Advanced Synthesis of Asymmetric Heptathiophene for High-Performance Organic Electronics Manufacturing
Advanced Synthesis of Asymmetric Heptathiophene for High-Performance Organic Electronics Manufacturing
The rapid evolution of organic electronics demands materials with precisely tuned electronic properties and morphological stability. Patent CN108864143B introduces a groundbreaking methodology for synthesizing asymmetric seven-membered fused thiophenes, addressing critical limitations in current organic semiconductor manufacturing. Unlike traditional symmetric oligomers, these asymmetric structures provide unique molecular packing capabilities that can significantly enhance charge transport characteristics in devices such as organic field-effect transistors and solar cells. The innovation lies not just in the final molecular architecture but in the robust chemical pathway designed to access these complex scaffolds with high fidelity. By leveraging advanced cross-coupling techniques and controlled ring-closure strategies, this technology enables the production of high-purity materials essential for next-generation display and energy applications.
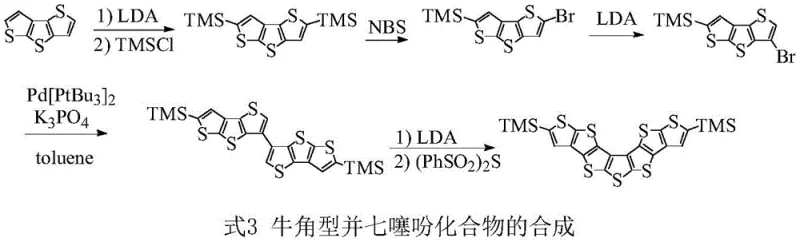
For procurement specialists and supply chain managers, understanding the synthetic accessibility of such complex molecules is paramount. The disclosed method utilizes readily available dithienothiophene building blocks, functionalized with trimethylsilyl (TMS) groups to improve solubility and processing characteristics. This strategic functionalization is not merely a synthetic handle but a critical design element that facilitates purification and film formation in device fabrication. The transition from laboratory-scale discovery to industrial viability hinges on the reproducibility of the coupling steps and the efficiency of the final cyclization. As a reliable organic semiconductor material supplier, recognizing the value of such patented processes allows for better forecasting of material availability and cost structures in the competitive electronic chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused thiophene oligomers has been plagued by significant challenges regarding regioselectivity and product isolation. Conventional approaches often rely on direct halogen-lithium exchange followed by copper-mediated coupling or non-selective palladium catalysis. These methods frequently result in statistical mixtures containing both homocoupled byproducts and the desired heterocoupled species. Because these isomers possess nearly identical solubility profiles and polarities, separating the target asymmetric molecule from its symmetric counterparts requires extensive and yield-depleting chromatographic purification. Furthermore, traditional routes to linear or helical heptathiophenes often lack the structural diversity needed to fine-tune intermolecular stacking, limiting the ultimate performance ceiling of the resulting electronic devices. The inability to efficiently access specific asymmetric isomers has thus remained a bottleneck in the development of high-mobility organic semiconductors.
The Novel Approach
The methodology described in the patent data overcomes these hurdles through a meticulously planned Suzuki-Miyaura cross-coupling strategy. By differentiating the two coupling partners—one functionalized as a boronate ester and the other as an aryl bromide—the process inherently suppresses the formation of homocoupling impurities. This chemoselective approach ensures that the reaction predominantly yields the desired unsymmetrical biaryl intermediate, drastically reducing the burden on downstream purification processes. The use of trimethylsilyl protecting groups further aids in managing the reactivity of the thiophene rings, preventing unwanted side reactions during the harsh conditions of lithiation and coupling. This strategic divergence from conventional non-selective coupling represents a significant leap forward in synthetic efficiency, offering a scalable route to materials that were previously difficult to obtain in high purity.
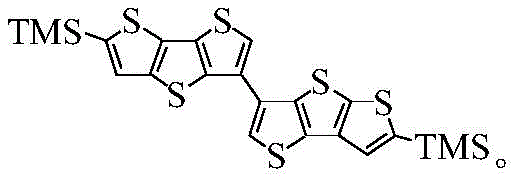
Mechanistic Insights into Suzuki Coupling and Electrocyclic Ring Closure
The core of this synthetic innovation involves a sophisticated interplay of organometallic transformations and electrophilic sulfur insertion. The initial phase relies on the precise generation of a boronic ester via lithium-halogen exchange at cryogenic temperatures, typically around -78°C to -90°C. This low-temperature control is critical to prevent metal-halogen exchange at unintended positions on the fused thiophene core, ensuring that the boron moiety is installed exclusively at the target site. Subsequent palladium-catalyzed coupling with a complementary bromo-thiophene derivative proceeds under mild aqueous conditions, utilizing bases like potassium carbonate or cesium carbonate to facilitate the transmetallation step. The robustness of the Suzuki mechanism allows for tolerance of the sensitive sulfur-containing heterocycles, which might otherwise be susceptible to oxidation or decomposition under more aggressive coupling conditions.
The final and perhaps most chemically intriguing step is the formation of the central seven-membered ring via an electrocyclic closure mediated by diphenylsulfonyl sulfide. Following deprotonation of the biaryl precursor with a strong base such as LDA (lithium diisopropylamide), the resulting dianion reacts with the sulfur transfer reagent to forge the new carbon-sulfur bonds. This intramolecular cyclization effectively locks the molecular conformation, creating the rigid, planar fused system required for effective pi-orbital overlap. The choice of diphenylsulfonyl sulfide as the sulfur source is particularly advantageous as it provides a controlled release of sulfur, minimizing the formation of polysulfide impurities. This mechanistic precision ensures that the final asymmetric heptathiophene possesses the structural integrity necessary for high-performance charge transport in organic electronic devices.
