Advanced Manufacturing of 2-Amino-5-Fluorobenzonitrile via Optimized Three-Step Route
The pharmaceutical industry continuously demands more efficient and cost-effective pathways for synthesizing key intermediates, particularly fluorinated aromatic compounds which are ubiquitous in modern drug design. Patent CN113354558B, published in early 2022, introduces a groundbreaking preparation method for 2-amino-5-fluorobenzonitrile, a critical building block for various bioactive molecules. This innovation addresses significant bottlenecks found in prior art by streamlining the synthetic sequence from four steps down to just three, thereby enhancing overall throughput and reducing operational complexity. By utilizing 2,5-difluorobenzaldehyde as a readily accessible starting material, the process leverages a robust condensation-dehydration-aminolysis strategy that avoids the use of precious metal catalysts. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a substantial leap forward in process chemistry, offering a pathway that is not only chemically elegant but also commercially viable for multi-ton production scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-5-fluorobenzonitrile has been plagued by inefficiencies inherent in older synthetic strategies, such as the route disclosed in WO2006/129120. As illustrated in the reaction scheme below, the conventional approach typically necessitates a four-step sequence that culminates in a palladium-carbon catalyzed hydrogenation step. This reliance on heterogeneous hydrogenation introduces severe logistical and safety challenges, including the requirement for high-pressure reactors and the handling of pyrophoric catalysts which pose significant fire hazards in large-scale manufacturing environments. Furthermore, the removal of trace palladium residues to meet stringent pharmaceutical purity specifications often requires additional purification steps, such as specialized filtration or scavenging treatments, which drastically increase both processing time and production costs. The cumulative yield of such multi-step processes often suffers due to material losses at each isolation stage, making the final product economically unattractive for cost-sensitive applications.
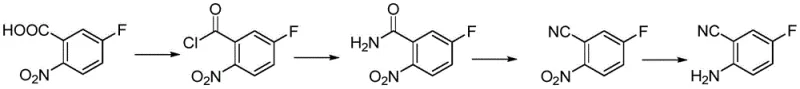
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach detailed in the patent data utilizes a concise three-step trajectory that begins with the condensation of 2,5-difluorobenzaldehyde. This streamlined pathway, depicted in the following figure, effectively bypasses the need for reduction chemistry entirely by constructing the nitrile functionality through dehydration of an oxime intermediate. The elimination of the hydrogenation step not only removes the safety risks associated with high-pressure hydrogen gas but also eradicates the need for expensive palladium catalysts, leading to immediate cost reduction in pharma manufacturing. The process operates under mild thermal conditions, with reaction temperatures ranging from 20°C to 80°C, which significantly lowers energy consumption compared to high-temperature or high-pressure alternatives. By simplifying the workflow and utilizing common organic solvents like dichloromethane and DMF, this method offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates with enhanced reliability.
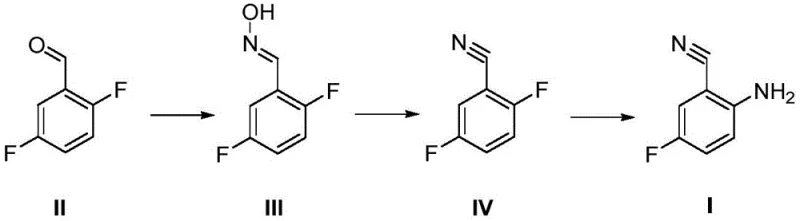
Mechanistic Insights into Oxime Formation and Dehydration
The core of this synthetic innovation lies in the efficient transformation of the aldehyde group into a nitrile via an oxime intermediate, followed by a selective nucleophilic substitution. In the first stage, 2,5-difluorobenzaldehyde undergoes a condensation reaction with hydroxylamine hydrochloride in the presence of triethylamine, which acts as a base to scavenge the generated hydrochloric acid. This step proceeds rapidly at ambient temperatures (20-40°C) to form the corresponding oxime (Compound III) with exceptional conversion rates, often exceeding 95% yield as demonstrated in the experimental examples. The subsequent dehydration step employs phosphorus oxychloride (POCl3) in N,N-dimethylformamide (DMF), a classic Vilsmeier-Haack type activation environment that facilitates the elimination of water to generate the nitrile group (Compound IV). This dehydration is critical as it establishes the carbon-nitrogen triple bond without affecting the sensitive fluorine substituents on the aromatic ring, ensuring high regioselectivity and minimizing the formation of defluorinated byproducts.
Following the formation of the nitrile, the final aminolysis step introduces the amino group ortho to the nitrile functionality through a nucleophilic aromatic substitution mechanism. By introducing ammonia gas into a solution of the nitrile intermediate in polar aprotic solvents like DMF or DMSO, the fluorine atom at the 2-position is displaced by the amine nucleophile. This reaction is highly dependent on the electron-withdrawing nature of the adjacent nitrile and nitro-like activation (though here it is the nitrile and the remaining fluorine), which activates the ring towards nucleophilic attack. The process is carefully controlled at temperatures between 20°C and 40°C to prevent over-reaction or polymerization, resulting in the final 2-amino-5-fluorobenzonitrile product with high purity. Impurity control is inherently built into this mechanism, as the mild conditions avoid the harsh reducing environments that often lead to side-reactions such as the reduction of the nitrile group to an amine or the formation of azo-compounds.
