Advanced Metal-Free Synthesis of 1,2-Dihydropyridine Derivatives for Commercial Scale-Up
Introduction to Novel 1,2-Dihydropyridine Synthesis
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly dihydropyridines, which serve as critical pharmacophores in numerous therapeutic agents. A groundbreaking approach detailed in patent CN103420901A introduces a highly efficient pathway for preparing 1,2-dihydropyridine derivatives through the cycloisomerization of 3-aza-1,5-enyne derivatives. Unlike traditional methods that often rely on costly transition metal catalysts, this innovation utilizes simple non-metal catalysts such as DDQ or iodine to drive the reaction with exceptional atom economy. This technological leap addresses long-standing challenges in impurity control and process safety, making it an attractive option for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines. The significance of this method lies not only in its chemical elegance but also in its potential to drastically reduce the environmental footprint associated with heterocycle synthesis.
Furthermore, the versatility of this synthetic route allows for the incorporation of diverse substituents, enabling the rapid generation of compound libraries for drug discovery programs. By starting from cheap and readily available raw materials such as aldehydes, sulfonamides, and terminal alkynes, the process ensures a stable supply chain foundation. The ability to access the less common 1,2-dihydropyridine isomer, as opposed to the more ubiquitous 1,4-isomer, opens new avenues for exploring unique biological activities and structure-activity relationships. For R&D directors focused on novelty and IP positioning, this metal-free strategy offers a distinct competitive advantage in the development of next-generation APIs and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydropyridine derivatives has been dominated by multicomponent reactions or transition metal-catalyzed cyclizations that present significant operational hurdles for large-scale manufacturing. Conventional routes frequently employ palladium, copper, or rhodium catalysts, which introduce severe complications regarding residual metal removal, a critical quality attribute for any pharmaceutical intermediate intended for human consumption. The stringent regulatory limits on heavy metals necessitate additional purification steps, such as scavenging treatments or repeated recrystallizations, which inevitably lower overall throughput and increase production costs. Moreover, many traditional protocols require harsh reaction conditions, including high temperatures or strong bases, which can compromise the integrity of sensitive functional groups and lead to the formation of complex impurity profiles that are difficult to characterize and control.
In addition to the economic and regulatory burdens, the reliance on precious metals creates supply chain vulnerabilities, as the availability and price of these catalysts can fluctuate wildly based on geopolitical factors. The waste streams generated from metal-catalyzed processes also pose environmental compliance challenges, requiring specialized treatment facilities to handle toxic effluents. For procurement managers and supply chain heads, these factors translate into higher risk profiles and reduced flexibility in sourcing strategies. The inability to easily scale these metal-dependent reactions without compromising purity or yield has often forced manufacturers to seek alternative, albeit less efficient, synthetic pathways, highlighting the urgent need for a greener and more sustainable solution like the one proposed in the referenced patent data.
The Novel Approach
The novel methodology described in the patent data revolutionizes this landscape by employing a metal-free cycloisomerization strategy that utilizes inexpensive organic oxidants like DDQ (2,3-dichloro-5,6-dicyano-1,4-benzoquinone) or elemental iodine. This shift from transition metals to non-metal catalysts fundamentally alters the economic equation of dihydropyridine manufacturing, eliminating the need for expensive catalyst loading and the subsequent costly removal processes. The reaction proceeds under relatively mild conditions, typically around 60°C in solvents like THF, which significantly enhances process safety and energy efficiency compared to high-temperature alternatives. By avoiding heavy metals entirely, the resulting product stream is inherently cleaner, simplifying downstream processing and ensuring that the final active pharmaceutical ingredients meet rigorous global purity specifications without extensive remediation.
