Advanced Synthesis of 2-Fluoro-5-[(4-Oxo-3H-2,3-Diazanaphthyl)Methyl]Benzoic Acid for Commercial Scale-Up
Introduction to Patent CN112624981B and Strategic Value
The pharmaceutical industry's relentless pursuit of efficient oncology treatments has placed significant spotlight on Poly (ADP-ribose) polymerase (PARP) inhibitors, with Olaparib standing as a cornerstone therapy for BRCA-mutated cancers. Central to the supply chain of this critical medication is the high-quality production of its key building block, 2-fluoro-5-[(4-oxo-3H-2,3-diazanaphthyl)methyl]benzoic acid. Patent CN112624981B, published in May 2021, introduces a transformative four-step synthetic methodology that addresses longstanding bottlenecks in yield and purity associated with traditional manufacturing routes. This technical disclosure outlines a robust pathway starting from o-carboxybenzaldehyde, leveraging a unique phosphonate intermediate to drive a Horner-Wadsworth-Emmons olefination, followed by a strategic palladium-catalyzed carbonylation. For global procurement and R&D teams, this patent represents a pivotal shift towards more sustainable and cost-effective API intermediate sourcing, promising to stabilize supply chains for one of the most vital anticancer agents on the market today.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical Olaparib intermediate has been plagued by significant chemical and economic inefficiencies that hinder large-scale production. One prominent prior art route, documented in J. Med. Chem. 2008, relies on a Horner-Wadsworth-Emmons reaction involving a nitrile-containing aldehyde, which subsequently requires harsh alkaline hydrolysis to convert the cyano group into the necessary carboxylic acid. This approach frequently suffers from extensive side reactions due to the sensitivity of the nitrile functionality, resulting in complex impurity profiles that are difficult and expensive to purge. Furthermore, an alternative route described in patent CN105820126B utilizes a Suzuki-Miyaura coupling strategy that necessitates the use of costly catechol borane reagents and expensive palladium catalysts. The reliance on these precious metal systems introduces severe downstream challenges, particularly regarding the rigorous removal of palladium residues to meet stringent pharmaceutical safety standards, thereby inflating both processing time and overall manufacturing costs significantly.
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN112624981B proposes a streamlined and chemically elegant solution that bypasses the problematic nitrile hydrolysis and expensive boronation steps entirely. The new strategy initiates with the formation of a stable phosphonate ester from o-carboxybenzaldehyde, which then undergoes olefination with 3-bromo-4-fluorobenzaldehyde to install the carbon backbone with high stereocontrol. Crucially, the route employs a palladium-catalyzed carbonylation reaction to directly convert the aryl bromide into the methyl ester, a transformation that is atom-economical and avoids the introduction of extraneous functional groups requiring later removal. This methodology not only simplifies the purification process through straightforward precipitation and pulping techniques but also ensures that the final cyclization with hydrazine hydrate proceeds with exceptional cleanliness. By eliminating the need for difficult post-treatment steps associated with cyano hydrolysis or boron residue clearance, this novel approach delivers a superior impurity profile and substantially lowers the barrier for industrial implementation.
![Final cyclization step converting the methyl ester intermediate to 2-fluoro-5-[(4-oxo-3H-2,3-diazanaphthyl)methyl]benzoic acid using hydrazine hydrate](/insights/img/olaparib-intermediate-synthesis-pharma-supplier-20260305121731-014.png)
Mechanistic Insights into Palladium-Catalyzed Carbonylation and HWE Olefination
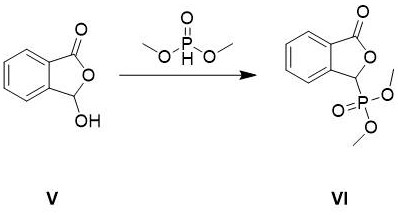
The core chemical innovation of this process lies in the precise orchestration of the Horner-Wadsworth-Emmons (HWE) reaction coupled with a subsequent carbonylation cycle, which together define the efficiency of the carbon skeleton construction. The initial formation of the (3-oxo-1,3-dihydroisobenzofuran-1-yl)dimethyl phosphate creates a highly reactive nucleophile that selectively attacks the aldehyde carbonyl of the fluoro-bromo benzaldehyde derivative. This step is critical because the resulting alkene linkage establishes the geometric foundation for the final phthalazinone ring system, and the use of the phosphonate moiety ensures that the reaction proceeds under mild basic conditions without epimerization or degradation of sensitive functional groups. Following this, the palladium-catalyzed carbonylation step operates through a classic oxidative addition-reductive elimination cycle, where the Pd(0) species inserts into the carbon-bromine bond of the intermediate. Under a pressurized carbon monoxide atmosphere, the resulting organopalladium complex undergoes migratory insertion of CO, followed by methanolysis to release the methyl ester product and regenerate the active catalyst. This mechanistic pathway is far superior to cross-coupling with boronates because it utilizes inexpensive carbon monoxide gas as the C1 source rather than pre-functionalized organometallic reagents, thereby reducing the atomic waste and chemical load on the reaction system.
