Advanced Synthesis of Asymmetric Thiophene[7]Helicene Isomers for High-Performance Organic Electronics
Advanced Synthesis of Asymmetric Thiophene[7]Helicene Isomers for High-Performance Organic Electronics
The rapid evolution of organic electronics demands materials with precisely tuned electronic properties, driving the search for novel helicene architectures. Patent CN111057087B discloses a groundbreaking methodology for synthesizing asymmetric thiophene[7]helicene isomers, a class of compounds previously inaccessible through conventional symmetric routes. This innovation addresses a critical gap in the field of organic semiconductors, offering materials with significantly enhanced charge carrier mobility and on-off ratios. For R&D directors and procurement specialists in the display and photovoltaic sectors, this patent represents a pivotal shift towards higher performance OLED and OFET materials. The disclosed method not only achieves high purity but also establishes a robust framework for the commercial scale-up of complex hetero-helicenes, ensuring a reliable supply chain for next-generation electronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiophene[7]helicenes has been restricted to symmetrical structures, which inherently limits the tunability of their electronic and optical properties. Conventional methods often rely on repetitive coupling strategies that result in rigid, centrosymmetric molecules, leading to suboptimal packing in thin films and restricted charge transport pathways. Furthermore, the lack of structural asymmetry prevents the fine-tuning of energy levels required for efficient injection and extraction of charges in organic field-effect transistors. Existing synthetic routes frequently suffer from low regioselectivity, generating complex mixtures of isomers that are difficult and costly to separate, thereby inflating the final cost of goods and delaying time-to-market for new electronic products.
The Novel Approach
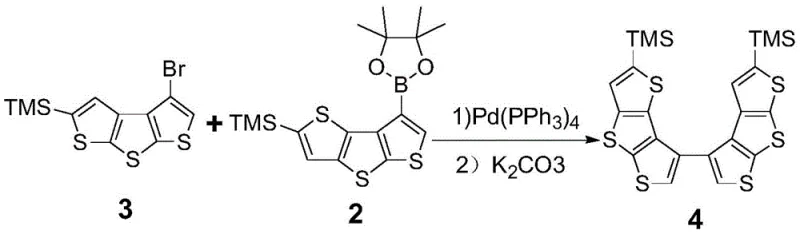
The patented methodology overcomes these barriers by introducing a strategic sequence of cross-coupling and oxidative cyclization reactions tailored to construct asymmetric scaffolds. By utilizing specific brominated and boronated precursors, the process enables the precise assembly of non-symmetric dithienothiophene dimers, which serve as the critical intermediates for the final helical closure. This approach allows for the deliberate placement of trimethylsilyl (TMS) groups and sulfur atoms to modulate solubility and intermolecular interactions without compromising the conjugated system. The result is a versatile platform capable of generating multiple isomers (TM-1, TM-2, TM-3) with distinct physical properties, providing material scientists with a broader toolkit for optimizing device performance in organic solar cells and light-emitting diodes.
Mechanistic Insights into Pd-Catalyzed Coupling and Oxidative Cyclization
The core of this synthesis lies in a highly efficient Suzuki-Miyaura cross-coupling reaction followed by a specialized oxidative ring-closing step. Initially, a brominated thiophene derivative undergoes lithiation with t-BuLi or n-BuLi at cryogenic temperatures (-70°C to -90°C), followed by quenching with a pinacol borate ester to form the reactive boronic species. This intermediate is then coupled with a complementary brominated unit using a palladium catalyst, such as Pd(PPh3)4 or Pd(OAc)2, in the presence of a carbonate base like K2CO3 or Cs2CO3. The reaction conditions, typically refluxing in THF or toluene at 100-110°C, ensure high conversion rates while minimizing homocoupling side products, which is essential for maintaining the structural integrity of the asymmetric backbone.
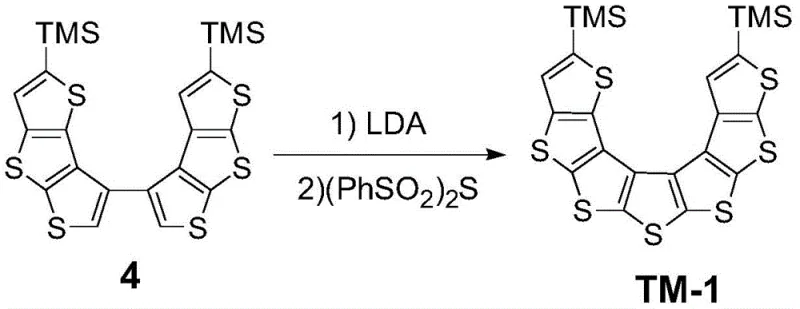
The final and most critical transformation involves the deprotonation of the dimeric precursor using lithium diisopropylamide (LDA) at low temperatures, followed by the addition of diphenyl disulfone ((PhSO2)2S) as the sulfur source. This oxidative cyclization step effectively stitches the open chain into the rigid, helical thiophene[7] structure. The mechanism proceeds through the generation of a carbanion which attacks the sulfur electrophile, facilitating the formation of the new carbon-sulfur bond that locks the helicene geometry. This step is performed under strict inert atmosphere conditions to prevent oxidation of the sensitive organolithium species, ensuring high yields of the target isomer. The precision of this cyclization is what differentiates this patent from prior art, enabling the exclusive formation of the desired asymmetric isomer over other potential regioisomers.
