Advanced Synthesis of Substituted Naphthoporphyrins and Metal Complexes for Industrial Catalysis
The landscape of functional macrocyclic compounds is undergoing a significant transformation, driven by the demand for high-performance materials in energy conversion and catalysis. Patent CN115093423A introduces a robust and versatile synthetic methodology for producing naphthoporphyrin (2.1.2.1) derivatives with varying substituents, addressing critical gaps in the current supply of advanced electronic chemicals. This technology leverages a strategic combination of palladium-catalyzed cross-coupling and acid-mediated condensation to construct highly curved, non-aromatic macrocyclic cavities that exhibit superior electrochemical properties. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a pivotal shift towards more accessible and tunable porphyrin architectures. The ability to systematically modify the meso-positions with groups such as pentafluorophenyl, phenyl, or p-tolyl allows for precise tuning of redox potentials, making these compounds ideal candidates for molecular catalysts in hydrogen production and carbon dioxide reduction cycles.
Furthermore, the inclusion of a streamlined metalation protocol for generating nickel and copper complexes expands the utility of these materials into the realm of industrial catalysis and fuel cell technology. The synthesis route described avoids excessively harsh conditions often associated with traditional porphyrin synthesis, utilizing moderate temperatures and common organic solvents that facilitate easier scale-up. As a reliable electronic chemical supplier, understanding the nuances of this patented pathway is essential for securing a competitive edge in the market for next-generation energy materials. The following analysis dissects the technical merits of this invention, highlighting its potential for cost reduction in catalyst manufacturing and its implications for supply chain stability in the production of high-purity naphthoporphyrins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing expanded porphyrins or fused porphyrinoid systems often suffer from low atom economy and rigorous reaction conditions that hinder commercial viability. Conventional routes frequently rely on statistical condensation of pyrroles and aldehydes, which can lead to complex mixtures of regioisomers that are difficult and expensive to separate, drastically increasing the cost of goods sold. Additionally, many existing protocols require high-temperature reflux in toxic solvents or the use of unstable intermediates that degrade rapidly upon exposure to air or moisture. These factors create significant bottlenecks for supply chain heads who require consistent quality and reliable delivery schedules for large-scale projects. The lack of modularity in older methods also means that introducing specific electron-withdrawing or electron-donating groups to tune the electronic properties of the macrocycle often requires redesigning the entire synthetic sequence from scratch, leading to prolonged development timelines.
The Novel Approach
The methodology outlined in patent CN115093423A overcomes these hurdles by employing a stepwise construction strategy that ensures high regioselectivity and purity. By first establishing the naphthalene-pyrrole scaffold through a controlled Suzuki-Miyaura coupling, the process guarantees the correct positioning of the pyrrole units before macrocyclization occurs. This pre-organization significantly reduces the formation of unwanted byproducts, simplifying the downstream purification processes which typically involve costly column chromatography. The subsequent deprotection and condensation steps are conducted under relatively mild conditions, utilizing ethylene glycol for deprotection and boron trifluoride etherate for cyclization at room temperature. This gentle approach preserves the integrity of sensitive functional groups, such as the pentafluorophenyl moiety, which might be compromised under more aggressive thermal conditions. Consequently, this novel approach offers a direct path to commercial scale-up of complex naphthoporphyrins with reproducible yields and defined structural characteristics.
Mechanistic Insights into Pd-Catalyzed Coupling and Acid-Mediated Cyclization
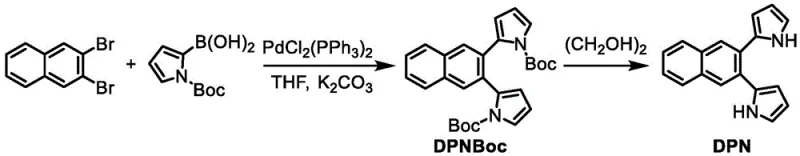
The core of this synthesis lies in the initial formation of the 2,3-bis(1H-pyrrol-2-yl)naphthalene (DPN) precursor, which serves as the rigid backbone for the macrocycle. The reaction begins with a palladium-catalyzed cross-coupling between 2,3-dibromonaphthalene and (1-(tert-butoxycarbonyl)-1H-pyrrol-2-yl)boronic acid. In this step, the PdCl2(PPh3)2 catalyst facilitates the oxidative addition into the carbon-bromine bonds of the naphthalene ring, followed by transmetallation with the organoboron species. The presence of potassium carbonate acts as a base to activate the boronic acid, promoting the formation of the carbon-carbon bond while the Boc group protects the pyrrole nitrogen from unwanted side reactions. This careful protection strategy is crucial for maintaining the nucleophilicity of the pyrrole alpha-carbons for the subsequent cyclization step. The reaction is typically carried out in tetrahydrofuran (THF) at temperatures ranging from 45°C to 80°C, with optimal results observed at 65°C over a 72-hour period, ensuring complete conversion of the dibromide starting material.
