Advanced Nickel-Catalyzed Synthesis of Doxepin Hydrochloride for Commercial Scale-Up
Introduction to Next-Generation Doxepin Hydrochloride Manufacturing
The pharmaceutical landscape for antidepressant intermediates is constantly evolving, driven by the need for more sustainable and cost-efficient manufacturing processes. Patent CN102924424A introduces a groundbreaking methodology for the synthesis of Doxepin Hydrochloride, a critical active pharmaceutical ingredient used globally for treating depression and anxiety neurosis. This technical disclosure moves away from traditional reliance on scarce and expensive noble metal catalysts, proposing a robust alternative that leverages abundant Nickel-based catalytic systems. For R&D directors and procurement managers alike, this shift represents a pivotal opportunity to optimize supply chains while maintaining rigorous quality standards. The innovation lies not just in the substitution of metals, but in the holistic redesign of the synthetic pathway to ensure high selectivity and environmental compliance.
This report analyzes the technical merits of this novel route, specifically focusing on its implications for commercial production. By replacing precious metals like Palladium or Platinum with a Nickel(II) acetate and triphenylphosphine system, the process drastically reduces raw material costs without compromising reaction efficiency. The patent data indicates a total yield reaching 62%, with key coupling steps achieving selectivity greater than 99%. Such metrics are crucial for establishing a reliable pharmaceutical intermediate supplier status, as they directly correlate to batch consistency and downstream purification costs. We will explore how this chemistry translates into tangible business value for stakeholders managing complex API portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex tricyclic antidepressants like Doxepin has often relied on transition metal-catalyzed cross-coupling reactions utilizing Palladium, Platinum, Ruthenium, or Rhodium. While chemically effective, these conventional methods present significant logistical and economic challenges for large-scale manufacturing. The primary drawback is the exorbitant cost of these noble metals, which creates volatility in production budgets and exposes supply chains to geopolitical risks associated with rare earth mining. Furthermore, the removal of residual heavy metals from the final API is a stringent regulatory requirement, necessitating expensive and time-consuming purification steps such as specialized scavenging resins or repeated recrystallizations.
Beyond cost, the environmental footprint of noble metal catalysis is substantial. The disposal of spent catalysts containing precious metals requires specialized hazardous waste handling protocols, increasing the operational overhead for chemical plants. Additionally, many traditional routes suffer from moderate selectivity, leading to complex impurity profiles that complicate quality control and reduce overall throughput. For a procurement manager, these factors translate into higher unit costs and longer lead times. The industry urgently requires a paradigm shift towards base metal catalysis that can deliver comparable or superior performance while mitigating these structural inefficiencies inherent in legacy synthetic pathways.
The Novel Approach
The methodology outlined in CN102924424A addresses these pain points through a cleverly engineered five-step sequence that culminates in a Nickel-catalyzed C-N bond formation. This approach replaces the expensive noble metals with a Ni(OAc)2/PPh3 system, which is not only orders of magnitude cheaper but also demonstrates remarkable stability and recyclability. The reaction pathway begins with a Grignard-type addition to form an alcohol intermediate, followed by dehydration and chlorination to set up the final coupling substrate. This logical progression ensures that each intermediate is robust and easily purified, laying a solid foundation for the high-yielding final steps.
