I) with an organolithium or Grignard reagent (Formula II) to form a tertiary alcohol intermediate (Formula III).
- Lithiate the intermediate and react with an electrophile (Formula IV), followed by etherification with an alkyl halide (Formula VI) and acid-catalyzed dehydration to yield Formula VII.
- Treat the ether compound (Formula VII) with a strong base to eliminate the alkoxy group, producing the target methylenecyclopentadiene (Formula VIII).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis route offers transformative benefits in terms of cost structure and supply reliability. The shift away from exotic starting materials and high-pressure processing equipment directly translates to reduced capital and operational expenditures. By utilizing commodity chemicals like 2-bromo-cyclopentenones and simple alkyl halides, manufacturers can secure raw material supplies from multiple global vendors, mitigating the risk of single-source dependency. This diversification of the supply base is critical for maintaining continuity in the production of downstream polymerization catalysts, which are essential for the plastics and elastomers industries.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, such as cobalt carbonyl, and the removal of high-pressure reactor requirements significantly lower the barrier to entry for production. Furthermore, replacing labor-intensive chromatographic purification with scalable vacuum distillation reduces solvent usage and waste disposal costs. The streamlined process flow, which avoids protection-deprotection sequences, shortens the manufacturing cycle time, allowing for faster throughput and better asset utilization without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials ensures that production schedules are not held hostage by the long lead times associated with custom-synthesized precursors. The robustness of the chemical steps means that the process is less susceptible to variability, leading to more predictable delivery timelines. This stability is paramount for downstream customers who require consistent quality and volume to maintain their own polymerization operations, thereby strengthening the overall resilience of the chemical supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving away from batch processes that are difficult to enlarge towards continuous or semi-continuous operations. The reduction in hazardous reagents and the avoidance of high-pressure carbon monoxide improve the safety profile of the facility, simplifying regulatory compliance and permitting. Additionally, the decreased solvent intensity and waste generation align with green chemistry principles, reducing the environmental footprint and potential liability associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology for industrial applications.
Q: What are the primary advantages of this new synthesis route over the Pauson-Khand reaction?
A: The new route eliminates the need for high-pressure carbon monoxide and expensive cobalt catalysts required in the Pauson-Khand reaction. It operates under milder conditions and avoids complex chromatographic purification steps, significantly enhancing scalability.
Q: How does this method address steric hindrance issues in ligand substitution?
A: By utilizing a specific sequence of nucleophilic attack followed by controlled etherification and dehydration, the process ensures substituents are placed exclusively at the position adjacent to the bridging point (alpha-position), overcoming the beta-substitution bias seen in direct nucleophilic attacks on methylenecyclopentadiene.
Q: Is this process suitable for industrial-scale manufacturing?
A: Yes, the process is designed for mass production. It utilizes commercially available starting materials like 2-bromo-3-methyl-2-cyclopenten-1-one and replaces labor-intensive chromatography with vacuum distillation, making it highly viable for large-scale supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ansa-Metallocene Precursor Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex academic innovations like CN1753853A into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry under inert atmospheres, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch of ansa-metallocene precursor meets the exacting standards required for high-performance catalysis.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific polymerization needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient pathway can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for specialty polymer additives.