How to Synthesize 2-Amino-5-Fluorobenzonitrile Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The process begins with the preparation of the oxime, where maintaining the molar ratio of aldehyde to hydroxylamine hydrochloride at approximately 1:1.2 ensures complete consumption of the starting material while minimizing excess reagent waste. Following isolation, the dehydration step must be monitored closely, typically requiring 2 to 6 hours at elevated temperatures to drive the equilibrium towards the nitrile product. The final aminolysis is a gas-liquid reaction that benefits from efficient mixing to ensure adequate mass transfer of ammonia into the solvent phase. For detailed operational protocols, safety data, and specific workup procedures required for GMP compliance, please refer to the standardized synthesis guide provided below.
- Condense 2,5-difluorobenzaldehyde with hydroxylamine hydrochloride using triethylamine in dichloromethane at 20-40°C to form the oxime intermediate.
- Dehydrate the oxime intermediate using phosphorus oxychloride in N,N-dimethylformamide at 40-80°C to generate the nitrile compound.
- Perform aminolysis on the nitrile compound by introducing ammonia gas in DMF or DMSO at 20-40°C to yield the final 2-amino-5-fluorobenzonitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages that extend beyond simple chemical yield improvements. By eliminating the dependency on palladium catalysts, manufacturers can achieve significant cost reduction in manufacturing, as the expense of precious metals and the associated recovery or disposal costs are completely removed from the balance sheet. Furthermore, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure requirements for production facilities, allowing for greater flexibility in choosing manufacturing sites that may not possess specialized high-pressure infrastructure. The use of commodity chemicals like 2,5-difluorobenzaldehyde ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or constrained starting materials that often plague the fine chemical sector.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the unit operations and the removal of expensive catalytic systems. Without the need for palladium on carbon, the downstream processing becomes significantly less complex, as there is no requirement for rigorous heavy metal testing or specialized filtration steps to meet regulatory limits. Additionally, the shorter reaction sequence reduces the total man-hours and utility consumption per kilogram of product, leading to substantial cost savings that can be passed down to the customer or reinvested into further process optimization. The high yield of each individual step, consistently reported above 90%, ensures that raw material utilization is maximized, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable pharmaceutical intermediate supplier, and this route excels by utilizing robust and widely available reagents. The starting material, 2,5-difluorobenzaldehyde, is a bulk chemical produced by multiple global vendors, reducing the risk of single-source supply disruptions. Moreover, the reaction conditions are mild and do not require extreme temperatures or pressures, which minimizes the risk of unplanned shutdowns due to equipment failure or safety incidents. This operational stability allows for predictable production scheduling and shorter lead times for high-purity pharmaceutical intermediates, enabling customers to maintain leaner inventory levels without compromising their own production timelines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for seamless transition from pilot plant to full commercial production. The solvents used, such as dichloromethane and DMF, are standard in the industry with well-established recovery and recycling protocols, facilitating effective waste management and reducing the environmental footprint. The absence of hydrogen gas eliminates the need for complex safety permits and explosion-proof zones, simplifying the regulatory approval process for new production lines. This ease of scale-up ensures that the technology can support growing market demand, from initial clinical trial batches of 100 kgs to commercial volumes exceeding 100 MT annually, without the need for fundamental process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding the process efficiency, safety profile, and quality attributes of the final product. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: How does this new method improve upon the traditional WO2006/129120 synthesis route?
A: The traditional route involves four steps and utilizes palladium-carbon hydrogenation, which requires expensive catalysts and high-pressure equipment. The new method shortens the process to three steps, eliminates the need for hazardous hydrogenation, and achieves a total yield of approximately 83% with individual step yields exceeding 90%.
Q: What are the critical reaction conditions for the dehydration step?
A: The dehydration step utilizes phosphorus oxychloride (POCl3) as the dehydrating agent in N,N-dimethylformamide (DMF). The reaction is conducted at temperatures between 40°C and 80°C for 2 to 6 hours, ensuring complete conversion of the oxime intermediate to the nitrile without significant degradation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It avoids high-pressure hydrogenation and uses readily available raw materials like 2,5-difluorobenzaldehyde. The mild reaction temperatures (20-80°C) and standard solvent systems (DCM, DMF, DMSO) make it ideal for commercial scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Fluorobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new pharmaceutical agents depends heavily on the reliability and quality of the underlying intermediate supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-amino-5-fluorobenzonitrile meets the highest industry standards for identity, assay, and impurity profiles. Our commitment to technical excellence allows us to offer a level of supply security and quality consistency that is essential for maintaining your own regulatory filings and production schedules.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your needs, ensuring that your partnership with us is built on a foundation of transparency, technical expertise, and mutual success.