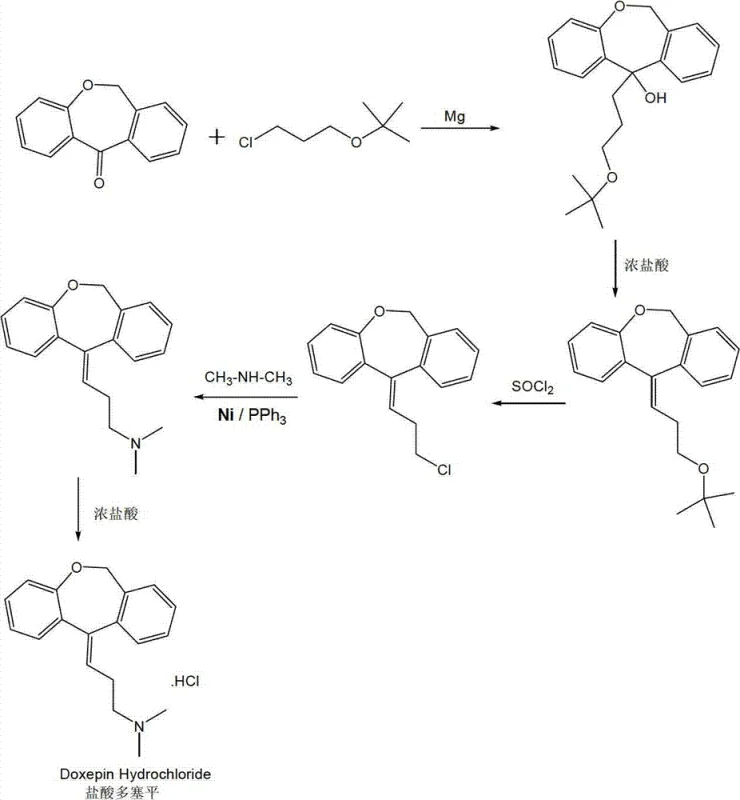
Crucially, the novel approach simplifies the operational complexity of the synthesis. The Nickel catalyst can be recovered through simple filtration after the reaction, allowing for multiple reuse cycles which further amortizes the catalyst cost over time. The process operates under relatively mild conditions, with the critical coupling step occurring at temperatures between 35°C and 45°C in DMF solvent. This thermal efficiency reduces energy consumption compared to high-temperature alternatives. For supply chain heads, this translates to a process that is easier to scale from pilot plant to commercial production, offering a reliable source of high-purity Doxepin Hydrochloride with a significantly reduced environmental impact and lower total cost of ownership.
Mechanistic Insights into Ni(OAc)2/PPh3 Catalyzed C-N Coupling
The heart of this synthetic innovation is the fourth step, where the chloro-intermediate undergoes nucleophilic substitution with dimethylamine facilitated by the Nickel catalyst. Mechanistically, this likely proceeds through a cycle involving oxidative addition of the aryl or alkyl chloride to the Nickel center, followed by ligand exchange with the amine and subsequent reductive elimination to form the C-N bond. The use of triphenylphosphine (PPh3) as a ligand is critical; it stabilizes the Nickel species and modulates its electronic properties to favor the desired coupling over competing side reactions such as dehalogenation or homocoupling. The patent specifies a ligand-to-substrate ratio of roughly 10:1, ensuring a saturated coordination sphere that maximizes catalytic turnover.
This mechanistic precision results in exceptional selectivity, reported to exceed 99% in the coupling step. High selectivity is paramount for R&D directors concerned with the impurity profile of the final API. By minimizing the formation of structural analogs or byproducts, the downstream purification burden is lightened, often requiring only standard recrystallization with petroleum ether to achieve HPLC purity greater than 99%. This level of control is difficult to achieve with less selective catalysts and underscores the sophistication of the Ni/PPh3 system. It effectively decouples the cost of the catalyst from the cost of purification, delivering a dual benefit of lower input costs and higher output quality.
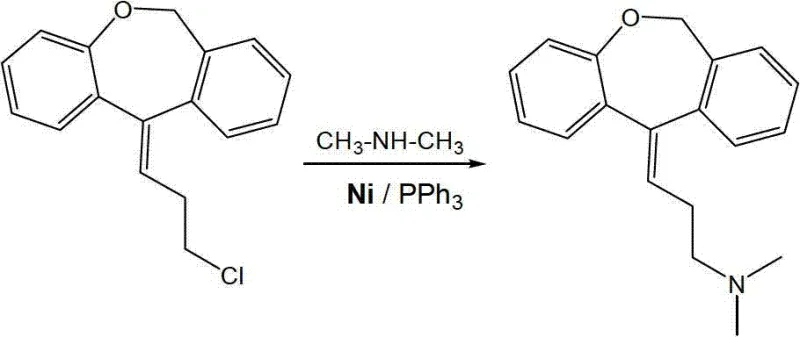
Furthermore, the robustness of the Nickel catalyst against poisoning by impurities or moisture is a significant advantage in an industrial setting. Unlike some sensitive Palladium systems that require rigorously anhydrous and oxygen-free environments, this Nickel system demonstrates tolerance that simplifies reactor setup and operation. The ability to recycle the catalyst via filtration means that the effective loading of fresh Nickel per batch can be kept extremely low, aligning with green chemistry principles. For technical teams evaluating process feasibility, this mechanism offers a clear path to a sustainable manufacturing process that meets both economic and regulatory demands for modern pharmaceutical production.
