Advanced Decarboxylation Strategy for High-Purity Trans-2-Substituent-5-Carboxyl-1,3-Dioxane Intermediates
Introduction to High-Selectivity Dioxane Synthesis
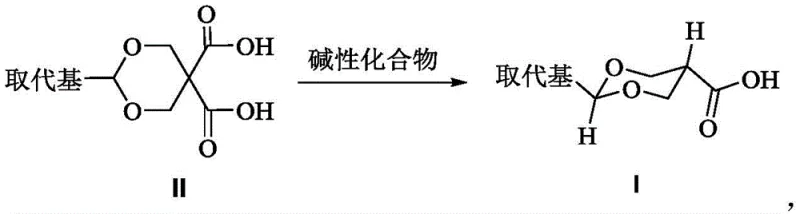
The pharmaceutical industry constantly demands intermediates with precise stereochemical configurations to ensure the efficacy and safety of final active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in patent CN108929304B, which discloses a novel preparation method for trans-2-substituent-5-carboxyl-1,3-dioxane derivatives. These compounds serve as critical building blocks for various bioactive molecules, where the trans-configuration is often the pharmacologically active form. The core innovation lies in a base-catalyzed decarboxylation strategy that transforms 2-substituent-5,5-dicarboxy-1,3-dioxane precursors into the desired trans-monocarboxylic acid products with exceptional stereoselectivity. This technological advancement addresses long-standing challenges in impurity control and process efficiency, positioning it as a vital asset for reliable pharmaceutical intermediate supplier networks seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted-5-carboxy-1,3-dioxanes has relied on methodologies described in earlier literature, such as the work by Bank et al. (JOC, 1973). These conventional approaches typically utilized simple aromatic amines like pyridine, aniline, or their methylated derivatives as both solvents and bases. While these methods could facilitate the decarboxylation reaction, they suffered from a critical deficiency: poor stereocontrol. The reaction conditions often resulted in a mixture of cis and trans isomers, with the unwanted cis-isomer constituting anywhere from 10% to 50% of the product mixture. For instance, when 2,6-dimethylpyridine was employed, the main product was actually the cis-isomer, necessitating complex and costly downstream purification processes to isolate the therapeutically relevant trans-configuration. This lack of selectivity not only reduces overall yield but also introduces significant variability in the quality of the intermediate, posing risks for downstream API synthesis.
The Novel Approach
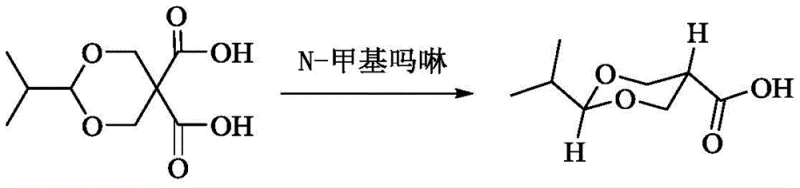
In stark contrast, the methodology outlined in CN108929304B introduces a paradigm shift by employing specific five- or six-membered heteroaromatic or heterosaturated ring compounds as the catalytic base. By selecting bases such as N-methylmorpholine or N-methylimidazole, the process achieves a dramatic improvement in stereoselectivity. As demonstrated in the patent examples, reacting 2-isopropyl-5,5-dicarboxy-1,3-dioxane with N-methylmorpholine in 1,4-dioxane at 80-105°C yields the trans-product with a cis:trans ratio of 1:116.6, and in scaled-up runs, the cis-configuration becomes undetectable by HPLC. This represents a quantum leap in process chemistry, effectively eliminating the need for difficult chiral separations or recrystallizations that plague older methods. The ability to drive the reaction almost exclusively towards the trans-isomer simplifies the workflow and ensures a much higher purity profile right out of the reactor.
Mechanistic Insights into Base-Catalyzed Decarboxylation
The success of this novel preparation method can be attributed to the specific electronic and steric properties of the chosen alkaline compounds. The reaction involves the thermal decarboxylation of a gem-dicarboxylic acid moiety within the dioxane ring system. The presence of a base facilitates the formation of a carboxylate anion, which subsequently undergoes decarboxylation. However, the stereochemical outcome is heavily influenced by the transition state stability. The use of N-substituted morpholines or imidazoles, as depicted in the general formula structures, likely creates a specific coordination environment or transition state geometry that favors the formation of the thermodynamically more stable trans-isomer while kinetically suppressing the formation of the cis-isomer. Unlike bulky bases that might force a different conformation, these specific heterocyclic amines appear to strike an optimal balance, allowing the substrate to adopt a conformation during the decarboxylation event that leads predominantly to the equatorial positioning of the substituent groups in the final product.
