Scalable Production of High-Purity (1R)-5-Bromo-Isoindole for Advanced Quinolone Antibiotics
Scalable Production of High-Purity (1R)-5-Bromo-Isoindole for Advanced Quinolone Antibiotics
The pharmaceutical landscape for quinolone antibiotics continues to evolve, driven by the persistent demand for safer and more effective antimicrobial agents like Gatifloxacin. A critical bottleneck in the supply chain for this next-generation drug has historically been the efficient synthesis of its key chiral intermediate, (1R)-5-bromo-2,3-dihydro-1-methyl-1H-isoindole. Recent intellectual property developments, specifically patent CN110938028B, have introduced a transformative three-step synthetic route that addresses long-standing issues regarding cost, purity, and operational safety. This technical insight report analyzes the mechanistic advantages of this novel preparation method, offering R&D and procurement leaders a clear pathway toward optimizing their API manufacturing strategies. By shifting away from hazardous reagents and complex multi-step sequences, this innovation represents a significant leap forward in process chemistry for fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoindoline derivatives has been plagued by inefficient routes that rely on expensive starting materials and harsh reaction conditions. For instance, literature methods such as those disclosed in Organic Letters (2018) utilize 3-bromo-1-phenylethylamine, which itself requires complex synthesis, and necessitate the use of approximately 12 equivalents of sodium borohydride for reduction. This excessive consumption of reducing agents not only inflates raw material costs but also generates substantial chemical waste, complicating downstream purification and environmental compliance. Other prior art routes, such as those starting from phthalic anhydride or p-bromoacetophenone, suffer from messy reaction systems, difficult separation of isomers, and the need for specialized high-pressure equipment that limits scalability. These conventional pathways often result in low overall yields and inconsistent optical purity, creating significant risks for supply chain continuity in the production of high-value antibiotics.
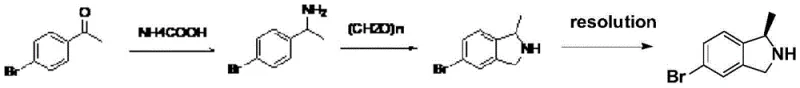
The Novel Approach
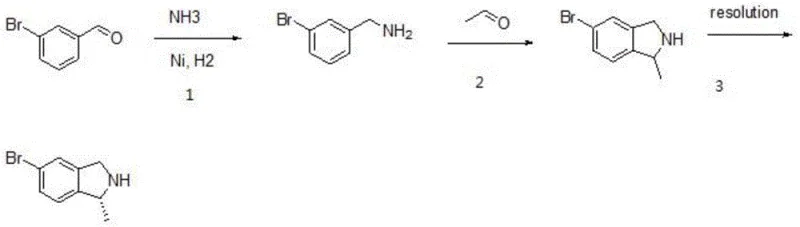
In stark contrast to these cumbersome legacy processes, the method described in CN110938028B employs a streamlined three-step sequence starting from the readily available and cost-effective 3-bromobenzaldehyde. The process initiates with a catalytic reductive amination under mild hydrogen pressure (1-4 atm) and moderate temperatures (10-50°C), achieving yields as high as 98.2% without the need for stoichiometric metal hydrides. The subsequent cyclization with acetaldehyde proceeds under reflux in an acidic medium, followed by a robust chiral resolution step using agents like L-(+)-tartaric acid. This approach eliminates the need for dangerous polybrominated precursors and simplifies the purification workflow, resulting in a final product with HPLC purity exceeding 99.5%. The operational simplicity and high atom economy of this route make it uniquely suited for industrial-scale manufacturing.
Mechanistic Insights into Catalytic Reductive Amination and Cyclization
The core of this technological breakthrough lies in the efficiency of the initial reductive amination step, where 3-bromobenzaldehyde reacts with ammonia water in the presence of a transition metal catalyst such as Raney nickel or palladium on carbon. Under a hydrogen atmosphere, the aldehyde functionality is converted directly to the primary amine (m-bromobenzylamine) with exceptional selectivity, avoiding the over-reduction or side reactions common in non-catalytic methods. This catalytic cycle operates effectively at low pressures (e.g., 2 atm) and temperatures around 30°C, minimizing energy consumption and thermal degradation of the sensitive bromo-aromatic substrate. The use of aqueous ammonia further simplifies the reaction medium, allowing for easy filtration of the catalyst and concentration of the crude amine product with minimal solvent waste.
Following the formation of the benzylamine intermediate, the mechanism shifts to an acid-catalyzed condensation and cyclization with acetaldehyde. By cooling the reaction mixture to -20°C before the dropwise addition of inorganic acid, the process controls the exothermic nature of the salt formation, ensuring safety and reproducibility. Heating the mixture to reflux facilitates the nucleophilic attack of the amine on the aldehyde carbonyl, followed by intramolecular electrophilic aromatic substitution to close the five-membered isoindoline ring. The final resolution step leverages the differential solubility of diastereomeric salts formed between the racemic base and chiral acids like camphorsulfonic acid. This thermodynamic control allows for the precise isolation of the desired (1R) enantiomer, crucial for the biological activity of the final Gatifloxacin API, while the mother liquor can potentially be recycled to improve overall mass balance.