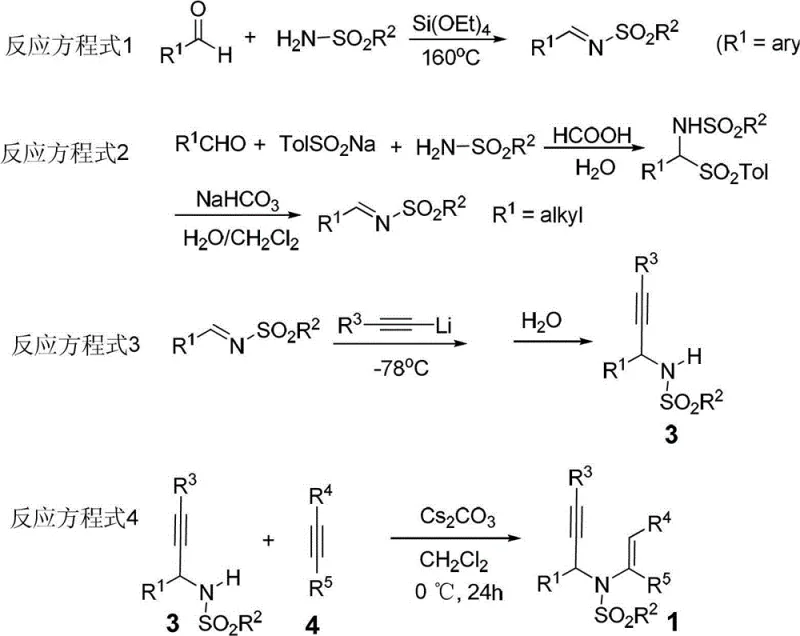
Moreover, the precursor synthesis involves a straightforward condensation of aldehydes and sulfonamides followed by alkynylation, steps that are well-established and easily scalable in standard chemical reactors. The use of cesium carbonate as a base in the precursor formation step ensures high conversion rates while maintaining compatibility with a wide range of functional groups. This robustness allows for the synthesis of diverse 3-aza-1,5-enyne derivatives, which then undergo the key cyclization step with high fidelity. For a reliable pharmaceutical intermediate supplier, adopting this technology means offering clients a product with a superior impurity profile and a more predictable cost structure, thereby strengthening long-term partnerships in the competitive fine chemical market.
Mechanistic Insights into DDQ-Catalyzed Cycloisomerization
The core of this innovative process lies in the oxidative cycloisomerization mechanism facilitated by DDQ, which acts as a hydride acceptor to drive the formation of the dihydropyridine ring system. The reaction initiates with the activation of the 3-aza-1,5-enyne substrate, where the electron-deficient nature of the DDQ molecule promotes the abstraction of a hydride ion from the propargylic position. This generates a reactive cationic intermediate that undergoes an intramolecular nucleophilic attack by the nitrogen lone pair onto the activated alkyne moiety. This cyclization event constructs the six-membered heterocyclic ring with precise regiocontrol, favoring the formation of the 1,2-dihydropyridine scaffold over other possible isomers. The subsequent rearomatization or stabilization steps, mediated by the reduced form of the catalyst, complete the transformation with high atom economy, ensuring that nearly all atoms from the starting materials are incorporated into the final product.
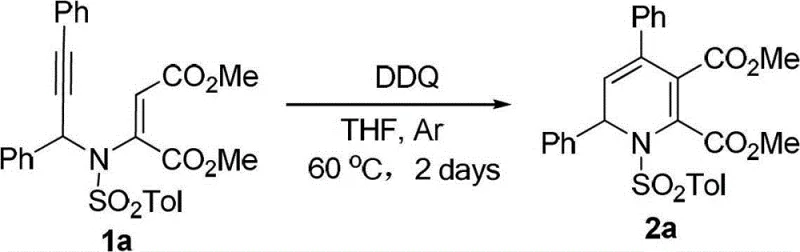
From an impurity control perspective, the mechanism is remarkably clean because it avoids radical pathways that often lead to polymerization or oligomerization side products common in metal-catalyzed alkyne chemistry. The absence of metal coordination complexes means there are no metal-ligand dissociation equilibria to manage, which simplifies the kinetic profile of the reaction and makes it more reproducible on a commercial scale. The specific choice of solvent, such as tetrahydrofuran, plays a crucial role in stabilizing the transition states and ensuring solubility of both the organic substrate and the catalyst. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as catalyst loading (typically 5-20 mol%) and reaction time (1-7 days), to maximize yield and minimize batch-to-batch variability, ensuring consistent quality for high-purity pharmaceutical intermediate batches.
How to Synthesize 1,2-Dihydropyridine Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must follow a disciplined protocol that emphasizes moisture control and precise stoichiometry to achieve the reported high yields. The process begins with the preparation of the 3-aza-1,5-enyne precursor, which requires careful handling of organolithium reagents at low temperatures to ensure successful nucleophilic addition to the imine intermediate. Once the precursor is isolated and characterized, the key cyclization step is performed under an inert argon atmosphere to prevent oxidation of sensitive intermediates by atmospheric oxygen. Detailed standardized synthetic steps, including specific workup procedures and purification techniques like silica gel column chromatography using petroleum ether and ethyl acetate mixtures, are essential for isolating the target 1,2-dihydropyridine derivatives in high purity.
