Optimizing Vilazodone Intermediate Production via Novel Catalytic Coupling and Reductive Amination Strategies
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex heterocyclic intermediates, particularly for central nervous system (CNS) agents like Vilazodone. Patent CN101163698B, published in May 2011, introduces a transformative methodology for the production of 5-(4-[4-(5-cyano-3-indolyl)-butyl]-1-piperazinyl)-benzofuran-2-carboxamide and its physiologically acceptable salts. This intellectual property discloses two distinct, highly effective synthetic strategies: a transition metal-catalyzed cross-coupling reaction and a reductive amination protocol. These approaches represent a significant departure from conventional multi-step syntheses, offering a streamlined route that minimizes operational complexity while maximizing yield and purity. For R&D directors and process chemists, understanding the nuances of these catalytic cycles is crucial for implementing robust manufacturing processes that meet stringent regulatory standards.
The core innovation lies in the strategic disconnection of the molecule, allowing for the late-stage assembly of the indole and benzofuran moieties. By leveraging modern palladium chemistry or classic reductive amination techniques, the patent provides a versatile toolkit for chemists aiming to optimize the supply chain for this critical antidepressant intermediate. The following analysis delves deep into the mechanistic underpinnings and commercial implications of these novel synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-(4-[4-(5-cyano-3-indolyl)-butyl]-1-piperazinyl)-benzofuran-2-carboxamide relied heavily on the alkylation of activated indole derivatives with halogenated benzofuran precursors or vice versa. Prior art, such as the methods cited in J. Med. Chem. (2004), often necessitated the use of reactive alkyl halides like 3-(4-chlorobutyl)indole-5-carbonitrile. These traditional alkylation pathways are fraught with challenges, including the potential for over-alkylation, the formation of quaternary ammonium salt by-products, and the requirement for rigorous purification steps to remove unreacted starting materials. Furthermore, conventional routes frequently involve lengthy synthetic sequences, sometimes extending up to ten discrete steps, which inherently accumulates material loss at each stage and drastically increases the cost of goods sold (COGS). The reliance on harsh alkylating agents also poses significant safety and environmental hazards, complicating waste management and requiring specialized containment infrastructure in manufacturing facilities.
The Novel Approach
In stark contrast, the methodology outlined in CN101163698B offers a paradigm shift by utilizing direct coupling or reductive amination to forge the critical carbon-nitrogen or carbon-carbon bonds. The first variant employs a palladium-catalyzed coupling between a 5-halobenzofuran-2-carboxamide and a piperazine-functionalized indole. This approach bypasses the need for unstable alkyl halides, utilizing stable aryl halides instead. 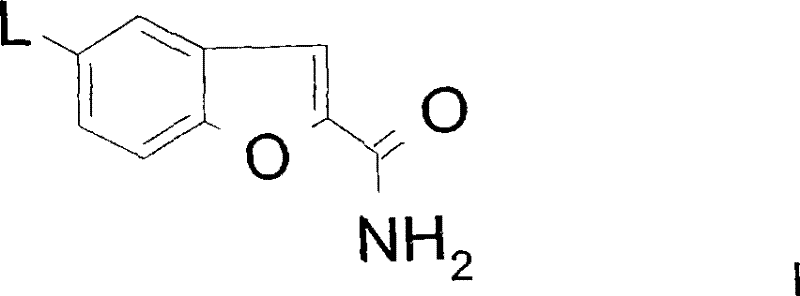 The second variant utilizes reductive amination between a benzofuran-piperazine base and an indole-aldehyde derivative. Both methods significantly reduce the step count, simplifying the overall process flow. The reduction in synthetic steps directly correlates to reduced solvent consumption, lower energy expenditure for heating and cooling cycles, and diminished labor costs. Moreover, the selectivity of these catalytic methods ensures a cleaner reaction profile, facilitating easier isolation of the final product through standard crystallization techniques rather than resource-intensive chromatography.
The second variant utilizes reductive amination between a benzofuran-piperazine base and an indole-aldehyde derivative. Both methods significantly reduce the step count, simplifying the overall process flow. The reduction in synthetic steps directly correlates to reduced solvent consumption, lower energy expenditure for heating and cooling cycles, and diminished labor costs. Moreover, the selectivity of these catalytic methods ensures a cleaner reaction profile, facilitating easier isolation of the final product through standard crystallization techniques rather than resource-intensive chromatography.
