Advanced Synthesis of Oxaspiro-Based Photoresist Monomers for High-Resolution Lithography
The semiconductor industry's relentless drive towards smaller node sizes demands photoresist materials with unprecedented resolution and etching resistance. Patent CN111777587A introduces a breakthrough class of degradable photoresist resin monomers synthesized from oxaspiro[4.5]decanedione, addressing critical limitations in current lithographic processes. These novel monomers incorporate a rigid oxaspiro core combined with dual polymerizable groups, creating a unique molecular architecture that balances solubility with mechanical stability. For R&D directors and procurement specialists in the electronic chemical sector, this technology represents a significant leap forward in achieving high-fidelity pattern transfer. The synthesis pathway described offers a robust method for generating these high-performance materials, utilizing well-established organic transformations that are amenable to rigorous quality control standards required in semiconductor manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoresist resins often rely on linear polymer chains functionalized with a single acid-sensitive group, which inherently limits their performance in advanced lithography applications. These conventional structures frequently suffer from inadequate etching resistance during the plasma etching steps, leading to pattern deformation and loss of critical dimensions. Furthermore, the dissolution contrast between exposed and unexposed regions in linear polymers is often insufficient, resulting in poor edge roughness and reduced resolution. The reliance on simple linear backbones means that the physical properties of the resist are heavily dependent on the specific acid-sensitive moiety alone, lacking the synergistic benefits of a cross-linked network. This structural simplicity becomes a bottleneck when attempting to push the boundaries of resolution for sub-micron and nanometer-scale device fabrication.
The Novel Approach
The innovative strategy outlined in the patent overcomes these deficiencies by introducing a monomer capable of forming a three-dimensional network structure upon polymerization. By incorporating two unsaturated carbon-carbon double bonds into the oxaspiro[4.5]decane framework, the resulting polymer achieves superior cross-linking density, which dramatically enhances etching resistance compared to linear analogues. 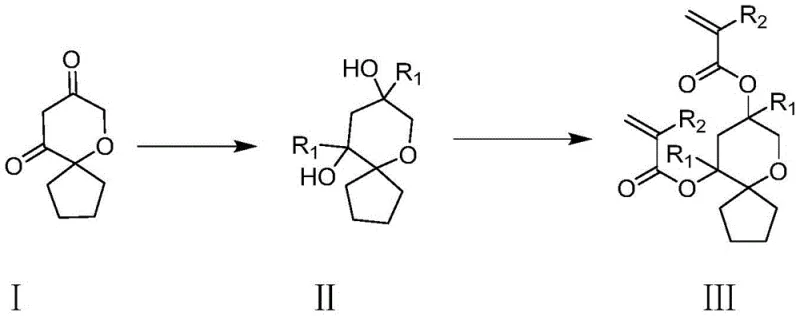 The presence of the oxygen-containing heterocyclic ring further contributes to the resolution improvement while maintaining high solubility in fat-soluble ester solvents used during spin-coating. This dual functionality ensures that the resin remains processable prior to exposure but becomes highly robust and resistant during the etching phase. The acid-cleavable nature of the ester linkages ensures that upon exposure and subsequent acid generation, the polymer backbone fragments into smaller, more soluble species, creating a sharp dissolution rate differential that is essential for high-resolution patterning.
The presence of the oxygen-containing heterocyclic ring further contributes to the resolution improvement while maintaining high solubility in fat-soluble ester solvents used during spin-coating. This dual functionality ensures that the resin remains processable prior to exposure but becomes highly robust and resistant during the etching phase. The acid-cleavable nature of the ester linkages ensures that upon exposure and subsequent acid generation, the polymer backbone fragments into smaller, more soluble species, creating a sharp dissolution rate differential that is essential for high-resolution patterning.
Mechanistic Insights into Grignard Addition and Esterification
The synthesis mechanism relies on a precise two-step sequence beginning with the nucleophilic addition of a Grignard reagent to the diketone precursor. In the first stage, 6-oxaspiro[4.5]decane-8,10-dione reacts with an alkyl or cycloalkyl magnesium halide under strictly anhydrous conditions to form a diol intermediate. This step is critical as it installs the R1 substituents which modulate the steric and solubility properties of the final monomer. Maintaining the reaction temperature between 0-30°C is essential to prevent side reactions and ensure high selectivity for the desired diol product. The use of anhydrous ether as a solvent facilitates the formation of the Grignard complex and stabilizes the intermediate organometallic species, ensuring a clean conversion to the hydroxyl-functionalized core.
Following the isolation of the diol intermediate, the second step involves an esterification reaction with acryloyl or methacryloyl chloride to install the polymerizable vinyl groups. This transformation is conducted under alkaline conditions, typically using triethylamine or pyridine as a base to scavenge the hydrogen chloride byproduct. 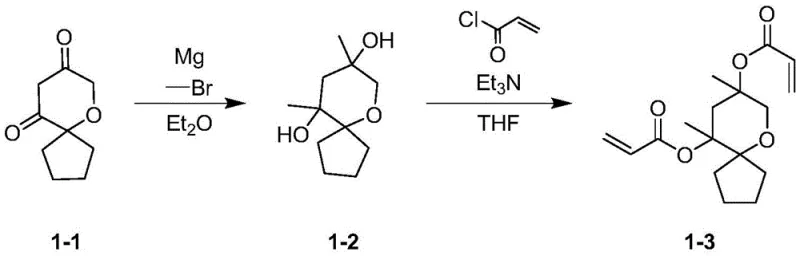 The reaction temperature is carefully controlled between 0-70°C in solvents such as tetrahydrofuran or toluene to optimize the reaction kinetics while minimizing thermal degradation of the sensitive acrylate groups. This esterification step effectively caps the hydroxyl groups with the photopolymerizable moieties, completing the synthesis of the target resin monomer. The rigorous control of reaction parameters in both steps ensures minimal impurity formation, which is paramount for preventing defects in the final photoresist film.