- Prepare the 3-aza-1,5-enyne precursor by reacting N-sulfonyl-allylamine with a terminal alkyne using Cs2CO3 in dichloromethane at 0°C to room temperature.
- Dissolve the 3-aza-1,5-enyne derivative in THF and add a non-metal catalyst such as DDQ (10 mol%).
- Heat the reaction mixture to 60°C under argon atmosphere for 2 days, then purify the resulting 1,2-dihydropyridine via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route presents a compelling value proposition centered on cost stability and operational simplicity. By eliminating the dependency on volatile precious metal markets, manufacturers can lock in more predictable raw material costs, shielding the supply chain from the erratic price fluctuations associated with palladium or platinum group metals. The simplified purification workflow, which no longer requires specialized metal scavengers or extensive washing protocols, translates directly into reduced cycle times and lower utility consumption per kilogram of product. This efficiency gain allows for a more agile response to market demand, enabling suppliers to maintain healthier inventory levels and shorter lead times for critical pharmaceutical intermediates without compromising on quality standards.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of transition metal catalysts, which are among the most expensive reagents in organic synthesis. Without the need for these metals, the bill of materials is significantly lowered, and the associated costs of waste disposal for heavy metal contaminants are virtually eliminated. Additionally, the use of commodity chemicals like DDQ and cesium carbonate, which are produced at scale globally, ensures a steady and affordable supply of catalysts. This structural cost advantage allows manufacturers to offer more competitive pricing to downstream API producers while maintaining healthy margins, fostering a more sustainable economic model for fine chemical production.
- Enhanced Supply Chain Reliability: The reliance on widely available organic reagents and solvents like THF and dichloromethane reduces the risk of supply disruptions that often plague specialized catalyst supply chains. Since the starting materials such as aromatic aldehydes and sulfonamides are bulk commodities with multiple global sources, the risk of single-source dependency is minimized. This diversification of the supply base enhances the resilience of the production network, ensuring continuous operation even during periods of logistical stress or regional shortages. For supply chain heads, this reliability is paramount for meeting the just-in-time delivery requirements of major pharmaceutical clients who cannot afford interruptions in their own manufacturing schedules.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, specifically high atom economy and the absence of toxic metals, align perfectly with increasingly stringent environmental regulations worldwide. Scaling this process from gram to tonnage does not introduce new hazardous waste streams, simplifying the permitting process for facility expansion and reducing the burden on wastewater treatment plants. The mild reaction conditions further contribute to energy savings, as there is no need for extreme heating or cooling beyond standard industrial capabilities. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important in vendor selection criteria for top-tier pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 1,2-dihydropyridine synthesis technology, providing clarity for stakeholders evaluating its potential integration into their supply chains. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of adopting this method for specific project requirements and for anticipating any potential technical hurdles during technology transfer.
Q: What are the advantages of using DDQ over transition metal catalysts for this cyclization?
A: Using DDQ eliminates the need for expensive and toxic transition metals, simplifying purification and reducing heavy metal residue risks in pharmaceutical intermediates.
Q: What is the typical yield for the cycloisomerization of 3-aza-1,5-enyne derivatives?
A: According to patent data, the reaction can achieve high isolated yields, such as 92% for specific phenyl-substituted derivatives under optimized conditions.
Q: Can this method tolerate various functional groups on the aldehyde starting material?
A: Yes, the method demonstrates broad substrate scope, tolerating substituents like methoxy, halogens, and nitro groups on the aromatic rings of the starting aldehydes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Dihydropyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cycloisomerization technology and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 1,2-dihydropyridine derivatives meets the exacting standards of the international pharmaceutical industry. We are committed to leveraging our technical expertise to optimize this green synthesis route, delivering high-quality intermediates that support the development of life-saving medications while adhering to the highest principles of sustainability and safety.
We invite forward-thinking partners to collaborate with us to explore how this advanced manufacturing capability can enhance your product portfolio and reduce your overall cost of goods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and secure your supply chain for the future.