Furthermore, the choice of solvent plays a synergistic role in this mechanistic pathway. The patent highlights the effectiveness of solvents like 1,4-dioxane, toluene, and acetonitrile, which have boiling points ranging from 80°C to 200°C. These solvents provide the necessary thermal energy to overcome the activation barrier for decarboxylation while maintaining a homogeneous reaction phase that supports the catalytic cycle of the base. The interaction between the solvent polarity and the basicity of the nitrogen-containing catalyst ensures that the proton transfer steps required for the decarboxylation proceed efficiently. This mechanistic understanding allows for precise tuning of reaction parameters, ensuring that impurities arising from side reactions or isomerization are minimized, thereby delivering a product stream that meets the stringent purity specifications required for pharmaceutical manufacturing.
How to Synthesize Trans-2-Substituent-5-Carboxyl-1,3-Dioxane Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized conditions identified in the patent data to maximize yield and selectivity. The process is designed to be operationally simple, avoiding the need for exotic reagents or extreme conditions, which facilitates easy technology transfer. Operators should focus on maintaining the precise temperature window of 80-105°C and ensuring the molar ratio of the base to the substrate is sufficient to drive the reaction to completion without generating excessive waste. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational protocol for process development teams aiming to adopt this superior manufacturing route.
- Combine 2-substituent-5,5-dicarboxy-1,3-dioxane raw material with a specific alkaline compound (such as N-methylmorpholine) in a suitable solvent like 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 80-105°C and maintain stirring for approximately 4 hours until HPLC monitoring confirms complete consumption of raw materials.
- Remove solvent under reduced pressure, adjust pH to approximately 2.5 with hydrochloric acid, cool to 5°C for crystallization, and filter to obtain the high-purity trans-product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By achieving near-perfect stereoselectivity, manufacturers can bypass expensive and time-consuming chromatographic separations or multiple recrystallization steps that were previously necessary to remove cis-isomer impurities. This reduction in processing steps directly translates to lower operational expenditures and reduced solvent consumption, aligning with green chemistry principles and cost reduction in pharmaceutical intermediate manufacturing goals. Additionally, the use of common, commercially available solvents like 1,4-dioxane and toluene ensures that raw material sourcing remains stable and unaffected by supply chain disruptions associated with specialty chemicals.
- Cost Reduction in Manufacturing: The elimination of complex purification stages significantly lowers the cost of goods sold (COGS). Since the reaction yields the desired trans-isomer with high selectivity (cis:trans ratios exceeding 1:100), the loss of valuable material during impurity removal is virtually eliminated. This efficiency means that less starting material is required to produce the same amount of qualified product, optimizing raw material utilization. Furthermore, the recovery and recycling of solvents like dioxane are well-established industrial practices, allowing for further economic optimization without compromising product quality.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes to greater supply chain resilience. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. This compatibility with existing infrastructure reduces the capital expenditure required for scale-up. Moreover, the starting materials, specifically the 2-substituent-5,5-dicarboxy-1,3-dioxane precursors, are accessible via established synthetic pathways, ensuring a steady flow of inputs for continuous production schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates successful scaling, with Example 2 detailing a reaction using over 500 grams of starting material that maintained high yield and selectivity. This indicates a clear path to commercial scale-up of complex pharmaceutical intermediates. From an environmental perspective, the process generates fewer waste streams compared to traditional methods because there is less need for extensive washing and separation of isomers. The ability to operate with high atom economy and reduced solvent intensity supports corporate sustainability targets and simplifies regulatory compliance regarding waste disposal and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this decarboxylation technology. These insights are derived directly from the experimental data and claims within patent CN108929304B, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios. Understanding these nuances is crucial for making informed decisions about process adoption and supplier qualification.
Q: How does this method improve stereoselectivity compared to traditional pyridine-based routes?
A: Unlike traditional methods using pyridine which often yield 10-50% cis-isomer impurities, this patent utilizes specific N-substituted heterocyclic bases like N-methylmorpholine to achieve cis:trans ratios as high as 1:116.6, drastically reducing purification burden.
Q: What solvents are compatible with this decarboxylation process?
A: The process demonstrates flexibility with solvents having boiling points between 80-200°C, including nitriles, monocyclic aromatic hydrocarbons, ethers, and amides, with 1,4-dioxane and toluene showing particularly effective results for yield and selectivity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly details scalable examples (e.g., Example 2 using over 500g of starting material) achieving consistent yields around 70% with undetectable cis-impurities, indicating robustness for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-2-Substituent-5-Carboxyl-1,3-Dioxane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the advancements presented in CN108929304B and is fully equipped to leverage this high-selectivity decarboxylation technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless. Our facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of trans-2-substituent-5-carboxyl-1,3-dioxane we deliver meets the exacting standards required for global pharmaceutical applications.
We invite R&D directors and procurement leaders to collaborate with us to optimize their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how switching to this novel synthetic route can impact your overall project economics. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your specific project needs, ensuring a secure and efficient supply of this vital intermediate.