The reaction temperature is carefully controlled between 0-70°C in solvents such as tetrahydrofuran or toluene to optimize the reaction kinetics while minimizing thermal degradation of the sensitive acrylate groups. This esterification step effectively caps the hydroxyl groups with the photopolymerizable moieties, completing the synthesis of the target resin monomer. The rigorous control of reaction parameters in both steps ensures minimal impurity formation, which is paramount for preventing defects in the final photoresist film.
How to Synthesize Oxaspiro-Based Photoresist Monomers Efficiently
The practical execution of this synthesis requires careful attention to moisture exclusion and temperature management to achieve the high purity levels demanded by the electronics industry. The process begins with the preparation of the Grignard reagent, followed by its addition to the diketone solution, and concludes with the acylation of the resulting diol. Detailed operational parameters, including specific molar ratios and workup procedures, are critical for reproducing the high yields reported in the patent examples. For process chemists looking to implement this route, understanding the nuances of the quenching and purification steps is vital to removing magnesium salts and unreacted starting materials effectively.
- Perform a Grignard reaction between 6-oxaspiro[4.5]decane-8,10-dione and an alkyl or cycloalkyl Grignard reagent in anhydrous ether at 0-30°C to form the diol intermediate.
- Quench the reaction with water or dilute acid, separate the organic layer, and purify the intermediate via extraction and column chromatography.
- React the purified diol intermediate with acryloyl chloride or methacryloyl chloride in the presence of a base like triethylamine in THF at 0-70°C to yield the final resin monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthetic route offers substantial advantages due to its reliance on commodity chemicals and standard unit operations. The starting material, 6-oxaspiro[4.5]decane-8,10-dione, along with common alkyl bromides and acryloyl chlorides, are readily available from established chemical suppliers, reducing the risk of raw material shortages. The elimination of exotic catalysts or complex multi-step sequences simplifies the procurement landscape and lowers the overall cost of goods sold. Furthermore, the use of common solvents like ether and THF means that existing manufacturing infrastructure can often be utilized without significant capital expenditure on new equipment. This compatibility with standard chemical processing capabilities ensures a smoother transition from laboratory scale to commercial production.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of expensive transition metal catalysts or precious metal reagents, which significantly lowers the direct material costs associated with production. By utilizing a straightforward Grignard addition followed by esterification, the process minimizes the number of purification steps required, thereby reducing solvent consumption and waste disposal costs. The high efficiency of the reaction steps implies less raw material is wasted on byproducts, leading to a more atom-economical process overall. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs in large-scale manufacturing facilities.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that the supply chain is resilient against market fluctuations that often affect specialty reagents. Since the synthesis does not depend on single-source proprietary catalysts, procurement teams have the flexibility to source raw materials from multiple vendors, mitigating the risk of supply disruptions. The robustness of the chemical transformations means that batch-to-batch variability is minimized, ensuring consistent product quality and reliable delivery schedules for downstream photoresist formulators. This stability is crucial for maintaining continuous production lines in the fast-paced semiconductor industry.
- Scalability and Environmental Compliance: The process is inherently scalable as it utilizes reaction types that are well-understood and easily managed in large reactors. The absence of heavy metals simplifies the environmental compliance profile, making it easier to meet stringent regulatory standards for wastewater and waste solid disposal. The solvents used are common and can be efficiently recovered and recycled, further reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only aids in regulatory compliance but also enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel photoresist monomers. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing product portfolios. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: What are the key structural advantages of this oxaspiro-based monomer?
A: The monomer features a rigid oxaspiro[4.5]decane core which enhances etching resistance, and two polymerizable vinyl groups that enable cross-linking for a robust 3D network structure.
Q: How does this monomer improve lithography resolution?
A: Upon exposure, the acid-sensitive ester groups cleave, significantly increasing solubility in the developer. This sharp contrast in dissolution rates before and after exposure reduces edge roughness and improves pattern resolution.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes standard Grignard and esterification reactions with mild temperature conditions (0-70°C) and common solvents like ether and THF, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Resin Monomer Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for the electronic materials sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing complex organic syntheses, such as the oxaspiro-based route, to meet the stringent purity specifications required for semiconductor applications. We operate rigorous QC labs equipped with advanced analytical instrumentation to ensure that every batch of photoresist intermediate meets the highest standards of quality and consistency. Our commitment to process excellence ensures that we can deliver high-purity photoresist resin monomers that enable our clients to achieve superior lithographic performance.
We invite potential partners to contact our technical procurement team to discuss how we can support your specific material requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage you to reach out for specific COA data and route feasibility assessments to verify our capability to supply these advanced electronic chemicals reliably. Let us collaborate to accelerate the development of next-generation photoresist formulations that drive the future of semiconductor manufacturing.
