Advanced Synthesis of 5-(Aryl Methylene) Michaelis Acid for Pharmaceutical Intermediates
Advanced Synthesis of 5-(Aryl Methylene) Michaelis Acid for Pharmaceutical Intermediates
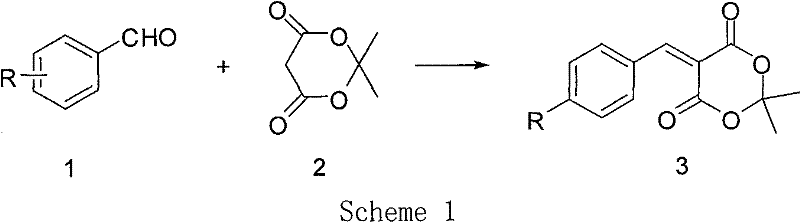
The landscape of fine chemical manufacturing is constantly evolving towards greener, more efficient methodologies, particularly for high-value intermediates used in complex heterocyclic synthesis. Patent CN101921258A introduces a significant breakthrough in the preparation of 5-(aryl methylene) Michaelis acid, also known as 5-arylidene-2,2-dimethyl-1,3-dioxane-4,6-dione. These compounds serve as highly active Michael addition acceptors and are pivotal precursors for synthesizing diverse heterocyclic structures with potential medical value through Diels-Alder reactions. The disclosed innovation addresses critical pain points in existing synthetic routes by replacing hazardous solvents and energy-intensive conditions with a benign, two-step protocol utilizing water and ethanol. This shift not only aligns with modern environmental compliance standards but also offers substantial operational advantages for commercial scale-up of complex pharmaceutical intermediates.
For R&D directors and process chemists, the structural versatility of these intermediates is paramount, as the substituent R can vary widely including hydrogen, halogens, alkyl, alkoxy, hydroxyl, nitro, and cyano groups. The ability to access these derivatives efficiently without compromising purity is a key driver for downstream drug discovery programs. The patent details a robust methodology that ensures high yields and minimizes the formation of stubborn impurities that often plague Knoevenagel condensations. By leveraging a Schiff base intermediate strategy, the process achieves a level of selectivity that direct condensation methods struggle to match, thereby simplifying the purification workflow and enhancing the overall quality of the high-purity pharmaceutical intermediates delivered to the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing 5-(aryl methylene) Michaelis acid have historically been fraught with significant technical and safety challenges that hinder efficient production. As illustrated in earlier literature such as Tetrahedron Letters (2001), direct condensation of aromatic aldehydes with Michaelis acid in water at elevated temperatures (75°C) often leads to the formation of undesirable byproducts. Specifically, this route is prone to generating compound 4, a bimolecular Michaelis acid condensation product, which is particularly prevalent when using halogen-substituted aromatic aldehydes. This impurity profile complicates downstream processing and reduces the overall yield of the desired active acceptor.
Furthermore, alternative approaches documented in Tetrahedron Letters (2007) attempted to mitigate these issues by employing dry benzene as a solvent under the catalysis of pyrrolidinium acetate. While this method improved selectivity to some extent, it introduced severe safety and environmental liabilities. Benzene is classified as a Class 1 solvent by the ICH due to its unacceptable toxicity and carcinogenicity, posing significant risks to operator health and requiring stringent containment measures. Additionally, the reliance on specific catalysts and extended reaction times of up to 24 hours at room temperature creates bottlenecks in production throughput, making these conventional methods less viable for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative strategy presented in patent CN101921258A fundamentally reimagines the synthetic pathway by decoupling the condensation into two distinct, highly controlled steps. Instead of forcing the aldehyde and Michaelis acid to react directly under harsh conditions, the process first converts the aromatic aldehyde into an arylidene aniline (Schiff base) using water as the sole solvent. This intermediate is then reacted with Michaelis acid in ethanol. This sequential approach effectively bypasses the formation of the troublesome bimolecular condensation impurity. The reaction conditions are remarkably mild, proceeding at room temperature with reaction times as short as 3 to 4 minutes for the final step.
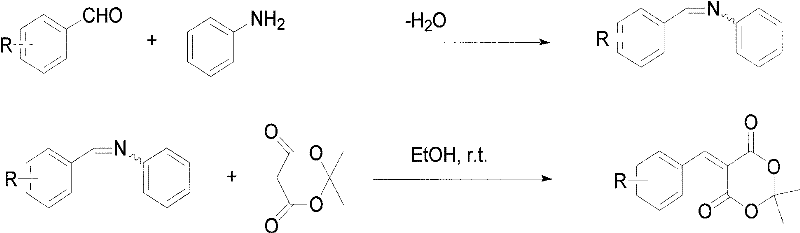
This novel route offers a compelling value proposition for supply chain leaders seeking reliable pharmaceutical intermediates supplier partnerships. By eliminating the need for toxic benzene and expensive catalysts, the process drastically simplifies the regulatory burden associated with solvent residuals and heavy metal clearance. The use of water and ethanol, both Class 3 solvents with low toxic potential, ensures that the final product meets stringent purity specifications with minimal effort. The rapid reaction kinetics observed in the second step allow for high throughput, enabling manufacturers to respond quickly to market demands while maintaining a smaller physical footprint for reaction vessels, thus optimizing capital expenditure.
Mechanistic Insights into Schiff Base Mediated Condensation
The core mechanistic advantage of this process lies in the modulation of electrophilicity through the formation of the arylidene aniline intermediate. In the first step, the aromatic aldehyde reacts with aniline in an aqueous medium to form the Schiff base. This transformation is driven by the nucleophilic attack of the amine nitrogen on the carbonyl carbon of the aldehyde, followed by dehydration. The use of water as a solvent here is counter-intuitive for imine formation but works effectively due to the insolubility of the resulting Schiff base, which precipitates out of the solution, driving the equilibrium forward according to Le Chatelier's principle. This step effectively 'masks' the aldehyde functionality, preventing it from undergoing uncontrolled self-condensation or reacting prematurely with the Michaelis acid in a non-selective manner.
