Scalable Synthesis of Benzothiadiazole Thiophene Derivatives for Next-Gen Photovoltaics
Scalable Synthesis of Benzothiadiazole Thiophene Derivatives for Next-Gen Photovoltaics
The rapid evolution of organic photovoltaics (OPV) demands precise molecular engineering to achieve higher power conversion efficiencies and better stability. As detailed in Chinese Patent CN103601740A, a novel class of 2,1,3-benzothiadiazolodithiophene derivative bromides has been developed to address these critical needs. Specifically, the patent discloses the synthesis of 5,8-bis(5-bromo-4-alkylthiophen-2-yl)thieno[2',3':3,4,3',2':5,6]benzo[1,2-c][1,2,5]thiadiazole, a sophisticated intermediate designed for narrow bandgap polymer donor materials. This molecule combines the rigidity of the benzothiadiazole core with the solubility-enhancing properties of alkyl-substituted thiophenes, creating an ideal building block for high-performance electronic materials. The strategic placement of bromine atoms at the 5-position of the terminal thiophene rings enables versatile downstream polymerization, making it a cornerstone for advanced optoelectronic applications.
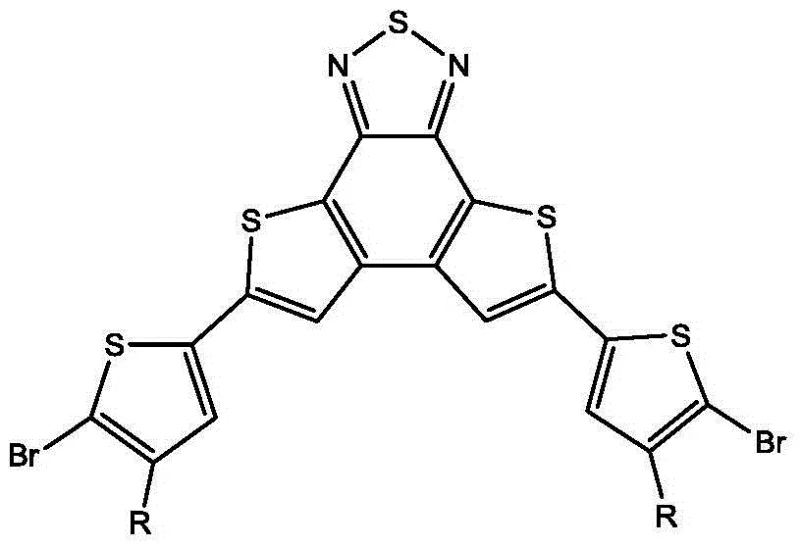
For R&D directors and procurement specialists seeking a reliable organic solar cell material supplier, understanding the structural nuances of this compound is paramount. The incorporation of alkyl chains (R groups ranging from C1 to C20) is not merely a solubility aid; it dictates the packing morphology of the resulting polymer films, which directly influences charge carrier mobility. By controlling the length and branching of these alkyl groups, manufacturers can fine-tune the material's processing window and final device performance. This level of molecular precision ensures that the resulting conjugated polymers possess the necessary thermal and chemical stability to withstand the rigorous conditions of device fabrication and operation, thereby extending the operational lifetime of organic solar modules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fused-ring electron-deficient units like benzothiadiazoles often suffers from harsh reaction conditions and poor regioselectivity during functionalization. Conventional routes frequently involve multiple protection and deprotection steps to achieve the desired substitution pattern, leading to significant yield losses and increased waste generation. Furthermore, direct bromination of thiophene-fused systems can result in mixtures of mono-, di-, and poly-brominated products, necessitating complex and costly purification processes such as repeated column chromatography or recrystallization. These inefficiencies not only drive up the cost of goods sold (COGS) but also introduce variability in the impurity profile, which is detrimental to the reproducibility of electronic devices. The reliance on unstable intermediates and sensitive reagents in older methodologies further complicates the scale-up process, posing risks to supply chain continuity for large-scale electronic chemical manufacturing.
