Advanced Green Synthesis of N-Methyl Heterocycles for Scalable Pharmaceutical Manufacturing
Advanced Green Synthesis of N-Methyl Heterocycles for Scalable Pharmaceutical Manufacturing
The global pharmaceutical and fine chemical industries are increasingly prioritizing sustainable manufacturing processes that minimize environmental impact while maintaining high efficiency and product purity. A pivotal advancement in this domain is detailed in patent CN112321480B, which discloses a novel methylation synthesis method for N-heterocyclic compounds. This technology represents a significant departure from traditional alkylation methods by utilizing dimethyl carbonate (DMC) as a green methylating agent under pressurized conditions. The innovation addresses critical pain points regarding toxicity, waste generation, and operational complexity that have long plagued the production of key intermediates like 1-methyl-1H-indole and its derivatives. By leveraging this proprietary methodology, manufacturers can achieve reaction conversion rates exceeding 98% while adhering to stricter environmental, health, and safety (EHS) standards. This report provides a deep technical and commercial analysis of this process, highlighting its viability for reliable pharmaceutical intermediate suppliers seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the N-methylation of heterocyclic scaffolds has relied heavily on hazardous reagents such as methyl iodide (MeI) and dimethyl sulfate (DMS). These traditional methylating agents, while effective, pose severe safety and environmental challenges that complicate large-scale manufacturing. Methyl iodide is a potent alkylating agent with high volatility and significant toxicity, requiring stringent containment measures and specialized personal protective equipment to prevent worker exposure. Furthermore, dimethyl sulfate is classified as a carcinogen and is known to methylate human DNA, presenting unacceptable risks in modern production facilities. Beyond safety concerns, the use of these reagents often necessitates complex workup procedures involving alkaline hydrolysis and pH adjustments to isolate the final N-methylated products. These additional steps increase processing time, generate substantial saline wastewater, and elevate the overall cost of goods sold (COGS). Consequently, reliance on these legacy chemistries creates supply chain vulnerabilities due to increasing regulatory scrutiny and the rising cost of compliance.
The Novel Approach
In stark contrast, the method described in patent CN112321480B utilizes dimethyl carbonate, a reagent renowned for its low toxicity and biodegradability, effectively eliminating the hazards associated with halogenated methylating agents. This novel approach operates under elevated pressure and temperature conditions, typically ranging from 80-250°C and 0.3-8.0 MPa, which drives the reaction kinetics to completion without the need for harsh bases or complex solvent systems. The process is characterized by its exceptional selectivity, producing target molecules such as 1,3,3-trimethyl-2-methylene indoline with minimal byproduct formation. 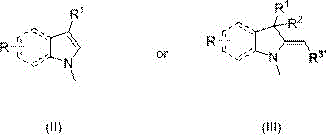 The simplicity of the operation allows for the direct recovery of unreacted dimethyl carbonate and the methanol byproduct through reduced pressure distillation, creating a closed-loop system that maximizes atom economy. This shift not only enhances the safety profile of the manufacturing site but also streamlines the purification process, resulting in a more robust and cost-effective production workflow for high-purity organic chemicals.
The simplicity of the operation allows for the direct recovery of unreacted dimethyl carbonate and the methanol byproduct through reduced pressure distillation, creating a closed-loop system that maximizes atom economy. This shift not only enhances the safety profile of the manufacturing site but also streamlines the purification process, resulting in a more robust and cost-effective production workflow for high-purity organic chemicals.
Mechanistic Insights into Dimethyl Carbonate Catalyzed Methylation
The core of this technological breakthrough lies in the activation of dimethyl carbonate under pressurized conditions to facilitate nucleophilic substitution at the nitrogen center of the heterocycle. Unlike ambient pressure reactions where DMC might act primarily as a solvent or mild reagent, the application of pressures up to 8.0 MPa ensures that the reagent remains in a supercritical or dense liquid phase, significantly increasing the collision frequency between reactant molecules. The reaction mechanism generally proceeds via a nucleophilic attack by the nitrogen lone pair of the heterocyclic substrate on the methyl carbon of the dimethyl carbonate. 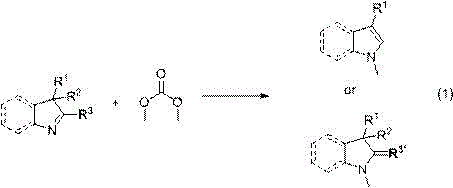 This process is often accelerated by the presence of specific catalysts, such as metal nickel, palladium, or platinum supported on carriers, or phase transfer catalysts like tetrabutylammonium salts. These catalytic systems lower the activation energy barrier, allowing the methylation to proceed efficiently even with sterically hindered substrates. The versatility of the system is evident in its tolerance for various substituents (R groups) on the heterocyclic ring, including halogens and alkoxy groups, without compromising the integrity of the sensitive functional groups.
This process is often accelerated by the presence of specific catalysts, such as metal nickel, palladium, or platinum supported on carriers, or phase transfer catalysts like tetrabutylammonium salts. These catalytic systems lower the activation energy barrier, allowing the methylation to proceed efficiently even with sterically hindered substrates. The versatility of the system is evident in its tolerance for various substituents (R groups) on the heterocyclic ring, including halogens and alkoxy groups, without compromising the integrity of the sensitive functional groups.
