Revolutionizing Dolutegravir Intermediate Production via Green Carbonylation and Reductive Amination
Introduction to Advanced Synthetic Routes for Antiretroviral Intermediates
The global demand for high-efficiency antiretroviral therapies continues to drive innovation in the synthesis of key pharmaceutical intermediates, specifically for integrase inhibitors like dolutegravir. Patent CN108752217B, published in August 2021, introduces a transformative methodology for producing 2,4-difluorobenzylamine, a critical building block in this therapeutic class. This patent addresses long-standing industry challenges by proposing a concise, green, and cost-effective production method that diverges significantly from traditional, hazard-prone synthetic pathways. By leveraging advanced carbonylation techniques and optimized reductive amination protocols, this technology offers a robust alternative that aligns with modern principles of green chemistry and sustainable manufacturing. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable supply chain for next-generation HIV treatments while mitigating regulatory and environmental risks associated with older chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,4-difluorobenzylamine has relied on routes that are increasingly untenable due to environmental regulations and safety concerns. One prevalent method involves starting from 2,4-dichlorobenzonitrile, which undergoes halogen exchange and subsequent reduction. As illustrated in the reaction scheme below, this pathway often suffers from difficult raw material sourcing and suboptimal fluorine yields, leading to significant economic inefficiencies.
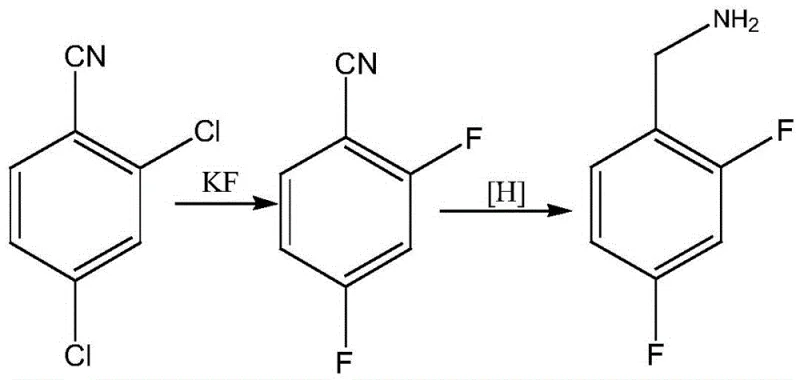
Furthermore, alternative approaches utilizing m-difluorobenzene via chloromethylation followed by urotropine condensation present severe ecological drawbacks. This traditional route, depicted in the following diagram, generates substantial quantities of waste acid and high-salinity wastewater containing ammonia nitrogen, creating a heavy burden on waste treatment facilities and increasing the overall carbon footprint of the manufacturing process.
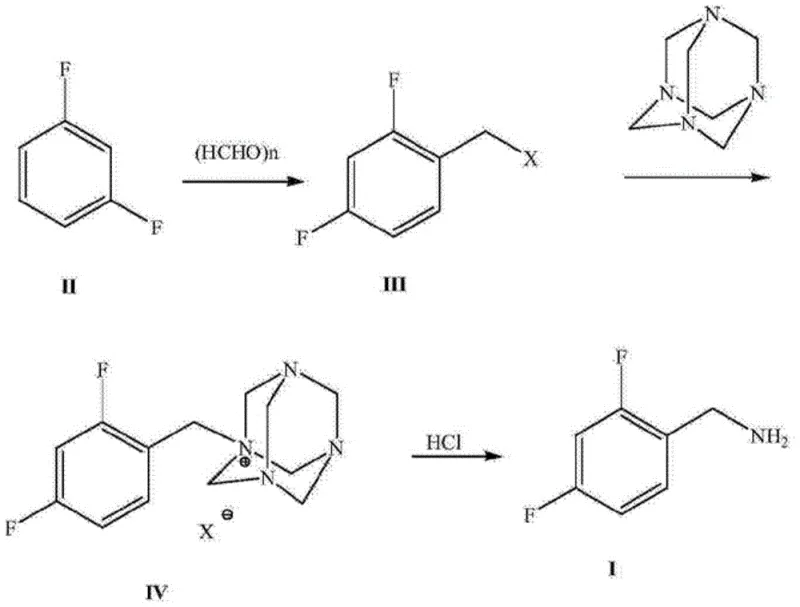
Additionally, direct reduction methods starting from 2,4-difluorobenzonitrile often employ hazardous reducing agents such as lithium aluminum hydride or borane complexes. These reagents pose significant safety hazards regarding thermal runaway and require stringent handling protocols, while also driving up operational costs due to the need for specialized equipment and extensive safety measures. The cumulative effect of these limitations is a supply chain that is fragile, expensive, and environmentally unsustainable.
