Advanced Microwave-Assisted Synthesis of Adavosertib for Commercial Scale-Up
Advanced Microwave-Assisted Synthesis of Adavosertib for Commercial Scale-Up
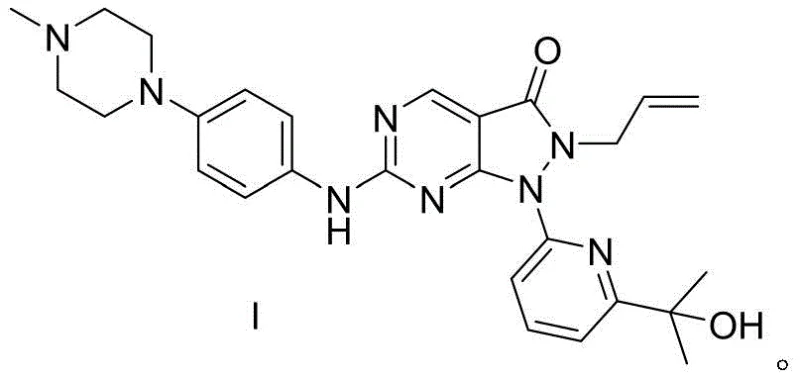
The pharmaceutical landscape for oncology treatments continues to evolve with the development of potent kinase inhibitors, among which Adavosertib (AZD-1775) stands out as a critical Wee1 inhibitor currently in advanced clinical trials for various solid tumors. As detailed in patent CN113735863A, a novel preparation process has been disclosed that addresses significant bottlenecks in the manufacturing of this high-value active pharmaceutical ingredient (API). This innovative synthetic route leverages microwave technology and optimized reagent selection to achieve superior total yields and enhanced industrial operability compared to prior art methods. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the technical nuances of this pathway is essential for securing a robust supply chain. The process outlines a streamlined four-step sequence that transforms readily available starting materials into the complex heterocyclic structure of Adavosertib with remarkable efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Wee1 inhibitors often suffer from cumbersome reaction conditions that hinder large-scale production and inflate manufacturing costs. Conventional methodologies frequently rely on the use of hazardous and expensive strong bases, such as sodium hydride (NaH), which require stringent anhydrous conditions and pose significant safety risks during scale-up. Furthermore, standard thermal heating methods for cyclization steps can be excessively time-consuming, often requiring several hours to days to reach completion, which drastically reduces throughput in a commercial setting. Purification processes in older protocols typically involve multiple column chromatography steps for intermediates, leading to substantial solvent consumption and product loss. These factors collectively contribute to a high cost of goods sold (COGS) and extended lead times, creating vulnerabilities in the supply chain for high-purity Adavosertib. Additionally, the formation of difficult-to-remove impurities, particularly those arising from non-selective halogenation or incomplete deprotection, often compromises the final quality of the API.
The Novel Approach
The methodology presented in the patent data introduces a paradigm shift by integrating microwave-assisted synthesis and greener reagent choices to overcome these historical challenges. A standout feature of this novel approach is the solvent-free microwave cyclization in the initial step, which completes the formation of the pyrazolopyrimidine core in merely 5 to 6 minutes with yields reaching up to 96%. This dramatic reduction in reaction time not only accelerates the overall process but also eliminates the need for large volumes of organic solvents at this stage. Subsequent alkylation steps strategically replace costly sodium hydride with inexpensive potassium carbonate (K2CO3), significantly lowering raw material expenses while maintaining high reaction efficiency. The process also employs a clever deprotection strategy using bromomethyl phenyl propane, which facilitates the removal of protecting groups under mild reflux conditions. By utilizing simple pulping purification techniques for intermediates rather than complex chromatography, the workflow becomes far more amenable to commercial scale-up of complex heterocycles, ensuring a more consistent and economical production cycle.