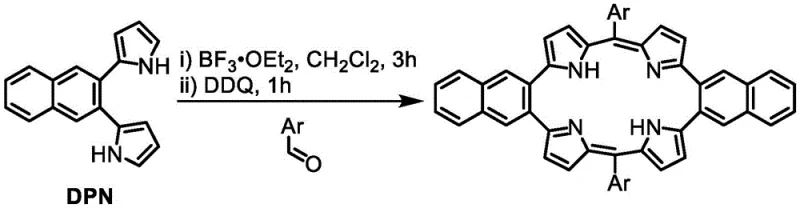
Following the coupling, the Boc protecting groups are removed using ethylene glycol at elevated temperatures (100°C to 180°C) under an inert argon atmosphere. This thermal deprotection generates the free amine DPN intermediate, which is then immediately subjected to macrocyclization. The cyclization mechanism involves the activation of aromatic aldehydes by the Lewis acid boron trifluoride etherate (BF3·OEt2), which increases the electrophilicity of the carbonyl carbon. The electron-rich alpha-positions of the pyrrole rings in DPN then attack the activated aldehyde, forming methine bridges that link the pyrrole units into a large macrocyclic structure. The final oxidation step using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) aromatizes the macrocycle, establishing the conjugated pi-system responsible for the compound's unique optical and electronic properties. This two-stage condensation and oxidation process is highly efficient, allowing for the incorporation of various aryl groups simply by changing the aldehyde feedstock.
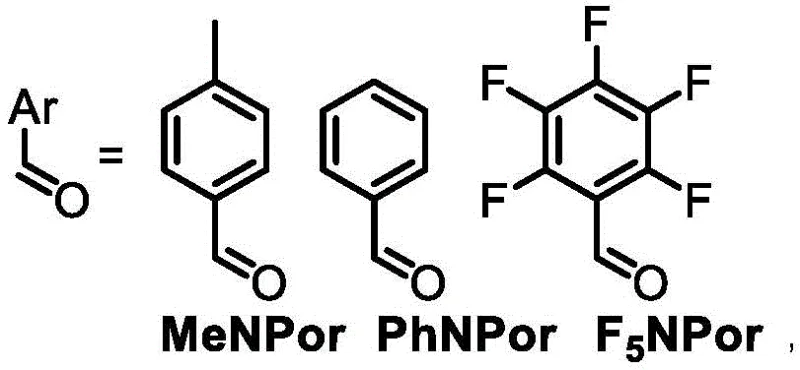
The versatility of this mechanism extends to the post-synthetic modification of the macrocycle through metalation. The central cavity of the naphthoporphyrin contains nitrogen atoms capable of coordinating with transition metal ions. When F5NPor is treated with metal salts such as nickel acetate or copper acetate in a mixture of dichloromethane and methanol, the metal ion displaces the inner protons to form a stable coordination complex. This process is driven by the thermodynamic stability of the metal-nitrogen bonds and is facilitated by the flexible nature of the macrocyclic cavity. The resulting metal complexes, such as F5NPorNi and F5NPorCu, exhibit distinct electrochemical behaviors compared to the free base, including irreversible two-stage oxidation and reduction potentials. These properties are critical for their application as molecular catalysts, where the metal center acts as the active site for electron transfer reactions in fuel cells or carbon capture systems.
How to Synthesize Substituted Naphthoporphyrin Efficiently
The synthesis of these high-value macrocycles requires precise control over reaction parameters to ensure high purity and yield. The process is divided into three distinct stages: the construction of the bipyrrole-naphthalene scaffold, the deprotection to reveal the reactive pyrroles, and the final macrocyclization with the desired aldehyde. Each step demands strict exclusion of oxygen and moisture, particularly during the palladium-catalyzed coupling and the metalation phases, to prevent catalyst deactivation or product degradation. The detailed standardized synthesis steps below outline the specific molar ratios, solvent volumes, and temperature profiles required to replicate the results described in the patent, providing a clear roadmap for laboratory scale-up and process optimization.
- Perform Suzuki coupling of 2,3-dibromonaphthalene with Boc-protected pyrrole boronic acid using PdCl2(PPh3)2 catalyst in THF/K2CO3 at 65°C to obtain DPNBoc.