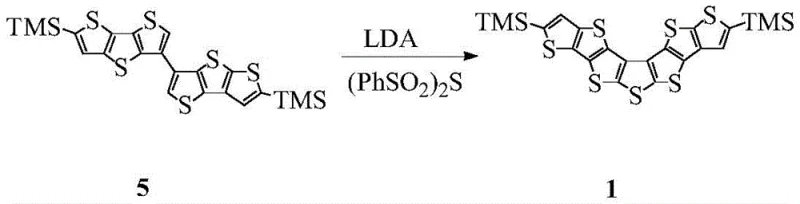
How to Synthesize Asymmetric Heptathiophene Efficiently
The synthesis of this high-value organic semiconductor intermediate requires strict adherence to inert atmosphere techniques and precise temperature control to maximize yield and purity. The process begins with the activation of the bromo-precursor, followed by the critical cross-coupling event that defines the molecular asymmetry. Operators must ensure that all solvents are thoroughly dried and degassed to prevent catalyst deactivation during the palladium cycle. The final ring-closing step demands careful quenching to preserve the integrity of the newly formed fused system. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized protocol below which outlines the critical control points for successful manufacturing.
- Perform lithium-halogen exchange on 3-bromo-6-trimethylsilyl-dithienothiophene followed by reaction with pinacol borate to generate the boronate ester intermediate.
- Execute a palladium-catalyzed Suzuki cross-coupling between the boronate ester and 5-bromo-2-trimethylsilyl-dithienothiophene to form the biaryl precursor.
- Conduct deprotonation using LDA followed by thiocyclization with diphenylsulfonyl sulfide ((PhSO2)2S) to close the central seven-membered ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for supply chain stability and cost management in the electronic chemicals sector. The reliance on Suzuki coupling, a well-established industrial process, means that the technology can be scaled using existing infrastructure without the need for exotic or hazardous reagents that complicate logistics. The high selectivity of the reaction directly translates to reduced solvent consumption and lower waste generation, as extensive chromatographic separation is minimized. This efficiency gain is crucial for maintaining competitive pricing while adhering to increasingly stringent environmental regulations. Furthermore, the use of stable boronate intermediates allows for potential stockpiling of key precursors, mitigating risks associated with raw material volatility and ensuring consistent supply continuity for downstream device manufacturers.
- Cost Reduction in Manufacturing: The elimination of difficult-to-separate byproducts significantly lowers the cost of goods sold by reducing purification time and solvent usage. Traditional methods often suffer from low effective yields due to the loss of material during the separation of symmetric isomers. By engineering the reaction to favor the cross-coupled product inherently, the process maximizes atom economy and reduces the overall consumption of expensive palladium catalysts and specialized ligands. This streamlined workflow allows for a more predictable cost structure, enabling better budget planning for large-scale procurement of organic semiconductor materials.
- Enhanced Supply Chain Reliability: The starting materials, primarily brominated and silylated thiophenes, are commercially accessible and can be sourced from multiple established chemical suppliers. This diversification of the supply base reduces dependency on single-source vendors and minimizes the risk of production stoppages due to raw material shortages. Additionally, the robustness of the synthetic steps ensures high batch-to-batch consistency, which is a critical requirement for qualifying materials in the highly regulated display and photovoltaic industries. Reliable delivery schedules can be maintained because the process is less susceptible to the variability often seen in complex multi-step organic syntheses.
- Scalability and Environmental Compliance: The process avoids the use of toxic tin reagents often found in Stille couplings, aligning with green chemistry principles and simplifying waste disposal protocols. The aqueous workup conditions typical of Suzuki reactions are easier to manage on a multi-kilogram scale compared to strictly anhydrous methods. This environmental compatibility not only reduces disposal costs but also facilitates regulatory approval for manufacturing facilities in regions with strict emission standards. The ability to scale this chemistry from gram to kilogram quantities without significant re-optimization makes it an ideal candidate for meeting the growing demand for high-performance organic electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of asymmetric heptathiophenes. These insights are derived directly from the patent specifications and are intended to clarify the advantages of this specific synthetic methodology for industry stakeholders. Understanding these nuances helps in making informed decisions regarding material selection and process integration.
Q: Why is the asymmetric structure of heptathiophene preferred over symmetric isomers?
A: Asymmetric heptathiophenes offer distinct molecular packing arrangements compared to symmetric linear or helical isomers. This structural asymmetry modulates intermolecular forces, potentially leading to improved charge carrier mobility and optimized energy levels for organic field-effect transistors and solar cells.
Q: What is the advantage of using Suzuki coupling in this synthesis route?
A: Traditional cross-coupling methods often yield mixtures of self-coupled and cross-coupled products that are difficult to separate due to similar polarity. The patented Suzuki strategy utilizing a specific boronate-bromide pairing significantly enhances selectivity for the desired cross-coupled intermediate, simplifying purification.
Q: What are the primary applications for these fused thiophene compounds?
A: These compounds serve as high-performance organic semiconductor materials. They are specifically designed for use in organic light-emitting diodes (OLEDs), organic field-effect transistors (OFETs), and organic photovoltaics (OPVs), demonstrating favorable mobility and switching ratios at room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Heptathiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced organic semiconductors play in the future of flexible electronics and renewable energy. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the asymmetric heptathiophene synthesis can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for OLED and OFET applications. Our commitment to quality assurance ensures that the electronic properties of the materials remain consistent, providing our partners with the reliability they need for their own product development cycles.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can accelerate your path to market with high-performance organic electronic materials.