How to Synthesize (1R)-5-Bromo-2,3-Dihydro-1-Methyl-1H-Isoindole Efficiently
Implementing this synthesis requires careful attention to the specific parameters outlined in the patent examples to maximize yield and optical purity. The process is designed to be robust, utilizing standard reactor equipment found in most fine chemical facilities, thereby reducing the barrier to entry for contract manufacturing organizations. Operators must strictly adhere to the temperature profiles during the acid addition and resolution phases to prevent racemization or oiling out of intermediates. The detailed标准化 synthesis steps provided below outline the precise molar ratios, solvent choices, and workup procedures necessary to replicate the high success rates reported in the patent data.
- Perform reductive amination of 3-bromobenzaldehyde with ammonia water and a metal catalyst (Ni/Pd) under hydrogen pressure (1-4 atm) at 10-50°C to obtain m-bromobenzylamine.
- React the resulting amine with acetaldehyde in the presence of inorganic acid (HCl/HBr) at reflux temperature for 5-15 hours to form the racemic isoindoline intermediate.
- Execute chiral resolution using a resolving agent like L-(+)-tartaric acid or camphorsulfonic acid at 40-80°C, followed by pH adjustment and crystallization to isolate the (1R) enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. By replacing expensive and hazardous reagents with commodity chemicals, the variable cost of goods sold (COGS) is drastically reduced, enhancing the margin profile for the final antibiotic product. Furthermore, the elimination of complex purification steps and the use of stable, shelf-stable starting materials mitigate the risk of production delays caused by raw material shortages or quality failures. This process stability translates directly into more reliable delivery schedules for downstream API manufacturers, ensuring a continuous supply of critical medication components.
- Cost Reduction in Manufacturing: The substitution of stoichiometric reducing agents like sodium borohydride with catalytic hydrogenation fundamentally alters the cost structure of the synthesis. This shift eliminates the expense associated with purchasing large quantities of hydride reagents and the subsequent costs of treating boron-containing waste streams. Additionally, the high yield of the first step (over 98%) ensures that the expensive brominated carbon skeleton is preserved throughout the process, minimizing material loss and maximizing the return on investment for every kilogram of starting aldehyde purchased.
- Enhanced Supply Chain Reliability: The reliance on 3-bromobenzaldehyde as the primary feedstock is a strategic advantage, as this chemical is a widely produced bulk intermediate with a mature global supply network. Unlike specialized precursors that may be sourced from single suppliers, 3-bromobenzaldehyde is available from multiple vendors, reducing the risk of supply disruption. The simplified three-step process also shortens the overall manufacturing lead time, allowing for faster response to market demand fluctuations and reducing the inventory capital tied up in work-in-progress materials.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method significantly lowers the environmental footprint of production. The avoidance of harsh bromination steps and the reduction of heavy metal waste simplify the wastewater treatment requirements, facilitating easier regulatory compliance in jurisdictions with strict discharge limits. The mild reaction conditions (low pressure and temperature) also reduce the energy load on the facility and lower the operational risk profile, making the process safer to scale from pilot plant to multi-ton commercial production without requiring extensive capital investment in specialized high-pressure reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of the synthesis. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: Why is the new synthesis route for (1R)-5-bromo-2,3-dihydro-1-methyl-1H-isoindole considered superior to prior art?
A: Unlike conventional methods that require expensive reagents like excessive sodium borohydride or harsh bromination conditions, this patent utilizes a mild catalytic hydrogenation step and readily available 3-bromobenzaldehyde, significantly reducing raw material costs and waste generation while improving overall yield and purity.
Q: What are the critical reaction conditions for the cyclization step in this process?
A: The cyclization step involves dissolving the intermediate amine in a solvent, cooling to -20°C, adding inorganic acid, and reacting with acetaldehyde at reflux temperature for 5-15 hours. This controlled acidic environment ensures efficient ring closure with minimal byproduct formation compared to traditional high-pressure or high-temperature methods.
Q: How does this manufacturing process impact the supply chain stability for Gatifloxacin production?
A: By utilizing 3-bromobenzaldehyde, a commercially abundant and stable starting material, and eliminating the need for specialized high-pressure equipment or difficult-to-separate intermediates, this method enhances supply chain reliability. The simplified three-step sequence allows for easier commercial scale-up and consistent batch-to-batch quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R)-5-Bromo-2,3-Dihydro-1-Methyl-1H-Isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceuticals depends on the reliability and quality of the underlying intermediate supply. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of market volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including the high optical purity required for chiral intermediates like (1R)-5-bromo-2,3-dihydro-1-methyl-1H-isoindole. We are committed to leveraging innovative processes like CN110938028B to deliver value-driven solutions that align with your cost and quality objectives.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with high-performance chemical intermediates.