How to Synthesize Doxepin Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly in the initial Grignard-like addition and the final coupling steps. The process is designed to be linear and scalable, utilizing common industrial solvents such as diethyl ether, THF, and DMF. The following guide outlines the critical operational phases derived from the patent examples, emphasizing the conditions necessary to achieve the reported yields and purities. Operators should note the importance of temperature control during the exothermic addition steps and the specific stoichiometry required for the Nickel catalytic system to function optimally.
- Perform Grignard-like addition of 6,11-dihydrodibenzo[b,e]oxepin-11-one with 3-chloropropyl-tert-butyl ether using Magnesium powder.
- Execute acid-catalyzed dehydration of the alcohol intermediate using concentrated hydrochloric acid to form the olefin.
- Convert the olefin to a chloro-intermediate using thionyl chloride (SOCl2) in benzene or toluene.
- Conduct the key C-N coupling reaction between the chloro-intermediate and dimethylamine using Ni(OAc)2 and PPh3 catalyst.
- Finalize the synthesis by forming the hydrochloride salt through reaction with concentrated hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Nickel-catalyzed route offers compelling economic arguments beyond simple raw material pricing. The shift from noble metals to base metals fundamentally alters the cost structure of Doxepin manufacturing. By eliminating the need for expensive Palladium or Rhodium complexes, the direct material cost is significantly reduced. Moreover, the recyclability of the Nickel catalyst means that the effective cost per kilogram of product decreases further with scale, as the catalyst inventory does not need to be replenished at the same rate as consumable reagents. This creates a more predictable and stable cost model for long-term supply contracts.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with Nickel acetate results in substantial cost savings on reagent procurement. Additionally, the simplified workup procedure, which avoids complex heavy metal scavenging steps, reduces the consumption of auxiliary chemicals and processing time. The high selectivity of the reaction minimizes waste generation, leading to lower disposal costs and improved overall process mass intensity. These factors combine to offer a highly competitive price point for the final API intermediate without sacrificing quality.
- Enhanced Supply Chain Reliability: Nickel and triphenylphosphine are commodity chemicals with robust global supply chains, unlike specialty noble metal catalysts which can be subject to supply disruptions. This availability ensures continuity of supply for large-scale production runs. The simplicity of the reaction conditions also means that the process can be transferred between manufacturing sites with minimal requalification effort, providing flexibility in sourcing and reducing the risk of single-point failures in the supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-liter reactors with consistent results. The use of less corrosive reagents and the ability to recycle catalysts align with increasingly strict environmental regulations regarding heavy metal discharge. This 'green' profile facilitates easier permitting for new production lines and enhances the corporate sustainability credentials of the manufacturer, which is a growing requirement for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational details highlighted in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement teams negotiating supply agreements based on this technology.
Q: Why is the Nickel-catalyzed method superior to Palladium for Doxepin synthesis?
A: The Nickel-catalyzed method (Ni(OAc)2/PPh3) offers a significant cost advantage over traditional Palladium, Platinum, or Ruthenium catalysts. Furthermore, the Nickel catalyst can be easily recovered and recycled via simple filtration, reducing heavy metal residue concerns and lowering overall production costs while maintaining high selectivity (>99%).
Q: What is the overall yield and purity profile of this synthesis route?
A: According to patent CN102924424A, the optimized five-step process achieves a total yield of approximately 62%. Critical steps, particularly the C-N coupling, demonstrate exceptional selectivity exceeding 99%, ensuring a high-purity final product suitable for pharmaceutical applications with minimal impurity burden.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like DMF, benzene, and ether, and operates under manageable conditions (e.g., 35-45°C for coupling). The elimination of expensive noble metals and the simplicity of workup procedures (filtration, recrystallization) make it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxepin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the Nickel-catalyzed route for Doxepin Hydrochloride. As a premier CDMO partner, we possess the technical expertise to translate such patent innovations into commercial reality. Our facilities are equipped to handle complex organic syntheses, ranging from early-stage process development to full-scale commercial manufacturing. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for Doxepin Hydrochloride and related antidepressant intermediates. By leveraging our capabilities in cost reduction in pharmaceutical intermediate manufacturing, we can help you achieve significant efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this novel chemistry can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
