Advanced Manufacturing Strategy for Pralsetinib via Streamlined Rieke Zinc Coupling
Advanced Manufacturing Strategy for Pralsetinib via Streamlined Rieke Zinc Coupling
The rapidly evolving landscape of oncology therapeutics demands robust, scalable, and highly efficient synthetic routes for next-generation kinase inhibitors. Patent CN111440151A, published on July 24, 2020, introduces a groundbreaking methodology for the preparation of Pralsetinib, a potent and selective RET kinase inhibitor currently under investigation for the treatment of non-small cell lung cancer (NSCLC) and medullary thyroid carcinoma (MTC). This intellectual property disclosure outlines a concise three-step synthetic pathway that markedly reduces operational complexity while enhancing the total yield and purity profile of the final active pharmaceutical ingredient (API). By leveraging advanced organometallic coupling techniques and optimized protection group strategies, this route addresses critical bottlenecks often encountered in the commercial manufacturing of complex heterocyclic drugs. For global supply chain stakeholders, this technology represents a significant leap forward in process chemistry, offering a viable solution for the reliable production of high-purity pharmaceutical intermediates.
As a leading entity in the fine chemical sector, understanding the nuances of such patented methodologies is essential for maintaining a competitive edge as a reliable API intermediate supplier. The disclosed route not only streamlines the synthesis but also introduces a novel intermediate, designated as compound 4, which had not been previously reported in the literature. This strategic innovation allows for better control over impurity profiles and facilitates easier scale-up from laboratory benchtop to multi-ton industrial reactors. The following analysis dissects the technical merits of this approach, providing R&D directors and procurement managers with the insights necessary to evaluate its potential for integration into existing manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing complex kinase inhibitors like Pralsetinib often suffer from excessive step counts, harsh reaction conditions, and cumbersome purification protocols that erode overall process efficiency. Conventional methods frequently rely on multiple protection and deprotection sequences that introduce unnecessary mass intensity and waste generation, thereby inflating the cost of goods sold (COGS). Furthermore, prior art routes often necessitate the use of silica gel column chromatography for intermediate purification, a technique that is notoriously difficult to translate from milligram-scale discovery chemistry to kilogram-scale commercial production due to solvent consumption and throughput limitations. The reliance on such labor-intensive purification steps can lead to significant product loss and variability in batch-to-batch consistency, posing substantial risks for supply chain continuity. Additionally, older methodologies may utilize less selective coupling reagents or catalysts that generate difficult-to-remove metal impurities, requiring expensive scavenging processes to meet stringent regulatory standards for residual metals in drug substances.
The Novel Approach
In stark contrast to these legacy challenges, the method disclosed in CN111440151A employs a highly convergent three-step strategy that maximizes atom economy and operational simplicity. The core of this innovation lies in the initial construction of the biaryl-like linkage using a Rieke zinc-mediated cross-coupling reaction, which proceeds under relatively mild thermal conditions compared to traditional Grignard or lithiation protocols. This novel approach effectively bypasses the need for cryogenic temperatures and minimizes side reactions associated with highly reactive organometallic species. Subsequent steps involve a straightforward hydrolysis to reveal the key carboxylic acid functionality followed by a final amide condensation, both of which are executed with high selectivity and yield. By designing the route to utilize acid-base extraction workups instead of chromatographic separation for the first intermediate, the inventors have created a process that is inherently more amenable to large-scale manufacturing. This shift from chromatographic to crystallization or extraction-based purification is a hallmark of mature process chemistry, directly translating to reduced manufacturing lead times and lower solvent recovery costs.