How to Synthesize Asymmetric Thiophene[7]Helicene Efficiently
The synthesis protocol described in the patent offers a clear pathway for laboratory and pilot-scale production, emphasizing strict temperature control and anhydrous conditions to maximize yield. The process begins with the preparation of the boronic ester intermediate, followed by the palladium-catalyzed dimerization, and concludes with the LDA-mediated cyclization. Each step has been optimized to balance reaction kinetics with product stability, ensuring that the sensitive helicene core is formed without degradation. For detailed operational parameters, stoichiometry, and workup procedures necessary for reproducible results, please refer to the standardized synthesis guide below.
- Perform lithiation of the bromo-thiophene precursor followed by borylation using pinacol borate to generate the boronic ester intermediate.
- Execute a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction between the boronic ester and a brominated dithienothiophene derivative to form the dimer.
- Conduct oxidative cyclization using LDA for deprotonation and diphenyl disulfone as the sulfur source to close the helical structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric synthesis route offers substantial strategic advantages over traditional helicene manufacturing. The use of commercially available starting materials and standard catalysts significantly reduces the dependency on exotic reagents, thereby stabilizing raw material costs and mitigating supply risks. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which simplifies downstream purification processes and reduces solvent consumption. This efficiency translates directly into a more sustainable manufacturing footprint and lower overall production costs, making high-performance organic semiconductors more accessible for mass-market applications.
- Cost Reduction in Manufacturing: The streamlined three-step sequence eliminates the need for complex multi-step functional group manipulations often required in symmetric helicene synthesis. By avoiding expensive chiral resolution steps and utilizing robust catalytic systems, the process drastically lowers the cost per gram of the final active material. The high yields reported in the examples, particularly in the coupling and cyclization stages, ensure that raw material utilization is maximized, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on common reagents such as palladium catalysts, lithium bases, and simple thiophene derivatives ensures a resilient supply chain that is less susceptible to geopolitical or logistical disruptions. Unlike processes requiring custom-synthesized building blocks with long lead times, this route leverages commodity chemicals that are readily available from multiple global suppliers. This flexibility allows for rapid scaling of production capacity to meet fluctuating market demands without the bottleneck of specialized precursor availability.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, with temperatures and pressures that can be safely managed in standard stainless steel reactors. The avoidance of heavy metal contaminants in the final product, due to efficient purification protocols, aligns with stringent environmental regulations for electronic materials. Additionally, the reduced solvent usage and higher atom economy of the coupling reactions contribute to a greener manufacturing profile, supporting corporate sustainability goals and compliance with increasingly strict waste disposal standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these asymmetric helicene isomers. Understanding these details is crucial for integrating this material into your existing R&D pipelines and supply chain strategies. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation.
Q: What are the electronic properties of the asymmetric thiophene[7]helicene isomers?
A: The synthesized isomers exhibit excellent semiconductor properties, including a hole mobility of 0.07-0.1 cm²/V·s, a threshold voltage ranging from -58V to -62V, and an on-off ratio exceeding 1.0 x 10^6, making them superior to existing symmetric analogues.
Q: How does the asymmetric structure benefit OLED applications compared to symmetric helicenes?
A: The asymmetric structure introduces unique molecular packing and dipole moments that can enhance charge transport and film morphology in organic light-emitting diodes, addressing the limitations of rigid symmetric scaffolds.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route utilizes standard reagents like Pd(PPh3)4 and LDA under controlled temperatures, avoiding exotic catalysts, which facilitates scale-up from gram to kilogram quantities for industrial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Thiophene[7]Helicene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply of high-purity OLED materials remains uninterrupted. We employ rigorous QC labs and stringent purity specifications to guarantee that every batch of asymmetric thiophene[7]helicene meets the exacting standards required for high-performance organic electronics.
We invite you to collaborate with us to optimize this synthesis for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your product development cycle and secure a competitive advantage in the global organic semiconductor market.