The Novel Approach
The methodology outlined in the patent offers a streamlined and robust alternative that overcomes these historical bottlenecks. By utilizing a stepwise approach starting from dithienobenzothiadiazole, the process achieves high regioselectivity through controlled bromination and subsequent cross-coupling reactions. The key innovation lies in the sequential construction of the molecular architecture: first establishing the dibromo core, then attaching the alkylated thiophene units via palladium-catalyzed coupling, and finally installing the terminal bromine handles. This logical progression minimizes side reactions and ensures that the final product is obtained with high purity and consistent quality. The use of commercially available reagents and standard solvent systems simplifies the operational complexity, making the transition from laboratory bench to pilot plant significantly smoother and more predictable for industrial partners.
Mechanistic Insights into Regioselective Bromination and Stille Coupling
The synthetic pathway relies heavily on the precise control of electrophilic aromatic substitution and transition metal catalysis. In the initial stages, the electron-rich thiophene rings fused to the benzothiadiazole core are selectively brominated using molecular bromine in a chloroform-acetic acid mixture. The reaction conditions, specifically heating to 60-70°C, are optimized to favor dibromination at the 5,8-positions while avoiding over-bromination which could degrade the conjugated system. Following this, the Stille coupling reaction serves as the pivotal step for introducing the solubilizing alkyl chains. Using tributyl(4-alkylthiophen-2-yl)tin and a tetrakis(triphenylphosphine)palladium catalyst in dimethylformamide (DMF) at elevated temperatures (155-165°C), the carbon-carbon bonds are formed with high fidelity. This step is critical as it defines the steric environment around the core, influencing the planarity and pi-stacking interactions of the final polymer.
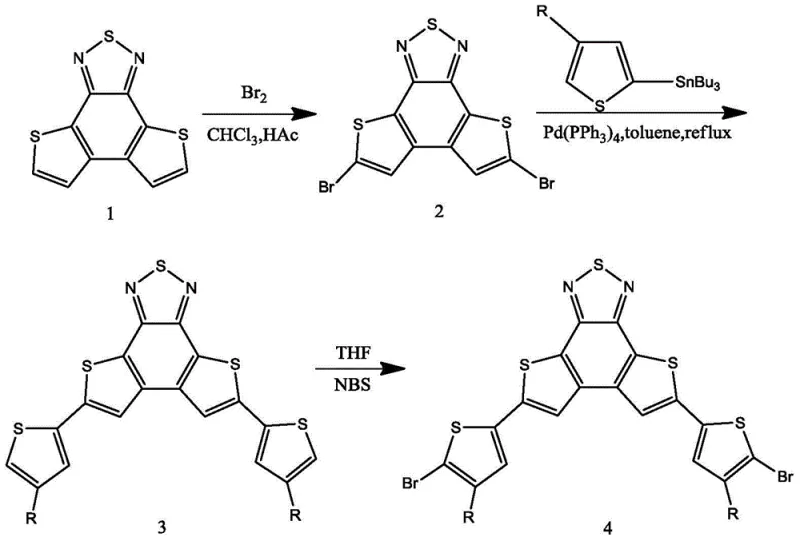
The final transformation involves the regioselective bromination of the terminal thiophene rings using N-bromosuccinimide (NBS). As illustrated in the reaction scheme, this step occurs under mild conditions at room temperature in tetrahydrofuran (THF), demonstrating the high reactivity of the alpha-positions on the thiophene rings relative to the beta-positions. The mechanism proceeds via an electrophilic attack where the succinimide radical or cation facilitates the replacement of the hydrogen atom with bromine. This mild condition is advantageous for preserving the integrity of the alkyl chains and the central heterocyclic core, preventing thermal degradation or isomerization. For quality control teams, this predictability in reaction outcome translates to a narrower impurity spectrum, reducing the burden on downstream purification and ensuring that the material meets the stringent specifications required for high-purity OLED material or photovoltaic applications.
How to Synthesize 5,8-bis(5-bromo-4-alkylthiophen-2-yl)thienobenzothiadiazole Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing this valuable intermediate with high reproducibility. The process is divided into three distinct operational phases, each optimized for yield and purity. The initial preparation of the dibromo core sets the foundation for the subsequent coupling, while the final bromination step installs the reactive handles necessary for polymerization. Operators should pay close attention to the stoichiometry of the reagents, particularly the molar ratios of bromine and NBS, to ensure complete conversion without excessive byproduct formation. The detailed standardized synthesis steps below outline the specific conditions and workup procedures required to achieve the reported results.