Mechanistic Insights into Pd-Catalyzed Coupling and Reductive Amination
The palladium-catalyzed variant (Method A) operates through a classic Buchwald-Hartwig amination mechanism, albeit adapted for the specific steric and electronic properties of the benzofuran and indole systems. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the 5-halobenzofuran-2-carboxamide, forming an aryl-palladium(II) complex. This step is facilitated by the electron-withdrawing nature of the carboxamide group, which activates the aryl ring towards oxidative addition. Subsequently, the piperazine nitrogen of the indole derivative coordinates to the palladium center, followed by deprotonation by a base such as sodium tert-butoxide. This generates a nucleophilic amido-palladium species. The cycle concludes with reductive elimination, which releases the coupled product and regenerates the active palladium(0) catalyst. The choice of ligand, such as tri-tert-butylphosphine, is critical here, as it stabilizes the low-valent palladium species and accelerates the reductive elimination step, ensuring high turnover numbers and minimizing catalyst loading requirements.
Alternatively, the reductive amination pathway (Method B) proceeds through the formation of an iminium ion intermediate. The reaction begins with the condensation of the primary or secondary amine group on the benzofuran-piperazine scaffold with the ketone functionality of 3-(4-oxo-butyl)-1H-indole-5-carbonitrile. 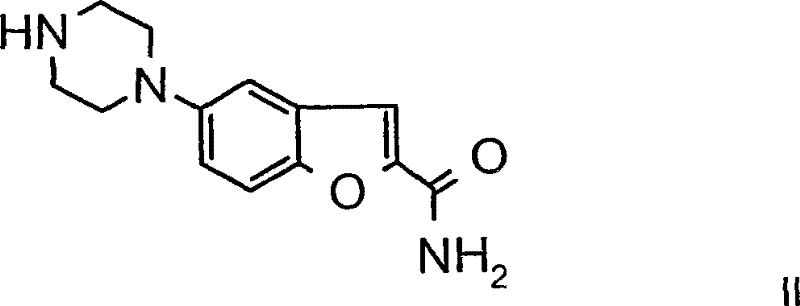 This equilibrium process yields an imine or iminium species, which is then selectively reduced by a hydride source, such as sodium cyanoborohydride. The use of a mild reducing agent is paramount to prevent the reduction of other sensitive functional groups, such as the nitrile group on the indole ring or the amide on the benzofuran. This chemoselectivity is a major advantage, as it prevents the formation of alcohol by-products or reduced nitrile impurities. The mechanism ensures that the butyl linker is formed with high fidelity, maintaining the structural integrity required for biological activity. Impurity control is further enhanced by the reversibility of the imine formation, which allows the system to self-correct towards the thermodynamically stable product before the irreversible reduction step locks the structure in place.
This equilibrium process yields an imine or iminium species, which is then selectively reduced by a hydride source, such as sodium cyanoborohydride. The use of a mild reducing agent is paramount to prevent the reduction of other sensitive functional groups, such as the nitrile group on the indole ring or the amide on the benzofuran. This chemoselectivity is a major advantage, as it prevents the formation of alcohol by-products or reduced nitrile impurities. The mechanism ensures that the butyl linker is formed with high fidelity, maintaining the structural integrity required for biological activity. Impurity control is further enhanced by the reversibility of the imine formation, which allows the system to self-correct towards the thermodynamically stable product before the irreversible reduction step locks the structure in place.
How to Synthesize 5-(4-[4-(5-cyano-3-indolyl)-butyl]-1-piperazinyl)-benzofuran-2-carboxamide Efficiently
Implementing these synthetic routes requires careful attention to reaction parameters, particularly temperature and stoichiometry, to ensure optimal conversion and minimal by-product formation. For the coupling route, maintaining an inert atmosphere is essential to prevent oxidation of the phosphine ligands and the palladium catalyst. The reaction typically proceeds in polar aprotic solvents like diglyme at elevated temperatures, ensuring complete dissolution of the organic substrates and efficient mass transfer. For the reductive amination route, pH control is critical during the workup phase to ensure the precipitation of the desired hydrochloride salt while keeping impurities in the aqueous phase. The patent details specific embodiments that serve as a robust foundation for scale-up, demonstrating that high purity can be achieved without resorting to preparative HPLC.
- Prepare the reaction vessel under inert atmosphere and charge with the appropriate palladium catalyst system (e.g., Pd2(dba)3 with P(t-Bu)3) or reducing agent (e.g., sodium cyanoborohydride) depending on the chosen route.