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN108752217B proposes a streamlined two-step strategy that fundamentally reshapes the production landscape. The core innovation lies in the direct formylation of m-difluorobenzene, a readily available and inexpensive commodity chemical, to generate 2,4-difluorobenzaldehyde. This can be achieved either through a catalytic carbonylation process using carbon monoxide or via a modified Reimer-Tiemann reaction using chloroform and a strong base. This shift eliminates the need for hazardous nitrile precursors and avoids the massive salt waste associated with chloromethylation, marking a significant leap forward in process efficiency and environmental stewardship.
Mechanistic Insights into Cobalt-Catalyzed Formylation and Reductive Amination
The mechanistic elegance of this new route is centered on the efficient conversion of m-difluorobenzene into the corresponding aldehyde. In the carbonylation variant, the process utilizes cobalt catalysts such as Co2(CO)8 or Na[Co(CO)4] under moderate pressure conditions. The cobalt species facilitates the insertion of a carbonyl group into the aromatic ring, likely proceeding through a coordination complex that activates the C-H bond ortho to the fluorine substituents. The presence of Lewis acids like AlCl3 further enhances the electrophilicity of the system, ensuring high regioselectivity for the 2,4-substitution pattern required for dolutegravir synthesis. Alternatively, the Reimer-Tiemann variation employs dichlorocarbene generated in situ from chloroform and potassium tert-butoxide, which attacks the electron-rich aromatic ring followed by hydrolysis to yield the aldehyde. Both pathways demonstrate superior atom economy compared to nucleophilic aromatic substitution routes.
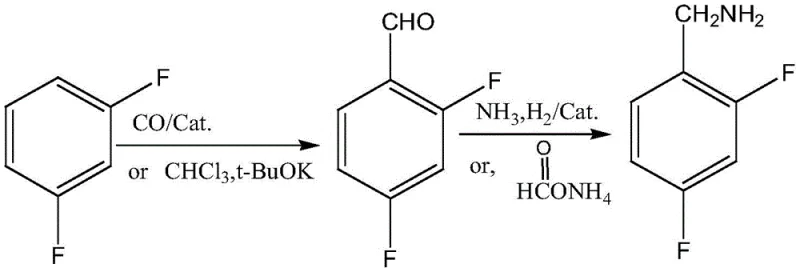
Following the formation of 2,4-difluorobenzaldehyde, the second critical stage involves reductive amination to install the primary amine functionality. This step is meticulously optimized to prevent over-reduction or defluorination, common side reactions in fluorinated aromatic systems. The patent details the use of heterogeneous catalysts like Raney nickel or Pd/C under an atmosphere of ammonia and hydrogen. The ammonia serves to protect the aldehyde as an imine intermediate, which is then selectively reduced to the amine. Crucially, the reaction conditions—specifically the controlled pressures of ammonia (0.2-0.5 MPa) and hydrogen (0.5-1.5 MPa)—are tuned to maximize yield while maintaining the integrity of the sensitive carbon-fluorine bonds. An alternative transfer hydrogenation method using ammonium formate is also described, offering a safer option for facilities without high-pressure hydrogen infrastructure, thereby broadening the technological applicability of this synthesis.