From an impurity control perspective, the mechanism inherently favors the formation of the N-methyl product over C-alkylation or over-methylation side reactions due to the specific electronic properties of the dimethyl carbonate molecule. The leaving group in this transformation is a methoxide ion, which subsequently captures a proton to form methanol, a benign and easily separable byproduct. This clean reaction pathway minimizes the formation of tars or polymeric impurities that are common in reactions involving methyl iodide. Furthermore, the ability to tune the reaction temperature between 100-200°C allows process chemists to optimize the balance between reaction rate and thermal stability of the product. The result is a crude reaction mixture that is significantly cleaner than those obtained from conventional methods, reducing the load on downstream purification units and ensuring consistent batch-to-batch quality.
How to Synthesize N-Methyl Heterocycles Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the sequential addition of the N-heterocyclic compound and dimethyl carbonate into a pressure-rated reaction kettle, followed by a thorough gas replacement cycle using nitrogen to remove oxygen and moisture. 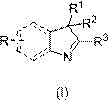 Once the inert atmosphere is established, the mixture is heated to the target temperature range while maintaining the system pressure within the specified 0.3-8.0 MPa window for a duration of 3 to 12 hours. The reaction progress is monitored until the conversion endpoint is reached, indicated by the consumption of the starting material. Following the reaction, the system is cooled and depressurized, allowing for the separation of the reaction mother liquor. The final step involves reduced pressure distillation, where the methylated product is isolated, and the valuable dimethyl carbonate and methanol are recovered for reuse.
Once the inert atmosphere is established, the mixture is heated to the target temperature range while maintaining the system pressure within the specified 0.3-8.0 MPa window for a duration of 3 to 12 hours. The reaction progress is monitored until the conversion endpoint is reached, indicated by the consumption of the starting material. Following the reaction, the system is cooled and depressurized, allowing for the separation of the reaction mother liquor. The final step involves reduced pressure distillation, where the methylated product is isolated, and the valuable dimethyl carbonate and methanol are recovered for reuse.
- Charge the reactor with the N-heterocyclic substrate and dimethyl carbonate, then perform nitrogen gas replacement to ensure an inert atmosphere.
- Heat the mixture to 80-250°C under a maintained pressure of 0.3-8.0 MPa for 3-12 hours to facilitate the methylation reaction.
- Upon completion, reduce pressure and subject the mother liquor to vacuum distillation to isolate the product and recover unreacted dimethyl carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dimethyl carbonate-based methylation technology offers transformative benefits that extend far beyond simple chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain risk profile by replacing highly regulated, hazardous raw materials with a commodity chemical. Dimethyl carbonate is produced on a massive industrial scale for the polycarbonate and battery electrolyte industries, ensuring a stable, abundant, and price-competitive supply compared to the niche and tightly controlled market for methyl iodide. This shift mitigates the risk of supply disruptions caused by regulatory crackdowns on toxic substances and reduces the administrative burden associated with handling scheduled chemicals. Additionally, the elimination of halogenated waste streams significantly lowers the cost of waste treatment and disposal, contributing to substantial overall cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high atom economy and the recyclability of the methylating agent. Since dimethyl carbonate acts as both reagent and solvent in many configurations, and unreacted material can be distilled and reused, the effective consumption of raw materials is minimized. The absence of expensive halogenated reagents and the reduction in neutralization salts (from avoiding alkaline hydrolysis steps) lead to lower raw material costs and reduced waste disposal fees. Furthermore, the high conversion rates observed in the patent examples, often exceeding 98%, mean that less starting material is lost to side reactions, directly improving the yield per batch and lowering the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: Relying on dimethyl carbonate enhances supply chain resilience because it is a bulk chemical with multiple global suppliers, unlike methyl iodide which has a limited supplier base due to its toxicity. This diversification of sourcing options prevents bottlenecks and ensures continuous production capability even during market fluctuations. The simplified logistics of transporting non-hazardous or low-hazard materials also reduce shipping costs and insurance premiums. By adopting this greener pathway, companies can future-proof their supply chains against tightening environmental regulations that may eventually ban or severely restrict the use of traditional methylating agents, ensuring long-term business continuity.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure reactor technology that is widely available in contract development and manufacturing organizations (CDMOs). The clean nature of the reaction, producing mainly methanol as a byproduct, aligns perfectly with modern green chemistry principles and corporate sustainability goals. This facilitates easier permitting for new production lines and reduces the environmental footprint of the manufacturing site. The ability to operate with excellent selectivity means that purification trains can be shorter and more energy-efficient, further supporting the goal of sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this methylation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific projects. Understanding these details is crucial for R&D teams planning technology transfers and for procurement teams assessing vendor capabilities.
Q: What are the safety advantages of using dimethyl carbonate over methyl iodide?
A: Dimethyl carbonate is significantly less toxic and non-carcinogenic compared to methyl iodide and dimethyl sulfate, reducing regulatory burdens and worker exposure risks.
Q: Can this methylation process be scaled for industrial production?
A: Yes, the process utilizes standard high-pressure reactor technology and achieves high conversion rates (over 98%), making it highly suitable for commercial scale-up.
Q: How are byproducts managed in this synthesis route?
A: The primary byproduct is methanol, and unreacted dimethyl carbonate can be efficiently recovered via distillation, minimizing waste generation and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, sustainable chemistries to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity organic chemicals that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By integrating the green methylation protocols described in CN112321480B into our manufacturing portfolio, we offer our partners a secure, compliant, and cost-effective source for essential N-heterocyclic building blocks.
We invite you to collaborate with us to leverage these technological advantages for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and reliability in your supply chain.