- Prepare 5,8-dibromodithienobenzothiadiazole by reacting the core monomer with bromine in chloroform and acetic acid at 60-70°C.
- Perform Stille coupling using tributyl(4-alkylthiophen-2-yl)tin and palladium catalyst in DMF at 155-165°C to attach alkyl thiophene units.
- Conduct final regioselective bromination using NBS in THF at room temperature to yield the target 5,8-bis(5-bromo-4-alkylthiophen-2-yl) derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that align with the goals of cost reduction in electronic chemical manufacturing. The elimination of exotic or highly unstable reagents reduces the dependency on specialized supply chains, thereby mitigating procurement risks. The use of standard solvents like chloroform, DMF, and THF allows for easier solvent recovery and recycling, contributing to a more sustainable and economically viable production process. Furthermore, the high yields reported in the patent examples suggest that the process is atom-economical, minimizing raw material waste and maximizing output per batch. For supply chain heads, this efficiency translates into shorter lead times for high-purity electronic chemicals and a more stable inventory management strategy.
- Cost Reduction in Manufacturing: The streamlined three-step sequence significantly reduces the number of unit operations compared to traditional multi-step syntheses. By avoiding complex protection group chemistry and utilizing robust catalytic systems, the process lowers the consumption of expensive reagents and catalysts. The simplified workup procedures, which involve standard aqueous washes and crystallization rather than extensive chromatography, drastically cut down on silica gel and solvent usage. These operational efficiencies cumulatively drive down the overall production cost, allowing for more competitive pricing in the global market for specialty organic intermediates.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 3-bromothiophene and common alkyl halides ensures a stable supply base. Unlike proprietary monomers that may be sourced from a single vendor, the precursors for this synthesis are commodity chemicals with multiple global suppliers. This diversification reduces the risk of supply disruptions due to geopolitical issues or manufacturer-specific problems. Additionally, the robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites without significant re-optimization, ensuring consistent product availability for downstream polymer manufacturers.
- Scalability and Environmental Compliance: The reaction conditions are well-suited for scale-up, with temperatures and pressures that are manageable in standard stainless steel reactors. The absence of cryogenic conditions or high-pressure hydrogenation simplifies the engineering requirements for commercial scale-up of complex organic intermediates. Moreover, the waste streams generated are primarily organic solvents and inorganic salts, which can be treated using established industrial wastewater treatment protocols. This alignment with environmental regulations reduces the compliance burden and potential fines, making the process attractive for companies aiming to improve their green chemistry metrics and corporate sustainability profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this benzothiadiazole derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the material's properties and the feasibility of its integration into existing manufacturing workflows.
Q: What are the solubility characteristics of this benzothiadiazole derivative?
A: The synthesized derivative exhibits excellent solubility in common organic solvents such as dichloromethane, chloroform, toluene, and THF, facilitating solution-processing for thin-film applications.
Q: Why is the alkyl chain introduction critical for this material?
A: Introducing alkyl chains at the 4-position of the thiophene rings significantly enhances the solubility of the rigid conjugated backbone without compromising the electronic properties required for efficient charge transport.
Q: Can this intermediate be used for polymer synthesis?
A: Yes, the terminal bromine atoms allow for further polymerization reactions, such as Stille coupling with distannyl compounds, to create narrow bandgap conjugated polymers for organic solar cells.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,8-bis(5-bromo-4-alkylthiophen-2-yl)thienobenzothiadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation renewable energy technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5,8-bis(5-bromo-4-alkylthiophen-2-yl)thienobenzothiadiazole meets the exacting standards required for organic photovoltaic applications. Our commitment to quality assurance means that you can rely on us for consistent material performance, batch after batch.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your R&D initiatives. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall material costs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your product development timeline. Partner with us to secure a stable supply of advanced electronic materials and drive innovation in the field of organic solar cells.