How to Synthesize 2,4-Difluorobenzylamine Efficiently
The practical implementation of this synthesis requires precise control over reaction parameters to ensure reproducibility and high purity. The process begins with the activation of m-difluorobenzene, followed by the careful introduction of the formylating agent, whether it be CO gas or the chloroform/base system. Subsequent workup involves standard extraction and distillation techniques to isolate the aldehyde intermediate, which is then subjected to the amination protocol. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and pressure settings derived from the patent examples, are outlined in the guide below.
- Formylation of m-difluorobenzene using CO/Co-catalyst or Reimer-Tiemann reaction to yield 2,4-difluorobenzaldehyde.
- Reductive amination of the aldehyde using ammonia/hydrogen with Raney Ni or Pd/C, or transfer hydrogenation with ammonium formate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers compelling strategic advantages that extend beyond mere technical feasibility. The shift towards using m-difluorobenzene as a starting material represents a significant optimization of the raw material basket, moving away from specialized, hard-to-source nitriles toward bulk commodities that are widely available in the global chemical market. This transition inherently stabilizes the supply chain against fluctuations in niche precursor availability and reduces the risk of production stoppages due to raw material shortages. Furthermore, the simplification of the synthetic route from multi-step sequences to a concise two-step process drastically reduces the overall manufacturing cycle time, allowing for faster response to market demand surges for dolutegravir.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reducing agents like lithium aluminum hydride, coupled with the use of cost-effective cobalt catalysts and commodity solvents, leads to a substantial decrease in direct material costs. Additionally, the avoidance of complex waste treatment procedures required for high-salt wastewater significantly lowers operational expenditures related to environmental compliance and disposal. The high yields reported in the patent examples, reaching up to 85 percent, further enhance the economic viability by maximizing output per unit of input, ensuring that every kilogram of raw material contributes effectively to the final product value.
- Enhanced Supply Chain Reliability: By relying on robust chemical transformations that tolerate standard industrial equipment, this method reduces the dependency on specialized, high-maintenance reactors often needed for cryogenic or ultra-high-pressure reactions. The ability to use transfer hydrogenation with ammonium formate provides a flexible backup strategy, ensuring continuity of supply even if hydrogen infrastructure is temporarily compromised. This resilience is critical for maintaining the uninterrupted production of life-saving antiretroviral medications, where supply interruptions can have severe consequences for public health outcomes.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by lower waste generation and the absence of heavy metal contaminants in the final product, simplifies the regulatory approval process for new drug master files. The process is inherently scalable, as demonstrated by the patent's emphasis on industrial applicability, allowing manufacturers to ramp up production from pilot scale to multi-ton commercial volumes without encountering the bottlenecks typical of batch processes involving unstable intermediates. This scalability ensures that the supply of 2,4-difluorobenzylamine can grow in tandem with the expanding global market for HIV integrase inhibitors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear framework for evaluating the technology's fit within existing manufacturing portfolios. Understanding these details is crucial for technical teams assessing the feasibility of technology transfer and for commercial teams negotiating licensing or supply agreements.
Q: What are the primary advantages of the new synthesis route over traditional chloromethylation?
A: The new route avoids the generation of large amounts of waste acid, high ammonia nitrogen, and saline wastewater associated with chloromethylation, significantly improving environmental compliance and reducing waste treatment costs.
Q: Which catalysts are preferred for the carbonylation step in this patent?
A: The patent specifies Co2(CO)8 or Na[Co(CO)4] as effective catalysts, with Na[Co(CO)4] being particularly preferred for its efficiency in converting m-difluorobenzene to the aldehyde intermediate.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes readily available raw materials like m-difluorobenzene and operates under manageable pressure conditions, making it highly scalable and economically viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Difluorobenzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of advanced pharmaceutical therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering 2,4-difluorobenzylamine with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and assay. Our capability to implement the green synthesis routes described in CN108752217B positions us as a forward-thinking partner capable of meeting the evolving sustainability goals of our global clientele.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this optimized supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply of this vital dolutegravir intermediate is secure, compliant, and cost-effective for the long term.