From an impurity control perspective, this synthetic design inherently minimizes the generation of hard-to-remove byproducts that typically plague Olaparib intermediate synthesis. In traditional routes involving nitrile hydrolysis, the harsh alkaline conditions often lead to the formation of ring-opened byproducts or over-hydrolyzed species that co-elute with the desired product during crystallization. By contrast, the carbonylation route described here operates at a moderate temperature of 80°C in a methanol solvent system, which preserves the integrity of the lactone ring and prevents unwanted decomposition. Furthermore, the final cyclization with hydrazine hydrate is executed in a tetrahydrofuran and water mixture, allowing for the selective formation of the diazanaphthyl ring while simultaneously hydrolyzing the methyl ester to the free acid in a telescoped manner. This convergence of steps reduces the number of isolation points, which is a primary driver of yield loss in multi-step syntheses, and ensures that the final product precipitates as a high-purity white solid upon pH adjustment, effectively excluding non-polar organic impurities and residual catalysts from the crystal lattice.
How to Synthesize 2-Fluoro-5-[(4-Oxo-3H-2,3-Diazanaphthyl)Methyl]Benzoic Acid Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the carbonylation and cyclization stages, to maximize yield and ensure reproducibility on a commercial scale. The process begins with the activation of o-carboxybenzaldehyde using triethylamine and dimethyl phosphite in dichloromethane, followed by acidification to isolate the phosphonate intermediate as a stable white solid. This solid is then reacted with 3-bromo-4-fluorobenzaldehyde under controlled cooling to manage the exotherm of the olefination, ensuring high conversion before proceeding to the high-pressure carbonylation step. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures optimized for industrial throughput, are provided in the guide below for technical reference.
- Preparation of (3-oxo-1,3-dihydroisobenzofuran-1-yl)dimethyl phosphate via reaction of o-carboxybenzaldehyde with dimethyl phosphite.
- Horner-Wadsworth-Emmons reaction with 3-bromo-4-fluorobenzaldehyde to form the bromo-substituted isobenzofuran intermediate.
- Palladium-catalyzed carbonylation under carbon monoxide pressure to convert the bromide to the methyl ester.
- Cyclization with hydrazine hydrate followed by hydrolysis to yield the final 2-fluoro-5-[(4-oxo-3H-2,3-diazanaphthyl)methyl]benzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend well beyond simple chemical yield improvements. The primary economic benefit stems from the complete elimination of expensive boron-based reagents, such as catechol borane, which are not only costly to purchase but also introduce significant hazards and waste disposal costs due to their reactivity. By replacing the Suzuki coupling with a carbonylation reaction that utilizes carbon monoxide gas, the process drastically reduces the raw material bill of materials (BOM) and removes the necessity for specialized handling equipment required for pyrophoric or moisture-sensitive organoboron compounds. Additionally, the simplified purification protocol, which relies on pulping and filtration rather than column chromatography or complex extraction sequences, significantly lowers the consumption of organic solvents and reduces the volume of hazardous waste generated per kilogram of product. These factors combine to create a manufacturing process that is not only more cost-effective but also more environmentally compliant, aligning with the increasing regulatory pressures on pharmaceutical supply chains to adopt greener chemistry principles.