Mechanistic Insights into Rieke Zinc-Mediated Cross-Coupling
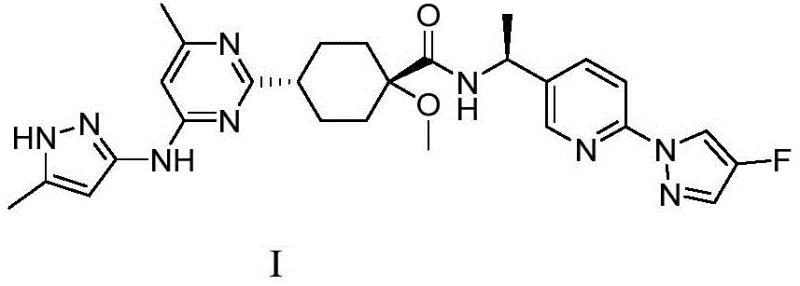
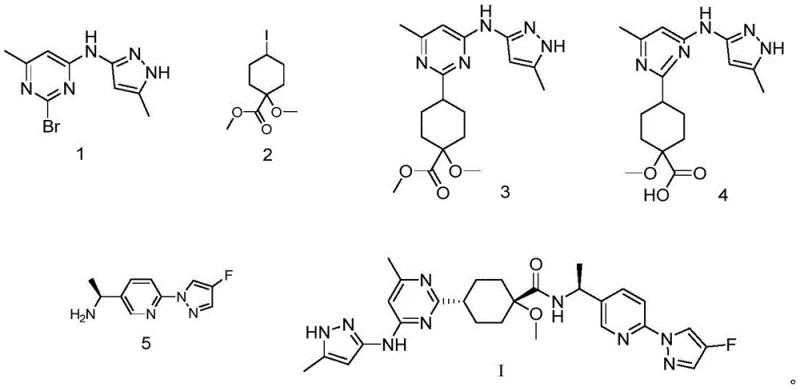
The cornerstone of this synthetic strategy is the first step, which involves the coupling of a brominated pyrimidine derivative (Compound 1, CAS: 2054922-17-5) with a functionalized cyclohexyl ester (Compound 2, CAS: 2097133-18-9). This transformation is catalyzed by a palladium complex, specifically [1,1'-bis(diphenylphosphino)ferrocene]palladium(II) dichloride (PdCl2(dppf)), in the presence of Rieke zinc. The use of Rieke zinc, a highly active form of zinc metal, facilitates the oxidative addition into the carbon-bromine bond at moderate temperatures ranging from 75°C to 85°C, typically optimized at 80°C. The reaction mechanism likely proceeds through the formation of an organozinc species in situ, which then undergoes transmetallation with the palladium catalyst before reductive elimination yields the coupled product, Compound 3. Experimental data from the patent indicates that maintaining a precise molar ratio of reactants, specifically a 1.1:1.1:1 ratio of Compound 1 to Rieke zinc to Compound 2, is critical for maximizing yield, which can reach up to 77.9% under optimal conditions. This careful stoichiometric balance prevents the formation of homocoupling byproducts and ensures high conversion of the valuable brominated starting material.
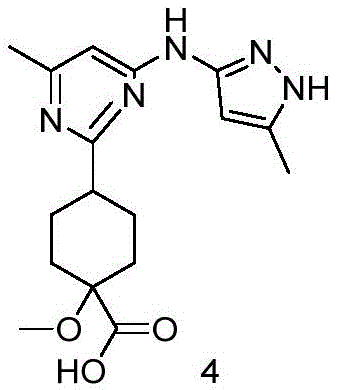
Following the coupling, the process moves to the hydrolysis of the dimethyl ester moiety in Compound 3 to generate the mono-acid Intermediate 4, a novel compound central to this invention. This step utilizes lithium hydroxide (LiOH) in a tetrahydrofuran (THF) and water mixture at 60°C, a condition chosen to selectively hydrolyze the ester without compromising the integrity of the sensitive heterocyclic rings. The patent highlights a crucial mechanistic advantage here: the reaction workup does not require pH-adjusted extraction, which is often problematic for molecules containing both carboxyl and methylene groups due to solubility issues. Instead, the product precipitates or is isolated directly upon concentration, avoiding product loss and simplifying the isolation protocol. The final step involves the amide bond formation between Intermediate 4 and the chiral amine fragment (Compound 5, CAS: 1980023-96-8) using HATU as the coupling agent and DIPEA as the base. This peptide-coupling style reaction is highly efficient, proceeding at room temperature to yield the final Pralsetinib molecule with an impressive HPLC purity exceeding 99.6%, demonstrating the robustness of the entire synthetic sequence.
How to Synthesize Pralsetinib Efficiently
The synthesis of Pralsetinib via this patented route offers a clear blueprint for process chemists aiming to establish a scalable manufacturing line. The procedure begins with the activation of zinc and the subsequent palladium-catalyzed coupling in DMF, followed by a straightforward aqueous workup to isolate the coupled ester. The second stage involves a controlled hydrolysis using lithium hydroxide to unveil the carboxylic acid handle, which is then immediately ready for the final conjugation. The detailed standardized synthesis steps, including specific reagent quantities, temperature profiles, and workup procedures derived from the patent examples, are provided below to guide technical implementation.