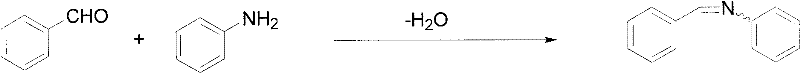
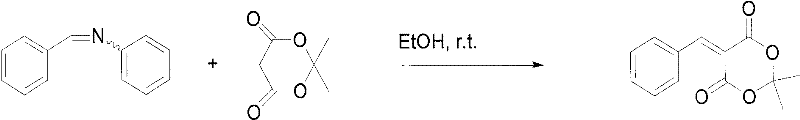
In the second mechanistic phase, the pre-formed arylidene aniline acts as a stabilized electrophile that reacts rapidly with the enol form of Michaelis acid. When the saturated ethanol solutions of the Schiff base and Michaelis acid are mixed, a swift Knoevenagel-type condensation occurs. The aniline moiety serves as an excellent leaving group in this context, facilitating the formation of the carbon-carbon double bond characteristic of the 5-(aryl methylene) Michaelis acid structure. The extreme speed of this reaction (3-4 minutes) is critical; it ensures that the concentration of reactive species remains low enough to prevent the dimerization of Michaelis acid, which is the root cause of the bimolecular impurity seen in direct methods. This precise kinetic control is what allows for the isolation of high-purity products simply by filtration and recrystallization.
How to Synthesize 5-(Aryl Methylene) Michaelis Acid Efficiently
The operational simplicity of this synthesis makes it an ideal candidate for technology transfer and scale-up. The process relies on standard unit operations such as dissolution, stirring, filtration, and recrystallization, requiring no specialized high-pressure or cryogenic equipment. The key to success lies in the preparation of saturated solutions for the second step, ensuring that the reactants are brought together in a state that promotes immediate precipitation of the product upon reaction. This 'precipitation-driven' synthesis not only simplifies isolation but also inherently purifies the material, as impurities tend to remain in the mother liquor. For detailed procedural specifics regarding stoichiometry and workup, please refer to the standardized guide below.
- Dissolve aromatic aldehyde and aniline in water at a molar ratio of 1.0: (1.0-1.2) and react at room temperature for 1-2 hours to form arylidene aniline.
- Prepare saturated ethanol solutions of the resulting arylidene aniline and Michaelis acid separately.
- Mix the saturated solutions, stir for 3-4 minutes at room temperature, then filter and recrystallize the precipitate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for procurement managers and supply chain heads focused on resilience and cost efficiency. The elimination of benzene removes a major regulatory hurdle and the associated costs of hazardous waste disposal and air emission monitoring. Furthermore, the substitution of expensive catalysts with simple aniline and the use of commodity solvents like water and ethanol significantly lowers the raw material cost base. The dramatic reduction in reaction time from 24 hours to mere minutes translates directly into increased asset utilization, allowing existing manufacturing infrastructure to produce significantly higher volumes without additional capital investment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of costly inputs and energy consumption. By operating at room temperature, the need for heating or cooling utilities is virtually eliminated, leading to substantial energy savings. Additionally, the avoidance of transition metal or organic catalysts removes the necessity for expensive scavenging steps to meet residual metal specifications, which is a common cost driver in API intermediate production. The high yield and simple filtration workup further reduce labor and processing time, contributing to a leaner cost structure.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of universally available raw materials. Aromatic aldehydes, aniline, and Michaelis acid are commodity chemicals with robust global supply chains, reducing the risk of shortages associated with specialized reagents. The simplicity of the process also means that it can be easily replicated across multiple manufacturing sites if necessary, providing a buffer against regional disruptions. The short cycle time ensures that inventory turnover is rapid, allowing for a more responsive just-in-time delivery model to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards or pressure build-up. The use of green solvents aligns perfectly with the sustainability goals of major multinational corporations, facilitating easier approval during vendor audits. The aqueous workup in the first step and ethanolic crystallization in the second generate waste streams that are easier to treat compared to halogenated or aromatic solvent wastes, ensuring long-term environmental compliance and reducing the liability associated with waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-(aryl methylene) Michaelis acid. These insights are derived directly from the patented methodology and are intended to clarify the operational benefits and chemical properties of the material for potential partners. Understanding these nuances is essential for integrating this intermediate into your specific synthesis pipelines effectively.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method eliminates the use of toxic benzene solvents and expensive catalysts required in prior art. It utilizes water and ethanol, operates at room temperature, and significantly reduces reaction time from hours to minutes while avoiding bis-condensation impurities.
Q: How does this process control impurities like the bimolecular condensation product?
A: By forming the arylidene aniline intermediate first, the reactivity is modulated. The subsequent reaction with Michaelis acid is extremely fast (3-4 minutes), preventing the prolonged exposure that leads to the formation of the unwanted bimolecular Michaelis acid condensation product (Compound 4).
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses common, low-cost solvents (water and ethanol) and mild conditions (room temperature), making it highly scalable. The simple filtration and recrystallization workup facilitate easy isolation of high-purity products suitable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(Aryl Methylene) Michaelis Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our capability to implement the green synthesis methods described in patent CN101921258A demonstrates our dedication to sustainable manufacturing practices that align with your corporate responsibility goals.
We invite you to engage with our technical procurement team to discuss how our optimized processes can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless integration of our high-performance intermediates into your manufacturing workflow.