Mechanistic Insights into Microwave-Assisted Cyclization and Selective Alkylation
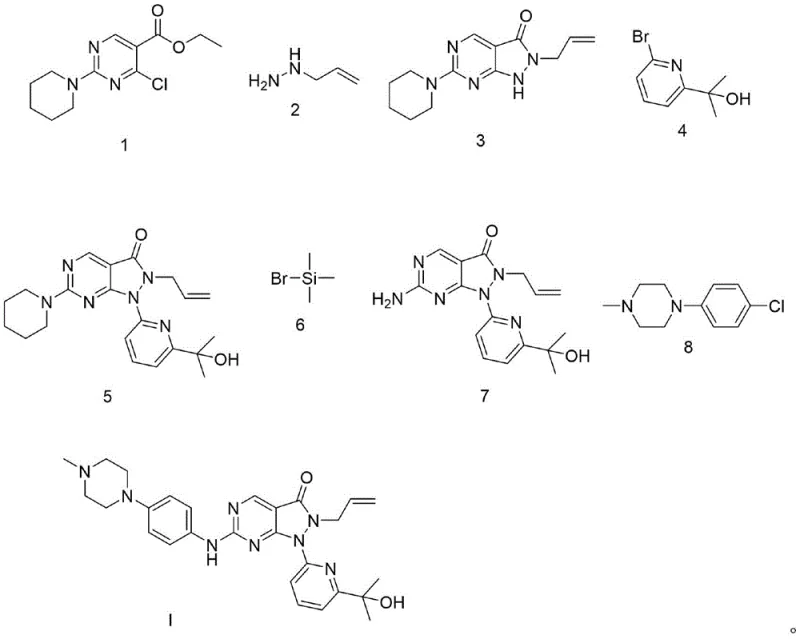
The core of this synthetic innovation lies in the mechanistic efficiency of the microwave-assisted cyclization between the chloro-pyrimidine ester (Formula 1) and hydrazine (Formula 2). Under microwave irradiation at 400W, the dipolar polarization of the reactants generates rapid internal heating, which lowers the activation energy required for the nucleophilic attack and subsequent ring closure. This mechanism allows for the rapid formation of the pyrazolo[3,4-d]pyrimidin-one core (Formula 3) without the thermal degradation often seen in prolonged conventional heating. The absence of solvent in this step further concentrates the reactants, driving the equilibrium towards product formation and minimizing side reactions. Following this, the N-alkylation with the bromo-pyridine derivative (Formula 4) proceeds via a classic SN2 mechanism, where the mild base K2CO3 effectively deprotonates the nitrogen without promoting elimination side reactions. This selectivity is crucial for maintaining the integrity of the sensitive allyl group present in the molecule.
Impurity control is rigorously managed through the specific choice of reagents in the later stages of the synthesis. In Step 4, the coupling of the amine intermediate (Formula 7) with the piperazine derivative (Formula 8) is conducted using K2CO3 in DMF at a controlled temperature of 60°C. This specific condition is vital to prevent the unwanted halogenation of the hydroxyl group on the side chain, a common impurity in less optimized routes. The patent data indicates that this selective alkylation ensures that the final product retains its structural fidelity, resulting in an HPLC purity of 99.5%. The deprotection step (Step 3) utilizes an excess of bromomethyl phenyl propane to drive the removal of the amino protecting group to completion, ensuring that no protected intermediates carry over into the final coupling step. This meticulous attention to reaction parameters and reagent stoichiometry demonstrates a deep understanding of the chemical kinetics involved, providing a robust framework for cost reduction in API manufacturing by minimizing waste and rework.
How to Synthesize Adavosertib Efficiently
The synthesis of Adavosertib via this patented route is designed for operational simplicity and high throughput, making it an ideal candidate for technology transfer to manufacturing sites. The process begins with the rapid microwave cyclization to build the core scaffold, followed by sequential functionalization steps that introduce the necessary side chains with high precision. Each step has been optimized to utilize common laboratory and plant equipment, such as standard microwave reactors and stirred tank reactors, avoiding the need for specialized high-pressure or cryogenic setups. The workup procedures are equally streamlined, relying largely on filtration and pulping, which are unit operations that scale linearly and predictably. For technical teams looking to implement this chemistry, the detailed standardized synthesis steps provided below outline the precise conditions, molar ratios, and purification methods required to achieve the reported yields and purity profiles consistently.