- Introduce the core building blocks: either react 5-halobenzofuran-2-carboxamide with 3-(4-piperazin-1-yl-butyl)-indole-5-carbonitrile for coupling, or react the benzofuran-piperazine base with 3-(4-oxo-butyl)-1H-indole-5-carbonitrile for reductive amination.
- Maintain precise temperature control (0°C to 120°C range) and reaction times, followed by standard aqueous workup, pH adjustment, and crystallization to isolate the high-purity hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of the methodologies described in CN101163698B offers substantial strategic benefits. The shift away from complex, multi-step alkylation sequences to direct coupling or reductive amination fundamentally alters the cost structure of manufacturing this intermediate. By reducing the number of unit operations, manufacturers can significantly lower the capital expenditure required for reactor occupancy time and utility consumption. This efficiency translates directly into a more competitive pricing model for the final API, providing a buffer against raw material price volatility. Furthermore, the use of widely available starting materials, such as halogenated benzofurans and piperazine derivatives, mitigates supply risk associated with bespoke, hard-to-source alkylating agents.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous alkylating agents, combined with the reduction in synthetic steps, leads to a drastic simplification of the production process. Fewer steps mean fewer opportunities for yield loss, less solvent usage for extractions and washes, and reduced waste disposal costs. The ability to use standard palladium catalysts or common reducing agents, which are available in bulk quantities from reliable chemical suppliers, further drives down the variable costs associated with production. This streamlined approach allows for a leaner manufacturing footprint, optimizing the utilization of existing facility infrastructure without the need for specialized equipment modifications.
- Enhanced Supply Chain Reliability: The robustness of the described chemical routes enhances supply chain continuity. Traditional methods relying on unstable intermediates often suffer from batch-to-batch variability and short shelf-lives, creating bottlenecks in production scheduling. In contrast, the stable precursors used in these novel methods can be stockpiled effectively, ensuring a steady feed of materials for continuous manufacturing campaigns. The simplified purification protocols, which rely on crystallization rather than chromatography, are inherently more scalable and less prone to technical failures, guaranteeing consistent delivery timelines to downstream API manufacturers.
- Scalability and Environmental Compliance: The processes described are designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale industrial reactors. The reduction in solvent volume and the avoidance of toxic alkyl halides align with green chemistry principles, reducing the environmental burden of the manufacturing process. This compliance with increasingly stringent environmental regulations minimizes the risk of production shutdowns due to waste treatment limitations. Additionally, the high selectivity of the reactions reduces the load on wastewater treatment plants, lowering the overall environmental compliance costs for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these synthetic routes. They are derived from a detailed analysis of the patent specifications and are intended to clarify the operational feasibility and quality implications of adopting these technologies. Understanding these aspects is vital for technical teams evaluating the transition from legacy processes to these advanced methodologies.
Q: What are the primary advantages of the Pd-catalyzed coupling route over traditional alkylation?
A: The Pd-catalyzed coupling route described in CN101163698B eliminates the need for harsh alkylating agents like 3-(4-chlorobutyl)indole derivatives. This results in a significantly shorter synthetic sequence compared to prior art methods which often require up to 10 steps, thereby reducing overall process complexity and potential impurity formation.
Q: How does the reductive amination method improve impurity profiles?
A: By utilizing 3-(4-oxo-butyl)-1H-indole-5-carbonitrile as the electrophile in a reductive amination with the benzofuran-piperazine core, the process avoids the generation of quaternary ammonium salts and other alkylation by-products common in traditional nucleophilic substitution reactions, leading to a cleaner crude profile.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly highlights the use of robust conditions and common solvents like diglyme, methanol, and dichloromethane. The simplified workup procedures, involving standard extraction and crystallization rather than complex chromatography for bulk production, indicate high scalability for industrial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(4-[4-(5-cyano-3-indolyl)-butyl]-1-piperazinyl)-benzofuran-2-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates. Our technical team has extensively analyzed the methodologies presented in CN101163698B and possesses the expertise to translate these laboratory-scale innovations into commercial reality. We offer comprehensive process development services tailored to optimize these coupling and reductive amination reactions for your specific production needs. With our state-of-the-art facilities, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply requirements are met with precision and consistency. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which employ advanced analytical techniques to verify the identity and purity of every batch.
We invite you to collaborate with us to leverage these advanced synthetic strategies for your Vilazodone supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to these optimized routes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of this essential intermediate, driving value and efficiency throughout your organization.