- Deprotect DPNBoc using ethylene glycol at 160°C under argon protection to yield the key intermediate 2,3-bis(1H-pyrrol-2-yl)naphthalene (DPN).
- Condense DPN with aromatic aldehydes using BF3·OEt2 in dichloromethane, followed by DDQ oxidation to form the final naphthoporphyrin macrocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in this patent offers substantial advantages in terms of raw material sourcing and process efficiency. The starting materials, including 2,3-dibromonaphthalene and various aromatic aldehydes, are commodity chemicals that are readily available from multiple global suppliers, reducing the risk of supply chain disruptions. Unlike specialized porphyrin precursors that may have long lead times or single-source dependencies, the inputs for this process can be sourced competitively, driving down the overall cost of goods. Furthermore, the use of common organic solvents like THF, dichloromethane, and ethylene glycol simplifies waste management and solvent recovery operations, contributing to a more sustainable and cost-effective manufacturing footprint. The elimination of exotic reagents or cryogenic conditions further lowers the barrier to entry for contract manufacturing organizations looking to produce these materials at scale.
- Cost Reduction in Manufacturing: The stepwise nature of the synthesis allows for the isolation and purification of intermediates, which minimizes the loss of valuable materials due to side reactions in one-pot processes. By optimizing the Suzuki coupling conditions to achieve high conversion rates, the consumption of the expensive palladium catalyst is maximized, reducing the effective cost per kilogram of the final product. Additionally, the ability to perform the macrocyclization at room temperature significantly reduces energy consumption compared to traditional high-temperature porphyrin syntheses. These cumulative efficiencies translate into a more favorable cost structure, enabling competitive pricing for high-purity naphthoporphyrins in the electronic materials market without compromising on quality or performance specifications.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for supply chain managers overseeing long-term contracts. The tolerance of the method to different aromatic aldehydes means that production can be easily switched between different product variants (e.g., F5NPor, PhNPor, MeNPor) without requiring major equipment changes or requalification of the entire process line. This flexibility allows manufacturers to respond quickly to shifting market demands for specific catalyst types. Moreover, the stability of the intermediates, such as the Boc-protected DPNBoc, allows for strategic stockpiling, buffering against potential fluctuations in the availability of fresh starting materials and ensuring continuous supply for downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard glassware like Schlenk tubes in the lab which can be directly translated to stirred tank reactors in pilot and commercial plants. The absence of highly toxic heavy metals in the final product (unless specifically metalated for catalysis) and the use of manageable solvents simplify regulatory compliance regarding environmental discharge. The purification steps, primarily involving column chromatography with standard stationary phases like silica gel or alumina, are well-understood unit operations that can be scaled using simulated moving bed (SMB) chromatography for industrial throughput. This alignment with established chemical engineering practices reduces the technical risk associated with scaling up novel fine chemical processes, facilitating a smoother transition from R&D to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these naphthoporphyrin derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating these materials into existing product lines or research pipelines.
Q: What are the optimal reaction conditions for the Suzuki coupling step?
A: The patent specifies using PdCl2(PPh3)2 as the catalyst in a THF and 1M potassium carbonate solution system, preferably reacting at 65°C for 72 hours to maximize the yield of the DPNBoc intermediate.
Q: Which metal salts are suitable for complexation with F5NPor?
A: A variety of nickel and copper salts can be used, including acetates, sulfates, chlorides, and nitrates. Nickel acetate and copper acetate are highlighted as preferred options for achieving high yields of the corresponding metal complexes.
Q: How does this synthesis method improve upon conventional porphyrin production?
A: This method utilizes a modular approach starting from commercially available 2,3-dibromonaphthalene, allowing for the introduction of diverse substituents via simple aldehyde variation, thereby enhancing structural tunability for specific catalytic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoporphyrin Supplier
The technological advancements detailed in patent CN115093423A underscore the growing importance of tailored macrocyclic compounds in the fields of renewable energy and advanced catalysis. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these sophisticated molecules to the global market. Our commitment to stringent purity specifications and the operation of rigorous QC labs ensures that every batch of naphthoporphyrin or metal complex meets the exacting standards required for high-performance applications. We understand that consistency is key in the electronic chemical sector, and our state-of-the-art facilities are equipped to handle the nuanced requirements of air-sensitive and light-sensitive syntheses described in this patent.
We invite industry partners to collaborate with us to explore the full potential of these materials in your specific applications. Whether you require custom synthesis of novel derivatives or bulk supply of standard catalog items, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these high-value catalysts into your supply chain. Together, we can accelerate the development of sustainable energy solutions through the power of advanced organic synthesis.