- Perform solvent-free microwave reaction between Formula 1 and hydrazine (Formula 2) at 400W for 5-6 minutes to form the pyrazolopyrimidine core (Formula 3).
- React Formula 3 with bromo-pyridine derivative (Formula 4) using K2CO3 in DMF at 60°C to install the side chain, yielding Formula 5.
- Execute deprotection by refluxing Formula 5 with bromomethyl phenyl propane (Formula 6) in dichloromethane to obtain the amine intermediate Formula 7.
- Complete the synthesis by coupling Formula 7 with the piperazine derivative (Formula 8) using K2CO3 in DMF, followed by chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this optimized synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of hazardous reagents like sodium hydride not only reduces the cost of raw materials but also simplifies the regulatory and safety compliance burden associated with handling pyrophoric substances. The shift towards microwave technology for the initial cyclization step represents a significant leap in process intensification, allowing for smaller reactor footprints and faster batch turnover times. This increased velocity in production translates to reducing lead time for high-purity intermediates, enabling suppliers to respond more agilely to fluctuating market demands. Furthermore, the reliance on simple purification techniques like pulping reduces the consumption of expensive chromatography silica and solvents, contributing to a lower environmental footprint and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The substitution of expensive strong bases with low-cost potassium carbonate creates a direct and substantial saving in bill of materials. Additionally, the solvent-free nature of the first step and the use of pulping for purification drastically cut down on solvent purchase and recovery costs. These cumulative efficiencies result in a significantly lower cost per kilogram of the final API, providing a competitive edge in pricing negotiations without compromising on quality standards.
- Enhanced Supply Chain Reliability: By utilizing commercially available starting materials and avoiding exotic catalysts or reagents, the supply chain for this synthesis route is inherently more robust and less prone to disruption. The simplified operational requirements mean that the process can be easily replicated across multiple manufacturing sites, diversifying supply risk. The high yields reported in each step ensure that less starting material is needed to produce a fixed amount of product, buffering against potential shortages of key precursors.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, minimizing solvent use and avoiding heavy metal catalysts that would require complex removal steps. This makes the scale-up from pilot plant to commercial tonnage smoother and more compliant with increasingly stringent environmental regulations. The ability to produce high-purity material with fewer purification steps also reduces the generation of hazardous waste, aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Adavosertib using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, offering clarity on yield expectations, purity profiles, and operational feasibility. Understanding these details is crucial for stakeholders evaluating the viability of this route for long-term supply agreements. The answers reflect the specific advantages of the microwave-assisted and base-optimized protocol described herein.
Q: How does the microwave-assisted step improve the synthesis of Adavosertib?
A: The microwave reaction in Step 1 significantly reduces reaction time to under 6 minutes compared to conventional heating, while achieving yields up to 96% without the need for organic solvents, thereby enhancing industrial operability.
Q: What cost advantages does this route offer over traditional methods?
A: This process replaces expensive strong bases like NaH with low-cost potassium carbonate (K2CO3) for key alkylation steps and utilizes simple pulping purification instead of complex chromatography for intermediates, drastically lowering raw material and processing costs.
Q: What is the purity profile of the final Adavosertib product?
A: The optimized process consistently delivers Adavosertib with an HPLC purity of 99.5% or higher, effectively minimizing halogenated byproducts through controlled reaction conditions and specific base selection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adavosertib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for next-generation oncology therapeutics like Adavosertib. Our team of expert chemists has extensively evaluated the patented microwave-assisted process and confirmed its potential for seamless translation from laboratory bench to industrial reactor. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the patent can be maintained at volume. Our state-of-the-art facilities are equipped with advanced microwave reactors and rigorous QC labs capable of meeting the stringent purity specifications required for clinical and commercial API supply. We are committed to delivering a consistent supply of this vital intermediate to support your drug development timelines.
We invite you to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. By leveraging our process expertise, we can provide a Customized Cost-Saving Analysis tailored to your volume needs, identifying further opportunities for efficiency gains. We encourage potential partners to contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. Let us collaborate to secure a sustainable and cost-effective supply chain for your Wee1 inhibitor programs, ensuring that you have the reliable support needed to bring life-saving medicines to patients faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