- Cost Reduction in Manufacturing: The economic impact of this new route is driven by the substitution of high-cost coupling partners with commodity chemicals like carbon monoxide and methanol. In traditional Suzuki couplings, the stoichiometric requirement for boron reagents and the associated ligand systems creates a substantial cost burden that scales linearly with production volume. This new method circumvents those expenses entirely, while the mild reaction conditions reduce energy consumption for heating and cooling. Furthermore, the high purity of the intermediates allows for direct use in subsequent steps without extensive recrystallization, saving both time and solvent costs. The cumulative effect is a significant reduction in the cost of goods sold (COGS) for the final API, providing a competitive edge in pricing negotiations for generic Olaparib formulations.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on widely available starting materials such as o-carboxybenzaldehyde and 3-bromo-4-fluorobenzaldehyde, which are produced by multiple global suppliers and are not subject to the same geopolitical or capacity constraints as specialized organometallic reagents. The robustness of the synthetic steps, characterized by their tolerance to minor variations in temperature and stoichiometry, ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. Moreover, the stability of the key phosphonate intermediate allows for potential stockpiling or campaign manufacturing, decoupling the production of the final acid from the immediate availability of upstream precursors. This flexibility is crucial for maintaining continuous supply to API manufacturers, especially during periods of high market demand or logistical disruptions.
- Scalability and Environmental Compliance: From an operational standpoint, the process is exceptionally well-suited for scale-up due to the absence of difficult-to-handle reagents and the simplicity of the workup procedures. The carbonylation step, while requiring pressure equipment, is a standard unit operation in modern fine chemical plants, and the use of heterogeneous filtration for product isolation facilitates automation and reduces manual labor requirements. Environmentally, the reduction in heavy metal usage (specifically avoiding the high palladium loads often needed for difficult Suzuki couplings) simplifies the wastewater treatment process and lowers the burden on effluent management systems. The overall E-factor of the process is improved through higher yields and reduced solvent usage, making it easier for manufacturing sites to meet increasingly strict environmental, health, and safety (EHS) regulations without compromising production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and comparative analysis presented in the patent documentation. These insights are intended to clarify the operational benefits and chemical rationale behind the new methodology for stakeholders evaluating its adoption. Understanding these nuances is essential for making informed decisions about technology transfer and vendor qualification.
Q: How does this new synthesis route improve upon previous methods for Olaparib intermediates?
A: The new route described in patent CN112624981B avoids the use of expensive catechol borane reagents and complex cyano hydrolysis steps found in prior art. By utilizing a palladium-catalyzed carbonylation strategy instead of Suzuki coupling with boronates, it significantly reduces raw material costs and simplifies the removal of metal residues, leading to higher purity and easier industrial scale-up.
Q: What are the critical reaction conditions for the carbonylation step?
A: The carbonylation step requires an autoclave environment with a carbon monoxide internal pressure maintained at approximately 50 Psi. The reaction utilizes a Pd(dppf)Cl2 catalyst in methanol with sodium acetate as a base, heated to 80°C. These mild conditions ensure high conversion rates while minimizing side reactions associated with harsher alkaline hydrolysis methods used in older synthetic pathways.
Q: Is this intermediate suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process is specifically designed for industrial viability. The intermediates are solids that are easily purified by pulping and filtration rather than complex chromatography. The avoidance of difficult-to-remove heavy metal residues and the use of readily available starting materials like o-carboxybenzaldehyde make it highly scalable for commercial production of Olaparib APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-[(4-Oxo-3H-2,3-Diazanaphthyl)Methyl]Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patent CN112624981B are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-fluoro-5-[(4-oxo-3H-2,3-diazanaphthyl)methyl]benzoic acid meets the exacting standards required for oncology drug substance manufacturing. Our commitment to quality assurance extends beyond simple compliance; we proactively monitor impurity profiles to ensure they remain well below regulatory thresholds, safeguarding the safety and efficacy of the final therapeutic product.
We invite global pharmaceutical companies and contract manufacturing organizations to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your volume requirements and regional logistics. We encourage you to contact us directly to obtain specific COA data from our pilot batches and to receive comprehensive route feasibility assessments tailored to your project timelines. Let us collaborate to secure a stable, high-quality, and cost-efficient supply of this critical Olaparib intermediate for your future production needs.