- Perform a palladium-catalyzed coupling between compound 1 and compound 2 using Rieke zinc and PdCl2(dppf) in DMF at 80°C to obtain compound 3.
- Hydrolyze the ester group of compound 3 using lithium hydroxide (LiOH) in THF at 60°C to generate the key intermediate compound 4.
- Condense intermediate compound 4 with amine compound 5 using HATU and DIPEA in DMF at room temperature to yield Pralsetinib with >99.6% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reduction in reaction steps from potentially longer linear sequences to just three convergent steps significantly compresses the manufacturing timeline, allowing for faster response to market demand fluctuations. The elimination of silica gel chromatography in the early stages of the synthesis is a major cost driver, as it removes the need for large volumes of chromatographic grade solvents and the associated disposal costs, leading to substantial cost savings in manufacturing operations. Furthermore, the use of commercially available starting materials with known CAS numbers ensures a stable supply chain, reducing the risk of raw material shortages that can plague custom synthesis projects. The high yields reported in the patent examples, particularly the near-quantitative yield in the hydrolysis step, minimize raw material waste and maximize the throughput of the final API, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by replacing expensive and time-consuming chromatographic purifications with simple acid-base extractions and filtrations. This operational shift drastically reduces solvent consumption and labor hours required for purification, resulting in a lower cost per kilogram of the produced intermediate. Additionally, the high atom economy of the Rieke zinc coupling ensures that expensive palladium catalysts and specialized reagents are utilized with maximum efficiency, further driving down the variable costs associated with production.
- Enhanced Supply Chain Reliability: By relying on robust, commercially sourced building blocks such as the brominated pyrimidine and the functionalized cyclohexyl ester, the supply chain becomes more resilient to disruptions. The mild reaction conditions, operating primarily between 60°C and 80°C, reduce the energy load on manufacturing facilities and decrease the wear and tear on reactor equipment, ensuring consistent production capacity over time. The ability to produce the key intermediate, Compound 4, as a stable, isolable solid also allows for strategic stockpiling, decoupling the production of the final drug from the immediate availability of the chiral amine fragment.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, utilizing common organic solvents like DMF, THF, and ethyl acetate that are easily recovered and recycled in modern pharmaceutical plants. The avoidance of cryogenic conditions and hazardous reagents simplifies the safety profile of the process, making it easier to scale from pilot plant batches to multi-ton commercial production without extensive re-engineering. The high purity of the final product (>99.6%) reduces the burden on downstream formulation teams and ensures compliance with strict regulatory guidelines for impurity limits, facilitating a smoother path to market approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Pralsetinib. These answers are derived directly from the experimental data and beneficial effects described in CN111440151A, providing clarity on the feasibility and advantages of this specific manufacturing route for industry stakeholders.
Q: What is the achieved purity of Pralsetinib using this novel synthetic route?
A: According to patent CN111440151A, the final Pralsetinib product obtained via this 3-step method achieves an HPLC purity of greater than 99.6%, demonstrating exceptional quality control suitable for pharmaceutical applications.
Q: What is the key novel intermediate identified in this process?
A: The process identifies the compound of formula 4 as a critical new intermediate. This carboxylic acid derivative is synthesized via hydrolysis of the dimethyl ester precursor and serves as the direct coupling partner for the final amide bond formation.
Q: How does this method improve purification efficiency compared to conventional routes?
A: The method eliminates the need for silica gel column chromatography in the first step by utilizing acid-base extraction properties of the imine salt. Additionally, the hydrolysis step avoids pH-adjusted extraction losses, significantly simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pralsetinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-quality synthesis routes in the competitive oncology drug market. Our team of expert process chemists has thoroughly analyzed the methodology presented in CN111440151A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. We are committed to delivering Pralsetinib and its key intermediates with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the highest international standards. Our state-of-the-art facilities are equipped to handle the specific requirements of Rieke zinc chemistry and palladium-catalyzed couplings, ensuring a seamless transition from development to full-scale manufacturing.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your project benefits from the most efficient and cost-effective manufacturing strategy available in the market.
